Clear Sky Science · en
Simple and efficient co-synthesis of gold nanoparticles and nanoclusters for HPV-16 detection
Why detecting a hidden virus matters
Cervical cancer is one of the few cancers we can largely prevent if we find the warning signs early. A major culprit is human papillomavirus type 16 (HPV‑16), a common sexually transmitted virus that can quietly damage cells for years before symptoms appear. Vaccination helps, but many women around the world remain unvaccinated or lack access to regular screening. This study describes a simple lab method that turns tiny particles of gold into a low-cost sensor, designed to spot HPV‑16’s genetic material quickly and without bulky instruments.
Turning gold into a disease detector
The researchers set out to build a test that could be both affordable and sensitive enough to pick up small amounts of HPV‑16 DNA, the virus’s genetic code. Instead of relying on complex equipment, they used gold at the nanoscale—particles so small they behave differently from ordinary metal. These gold nanoparticles and even smaller "nanoclusters" can change color and glow under light in very specific ways. By linking them to a short piece of DNA that recognizes part of the HPV‑16 genome, the team created a system where the presence of the virus triggers a visible color shift and a strong light signal. This dual response makes it easier to tell positive samples from negative ones, even for non-experts.
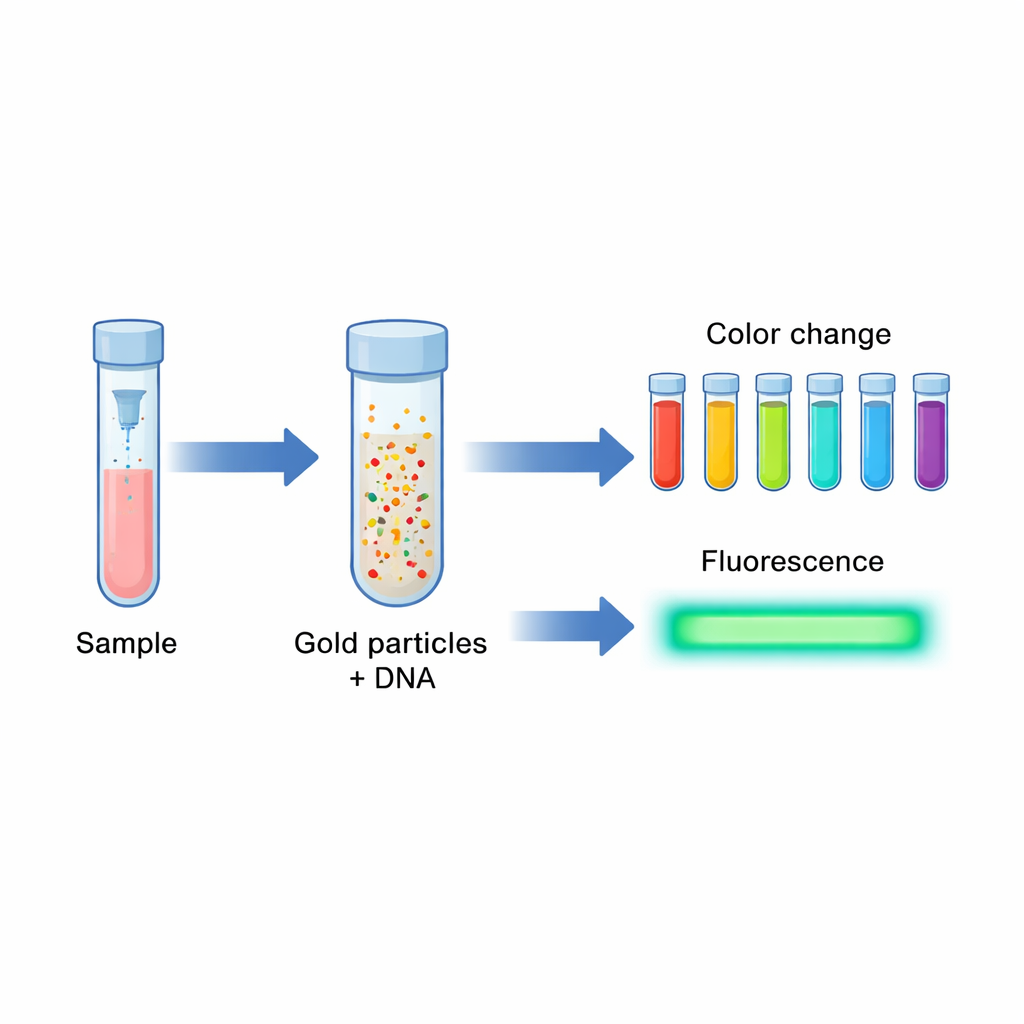
One simple recipe, two kinds of gold
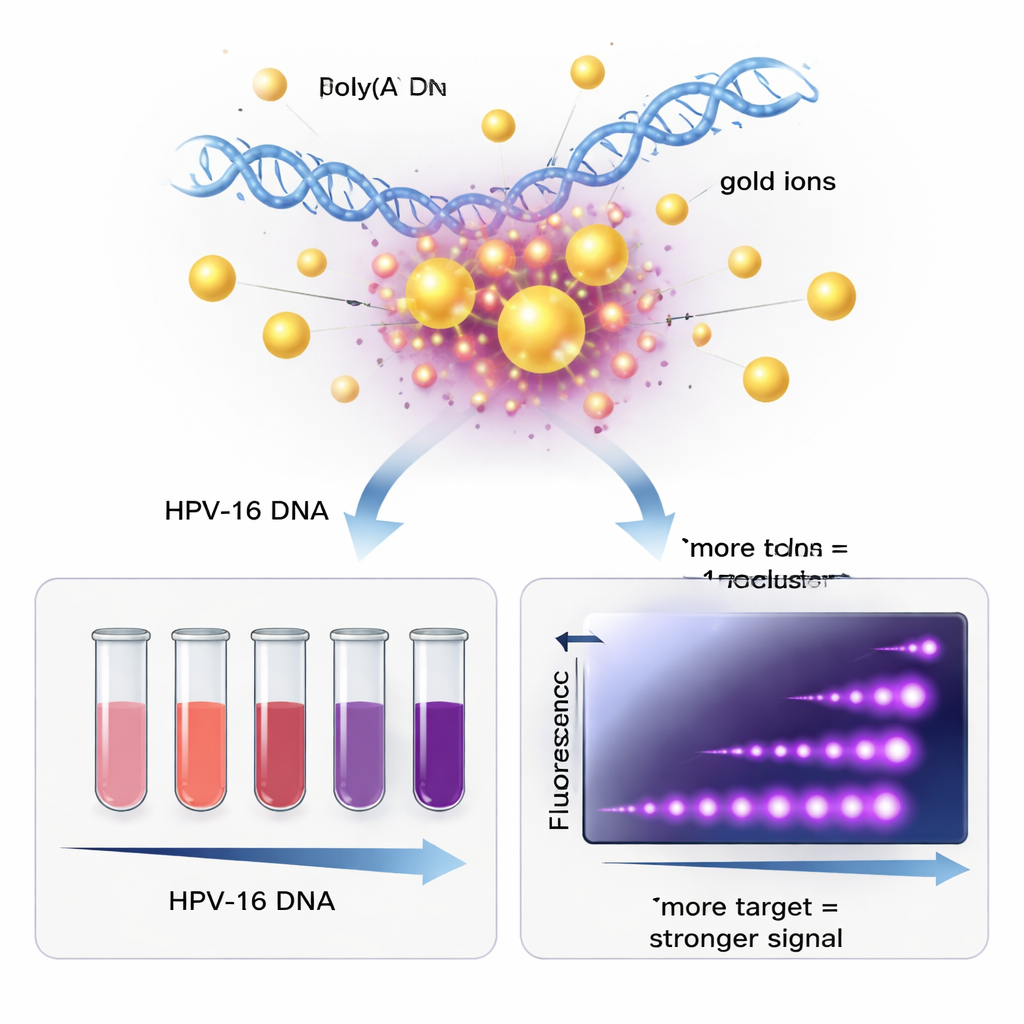
A key innovation of the work is that both the larger gold nanoparticles and the ultrasmall nanoclusters are made together in a single step. The team used a short DNA strand rich in the base adenine—essentially a tail of 20 "A" letters—mixed with a gold salt and a gentle chemical called HEPES. The adenine tail naturally grabs onto gold ions and helps them clump into either slightly larger particles that give a strong red color, or very tiny clusters that shine with blue‑violet fluorescence. Because the same DNA strand also contains a sequence that binds specifically to the HPV‑16 L1 gene, each gold particle ends up coated with many copies of a "probe" that can latch onto the virus’s DNA if it is present.
Reading the virus in color and light
To test the sensor, the researchers used a piece of circular DNA (a plasmid) carrying the HPV‑16 L1 gene, as well as DNA extracted from real patient samples. They heated the viral DNA briefly so its double helix opened, then cooled it down with the gold–DNA probes present. When the probe sequence found its matching HPV‑16 target, it formed a stable, double‑stranded structure attached to the gold. In tubes with the correct target, the solution stayed a clear red and produced a strong fluorescent signal. In control tubes without the right HPV‑16 sequence, the gold particles clumped together, the color shifted toward purple, and the light signal remained weak. By measuring how much the color and fluorescence changed, the team could estimate how much viral DNA was present over a useful range of concentrations.
Extra trick: gold that acts like an enzyme
The gold nanoparticles in this sensor also mimic the activity of certain natural enzymes. When the researchers added a common laboratory dye (TMB) and hydrogen peroxide, non‑clumped gold nanoparticles helped convert the dye into a deep blue form. The more HPV‑16 DNA bound to the probes, the more stable and separated the gold particles remained, and the stronger this blue color became. This gave the test a second, independent color readout—one based not just on the particles’ natural color, but on their enzyme‑like behavior. Using this effect, the team could detect HPV‑16 DNA at similarly low levels, confirming that both color pathways tell the same story.
What this could mean for future screening
Overall, the study shows that a simple mixture of gold, buffer, and short DNA strands can be turned into a reliable sensor for a high‑risk cancer virus. The method detects HPV‑16 without extra labels, complex amplification steps, or expensive instrumentation, yet still reaches low detection limits and clearly separates positive from negative samples. While more validation in larger patient groups and real‑world clinics is still needed, this dual‑mode gold sensor points toward future screening tools that could be run in modest laboratories—or potentially near the point of care—helping to bring early cervical cancer detection within reach of more women globally.
Citation: Saleh, M.A., Hosseinkhani, S., Nikkhah, M. et al. Simple and efficient co-synthesis of gold nanoparticles and nanoclusters for HPV-16 detection. Sci Rep 16, 4854 (2026). https://doi.org/10.1038/s41598-026-35246-6
Keywords: HPV-16, cervical cancer screening, gold nanoparticles, biosensor, nanodiagnostics