Clear Sky Science · en
Deglycosylation induces a novel distal conformation in the mannose receptor CD206
Why sugar coatings on immune sensors matter
Our immune cells rely on molecular "antennae" to detect threats such as microbes and cancer cells. One of these antennae, called the mannose receptor CD206, helps immune cells grab sugar-decorated molecules and even home in on tumors. Like many proteins, CD206 itself is covered with small sugar chains. This study asks a deceptively simple question with big implications: what happens to CD206’s shape and function when those sugars are removed?
A flexible arm that reads sugary signals
CD206 sits on the surface of certain immune cells, especially macrophages that patrol tissues and tumors. It has a long, jointed structure that reaches out from the cell membrane, ending in several “lectin” segments that recognize sugars on passing molecules. CD206 can also be shed into the bloodstream, where its levels correlate with inflammatory and fibrotic diseases, making it a potential marker for illness. The protein is heavily decorated with N-linked sugar chains, which earlier work showed can influence how well different segments of the receptor recognize sugars. Many of these sugar sites are far from the main binding pockets, hinting that they might act more like distant switches than simple blockers.
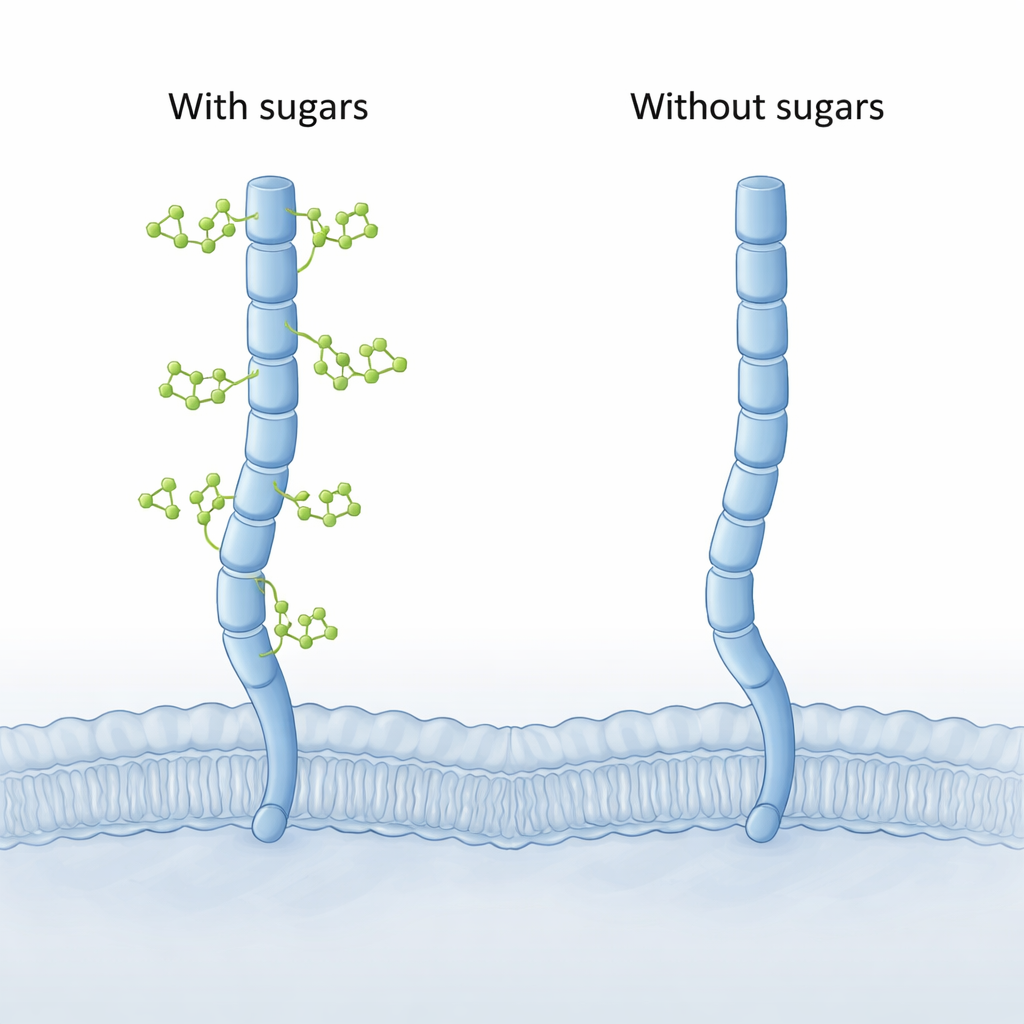
Computer models reveal a hidden bend
Because the full 3D structure of CD206 has not yet been captured experimentally, the authors turned to modern computational tools. They first used AlphaFold to build full-length models of the receptor, then ran long, detailed molecular dynamics simulations of CD206 with and without its attached sugars. Both versions behaved as an extended molecular arm, but the “sugar-free” form showed a striking new motion at its far end. The last two lectin segments (called CTLD7 and CTLD8) could swing inward to form a concave, inward-bent tip, a shape never seen in the sugar-decorated version, which stayed more outward-curved, or convex.
How a small change reshapes motion
To understand this difference, the team analyzed the dominant motions in their simulations. They found that the main movement distinguishing the two forms was this bending of the distal tip, and that it only appeared when sugars were missing. Removing sugars generally made the receptor more flexible, especially in the distant lectin segments and, to a lesser degree, near the N-terminal end. Correlation maps of how different parts of the protein move together showed that, without sugars, the last two lectin segments move in a more concerted way and are more tightly coupled to other regions. In essence, the sugars do not just sit on the surface; they help tune how the entire arm flexes and communicates along its length.
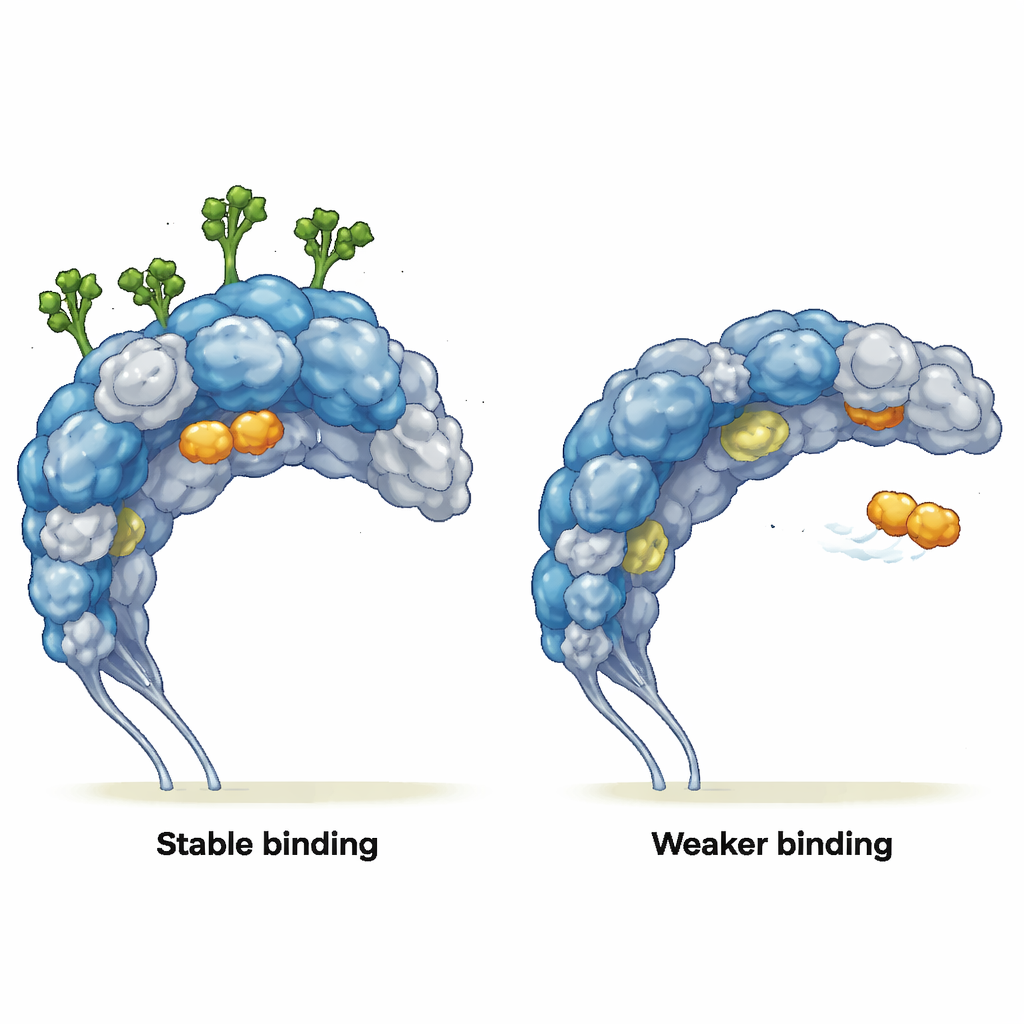
Changing the way partners bind
The authors then asked how this altered motion affects real binding partners. They simulated interactions with two ligands: a simple mannose-like sugar (MMA), which fits into a well-defined pocket, and a tumor-homing peptide called mUNO, which targets CD206-positive tumor-associated macrophages. When sugars were present, MMA stayed snugly in its binding pocket for the full simulation, maintaining key contacts, even though the nearby protein structure hardly changed. Without sugars, MMA escaped the pocket, skimming across the protein surface and forming only brief, superficial contacts. For mUNO, the glycosylated receptor displayed several stable binding poses with frequent, long-lasting contacts. In the deglycosylated receptor, mUNO’s binding was weaker and more scattered, and it also began to explore regions that are normally blocked by sugar chains, revealing new, previously hidden interaction sites.
What this means for disease and therapy
Taken together, these results show that the sugars attached to CD206 act as remote regulators of its shape and stickiness. By restricting certain bends and organizing how its segments move together, glycosylation helps the receptor hold onto some ligands more tightly and steer others toward preferred docking spots, while also shielding parts of its surface. In diseases like cancer, where overall sugar patterns on proteins are often altered, CD206 may adopt different shapes and binding behaviors, changing how immune cells sense their environment. Understanding this sugar-dependent “conformational switch” not only clarifies how CD206 works, but also suggests new ways to design drugs or imaging agents that exploit its changing landscape in tumors and other pathological conditions.
Citation: Alvarez, G., Di Lella, S., Pickholz, M. et al. Deglycosylation induces a novel distal conformation in the mannose receptor CD206. Sci Rep 16, 5239 (2026). https://doi.org/10.1038/s41598-026-35240-y
Keywords: mannose receptor, protein glycosylation, immune recognition, tumor-associated macrophages, molecular dynamics