Clear Sky Science · en
Synthesis and investigation of linear and nonlinear optical parameters of hafnium nitrosalicylate complex
Why a New Light-Bending Material Matters
From faster internet to sharper medical imaging and smarter sensors, many emerging technologies depend on materials that can control light with great precision. This study introduces a newly made compound built from the metal hafnium and an organic molecule called 5-nitrosalicylic acid. By carefully designing and testing this material, the researchers show that it can strongly absorb ultraviolet (UV) light while staying largely transparent to visible light—and it also bends and controls intense laser beams in unusual ways. These combined abilities make it a promising candidate for future photonic devices that switch, guide, and sense light without relying on electronics.
Building a Stable Light-Responsive Compound
The team first tackled a practical challenge: how to reliably make a hafnium-based compound that is both stable and easy to handle. They reacted hafnium salts with 5-nitrosalicylic acid under controlled conditions, optimizing temperature, ratios, and purification steps to reach a solid yield of about two-thirds of the starting materials. The result was a white crystalline solid known as tetrakis (5-nitrosalicylate) hafnium (IV). Tests showed that this compound does not break down until temperatures above 300 °C, an important feature for devices that must operate in harsh or long-term conditions. The organic part of the molecule wraps around the hafnium atom like a claw, forming ring-like structures called chelates that lock the metal in place and boost stability.
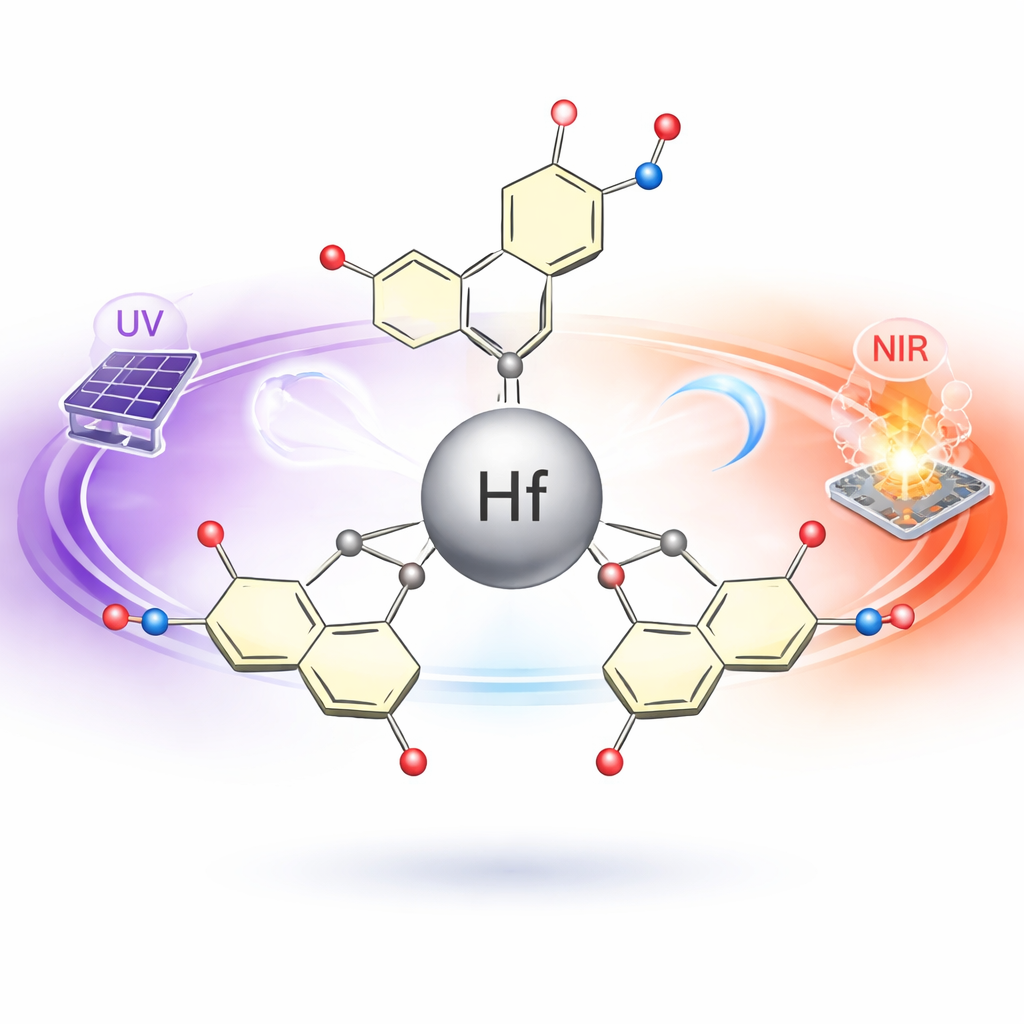
Peeking Inside with Structural Probes
To verify what they had made, the researchers combined several standard yet powerful techniques. Infrared spectroscopy was used to identify how chemical bonds vibrate, confirming that the organic rings were indeed attached to the metal in the expected way. X-ray diffraction patterns revealed that the material forms a well-ordered crystal, distinct from the starting ingredients, with a regular arrangement of hafnium centers and their surrounding ligands. Energy-dispersive X-ray analysis further confirmed that the elements—carbon, nitrogen, oxygen, and hafnium—are spread evenly throughout the sample. Complementary computer calculations helped map how electrons are distributed in the molecule, showing that when the compound absorbs light, electrons tend to move from the surrounding organic rings toward the central hafnium atom.
How the Material Handles Everyday Light
The next step was to learn how the compound interacts with ordinary, relatively weak light. Using a technique called spectroscopic ellipsometry, the team measured how much the material refracts (bends) and absorbs light across ultraviolet, visible, and near-infrared wavelengths. They found a striking dual behavior. In the UV region, the compound shows strong absorption linked to so-called ligand-to-metal charge transfer: light energy kicks electrons from the organic shell into hafnium-related states. In contrast, at visible and near-infrared wavelengths the material behaves more like a clear dielectric: it has a stable refractive index and much lower absorption, meaning it can transmit light efficiently. From these data, they determined that the energy gap between filled and empty electronic states is wider in the hafnium complex than in the free organic molecule, which generally contributes to stability and UV selectivity.
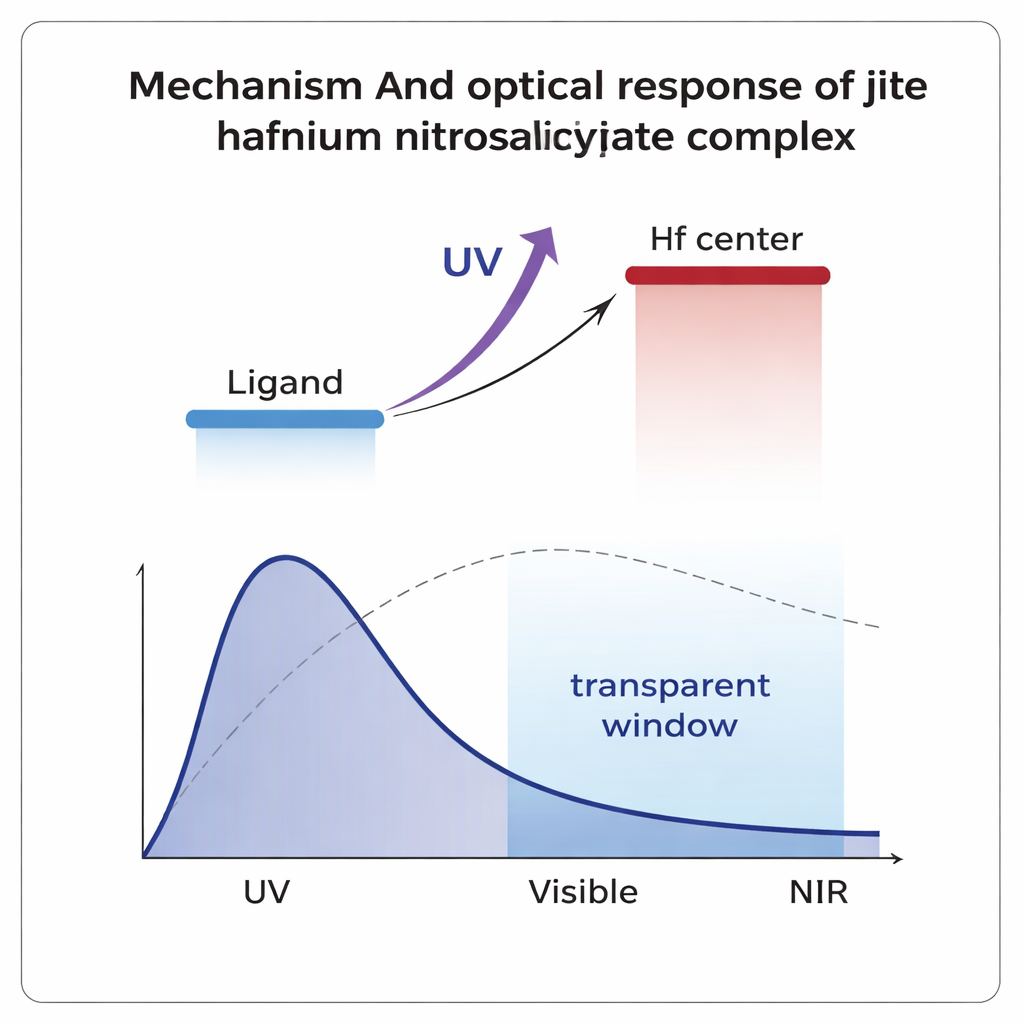
What Happens Under Intense Laser Light
When light becomes very intense—such as in focused laser beams—some materials respond in nonlinear ways: their transparency and refractive index start to depend on the light’s strength. Using a sensitive method called the Z-scan technique with a green laser, the researchers showed that the hafnium complex has a strong third-order nonlinear response. The material slightly defocuses the laser beam and also exhibits two-photon absorption, in which the material absorbs two photons at once. These effects were not seen in the free organic ligand alone, highlighting the crucial role of hafnium. When benchmarked against common reference liquids and oxides, the new compound shows nonlinear strengths orders of magnitude higher, and quantitative figures of merit suggest it could function in “all-optical switches” that use light to control light without converting signals back into electricity.
From Lab Sample to Future Devices
Overall, this work shows that by carefully choosing and arranging metal atoms and organic molecules, scientists can sculpt how a material responds to different colors and intensities of light. The hafnium nitrosalicylate complex acts as a UV-hungry absorber while remaining largely transparent in the visible range, and it strongly reshapes powerful laser beams. For non-specialists, the takeaway is that such materials could underpin next-generation photodetectors that see only UV, coatings that shield sensitive components from harmful radiation, and ultrafast optical switches that move information using photons instead of electrons. This study is an early but important step toward turning these concepts into practical optical and photonic technologies.
Citation: Azadegan, A., Jafari, A., Nikoo, A. et al. Synthesis and investigation of linear and nonlinear optical parameters of hafnium nitrosalicylate complex. Sci Rep 16, 4820 (2026). https://doi.org/10.1038/s41598-026-35221-1
Keywords: hafnium complex, ultraviolet photonics, nonlinear optics, metal–organic materials, all-optical switching