Clear Sky Science · en
Chitosan-dextran sulfate nanocapsules for enhanced tigecycline efficacy against non-typhoidal Salmonella enterica
Why this matters for food safety
Most people know Salmonella as a nasty cause of food poisoning, often linked to undercooked poultry. Fewer realize that some strains have become so resistant to antibiotics that even powerful “last‑resort” drugs can fail. This study explores a nanotechnology-based way to rescue one such drug, tigecycline, by packaging it inside tiny sugar-based capsules that help it slip into infected cells and shut down key resistance mechanisms in Salmonella.
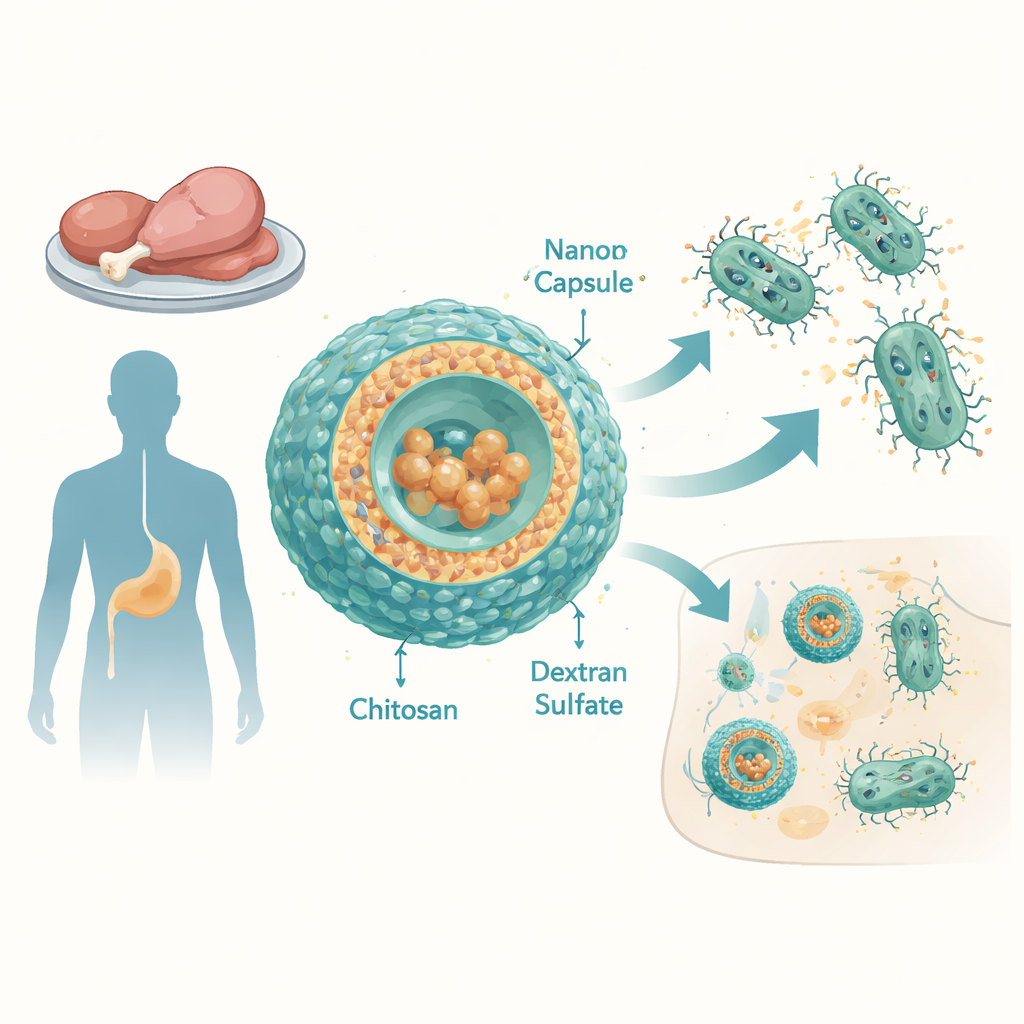
The growing threat from tough Salmonella
Non-typhoidal Salmonella (NTS) serovars are a leading cause of diarrhea and bloodstream infections worldwide, especially in low- and middle-income countries. The researchers examined 12 Salmonella enterica strains taken from chicken and duck meat in Egypt. These strains were resistant to many commonly used antibiotics, including penicillins, cephalosporins, tetracyclines and others, and showed high multiple-antibiotic-resistance scores. Even tigecycline, a broad-spectrum drug reserved for difficult cases, worked poorly: the bacteria could tolerate very high concentrations of it. A major reason was overactive “efflux pumps”—molecular machines that sit in the bacterial membrane and constantly pump antibiotics back out of the cell.
Building a smarter drug capsule
To overcome this, the team designed nanoscale capsules made from two naturally derived polymers: chitosan, obtained from chitin (the material in shellfish shells), and dextran sulfate, a branched sugar molecule. By carefully tuning the proportions of these components, they produced stable particles about 100–150 nanometers across, with a positive surface charge that helps them interact with bacterial and host cell membranes. Tigecycline was loaded into these particles with remarkably high efficiency—essentially all of the drug ended up inside the capsules. In lab tests mimicking conditions in the bloodstream, the capsules released most of their tigecycline within a few hours, suggesting they could deliver a strong burst of antibiotic at infection sites.
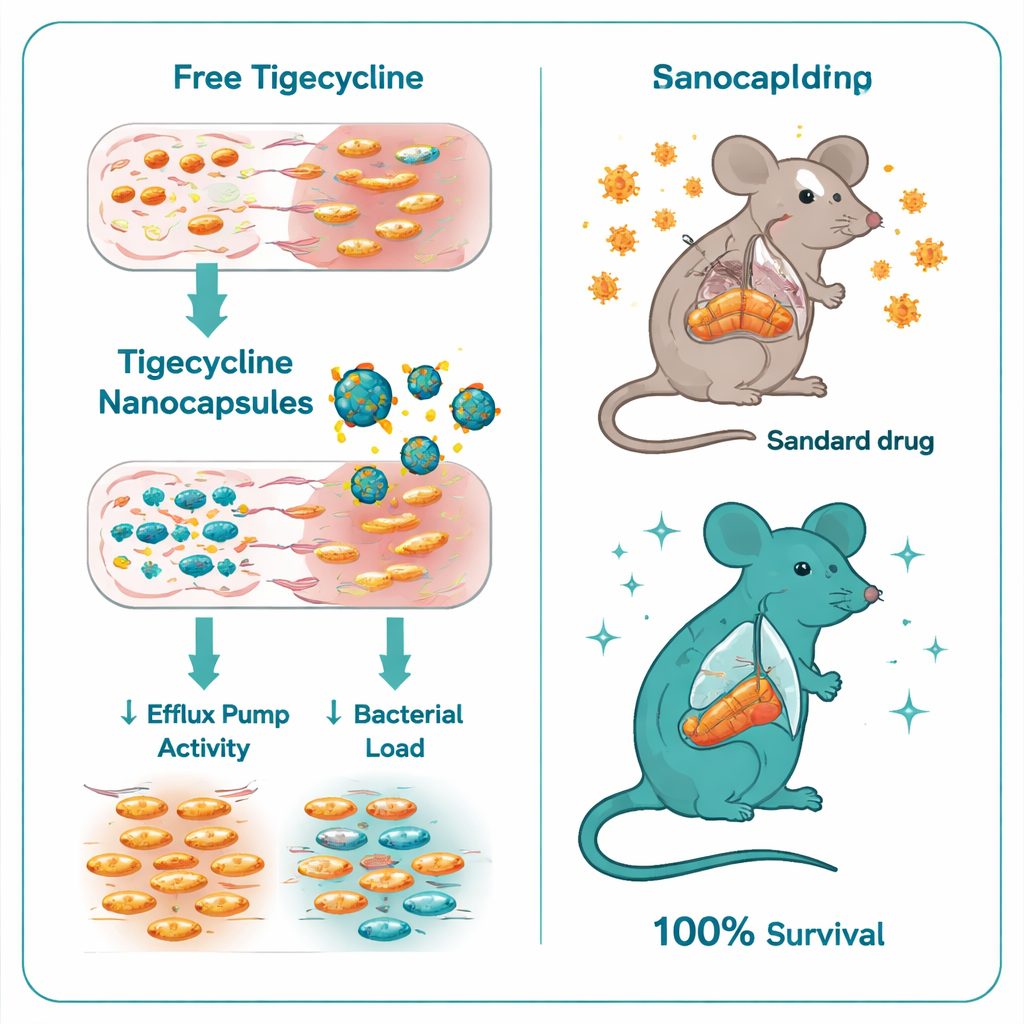
Turning down bacterial defenses
When the researchers compared plain tigecycline with the tigecycline-loaded chitosan–dextran sulfate nanocapsules, the difference was striking. The minimum drug concentration needed to stop bacterial growth dropped from 32–128 micrograms per milliliter for plain tigecycline down to just 0.5–1 microgram per milliliter with the nanocapsules—a six- to seven-fold improvement. Time-kill experiments showed that the encapsulated form killed Salmonella much more rapidly and thoroughly. At the genetic level, the nanocapsules also quieted key resistance regulators: expression of the ramA and acrB genes, which help control and form major efflux pumps, fell several-fold in treated bacteria. In other words, the capsules did not just deliver more drug; they helped switch off one of the bacteria’s main escape routes.
Protecting infected mice from lethal disease
The team then tested the new formulation in a mouse model of severe Salmonella Typhimurium infection. Untreated infected mice all died within eight days. Mice receiving standard tigecycline fared better but still suffered high mortality, with only 40% surviving. In sharp contrast, every mouse treated with the tigecycline-loaded nanocapsules survived. These animals had far fewer bacteria in their livers and intestines, blood tests that were closer to normal liver and kidney function, and much milder tissue damage upon microscopic examination. Even the “empty” chitosan–dextran sulfate capsules, without antibiotic, offered some protection, likely because chitosan itself has modest antimicrobial and anti-inflammatory effects, but the combination with tigecycline was clearly superior.
What this could mean for future treatments
For non-specialists, the central message is that packaging an existing antibiotic inside a smart, biocompatible nanocapsule can restore its punch against highly resistant Salmonella. By helping the drug reach bacteria hiding inside cells and by dialling down the pumps that normally kick the drug out, the chitosan–dextran sulfate system turned a struggling last-resort antibiotic into a highly effective treatment in mice, with 100% survival. While more work is needed before this approach can be used in people or in farm animals, it offers a promising path to extend the useful life of important antibiotics and improve the safety of our food supply without having to invent entirely new drugs from scratch.
Citation: Omar, M.R., Saeed, A.A., Malhat, S.M. et al. Chitosan-dextran sulfate nanocapsules for enhanced tigecycline efficacy against non-typhoidal Salmonella enterica. Sci Rep 16, 5016 (2026). https://doi.org/10.1038/s41598-026-35229-7
Keywords: Salmonella, antibiotic resistance, nanoparticles, drug delivery, tigecycline