Clear Sky Science · en
Enhanced crystal violet adsorption from aqueous solutions on ozone-aged polyvinyl chloride microplastics
Why tiny plastic bits and a purple dye matter
Most of us think of plastic waste as bottles and bags bobbing on the ocean, but much of it breaks down into particles smaller than a grain of sand, called microplastics. This study looks at one common plastic, polyvinyl chloride (PVC), and a vivid purple dye named crystal violet, a pollutant used in industries from textiles to labs. The twist is that the same ozone used to clean drinking water can change these microplastics in ways that make them better at grabbing and carrying this toxic dye through rivers, lakes, and treatment plants.
From everyday plastics to invisible specks
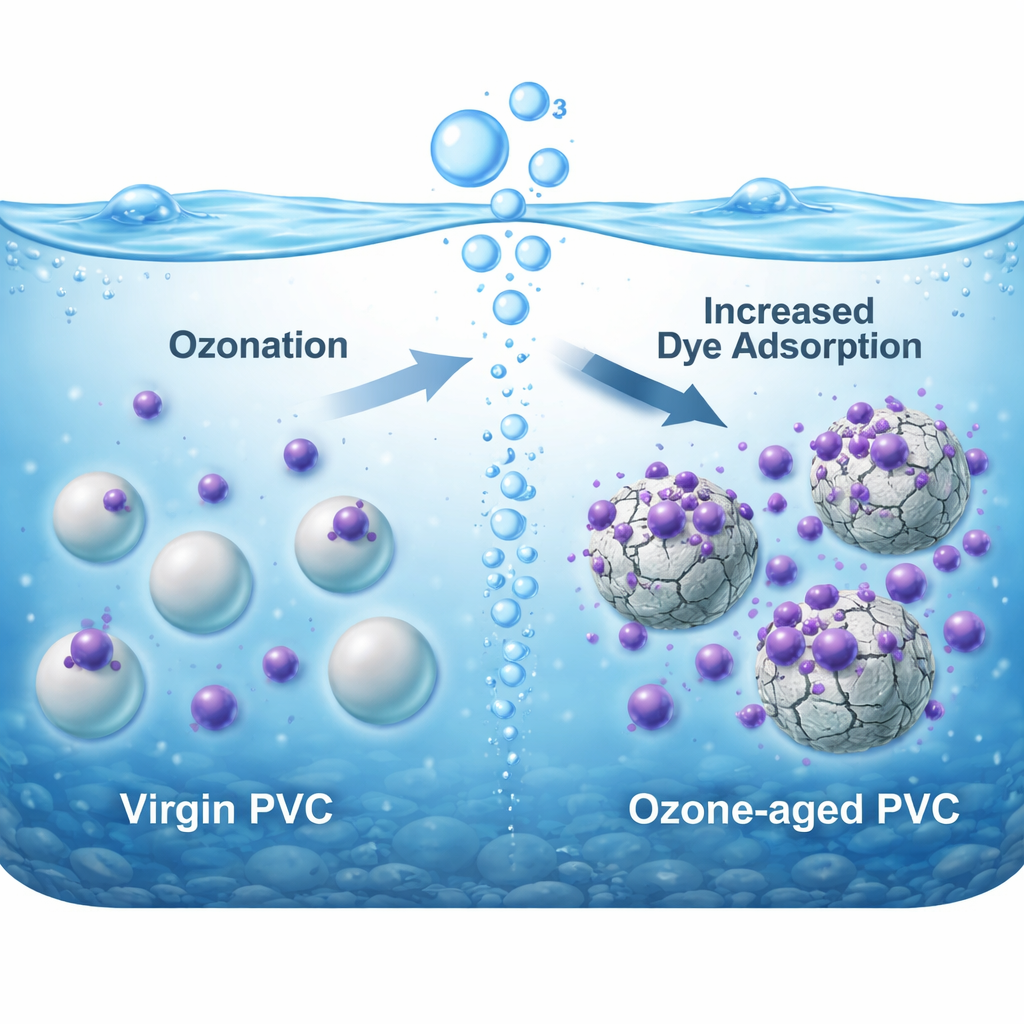
Plastics are designed to last, which is why they are so useful—and so troublesome. Over time, sunlight, heat, and chemicals break large PVC items into microplastics that now show up in freshwater and the sea. These tiny particles can attract other pollutants, including dyes, pesticides, and metals, turning them into mobile packages of contamination. Yet many experiments have assumed that these plastics were fresh from the factory, not aged by real-world conditions. The authors set out to mimic one specific aging process—exposure to ozone, a strong oxidant commonly used in water treatment—to see how it changes PVC microplastics and their ability to bind crystal violet.
Simulating years of wear with an hour of ozone
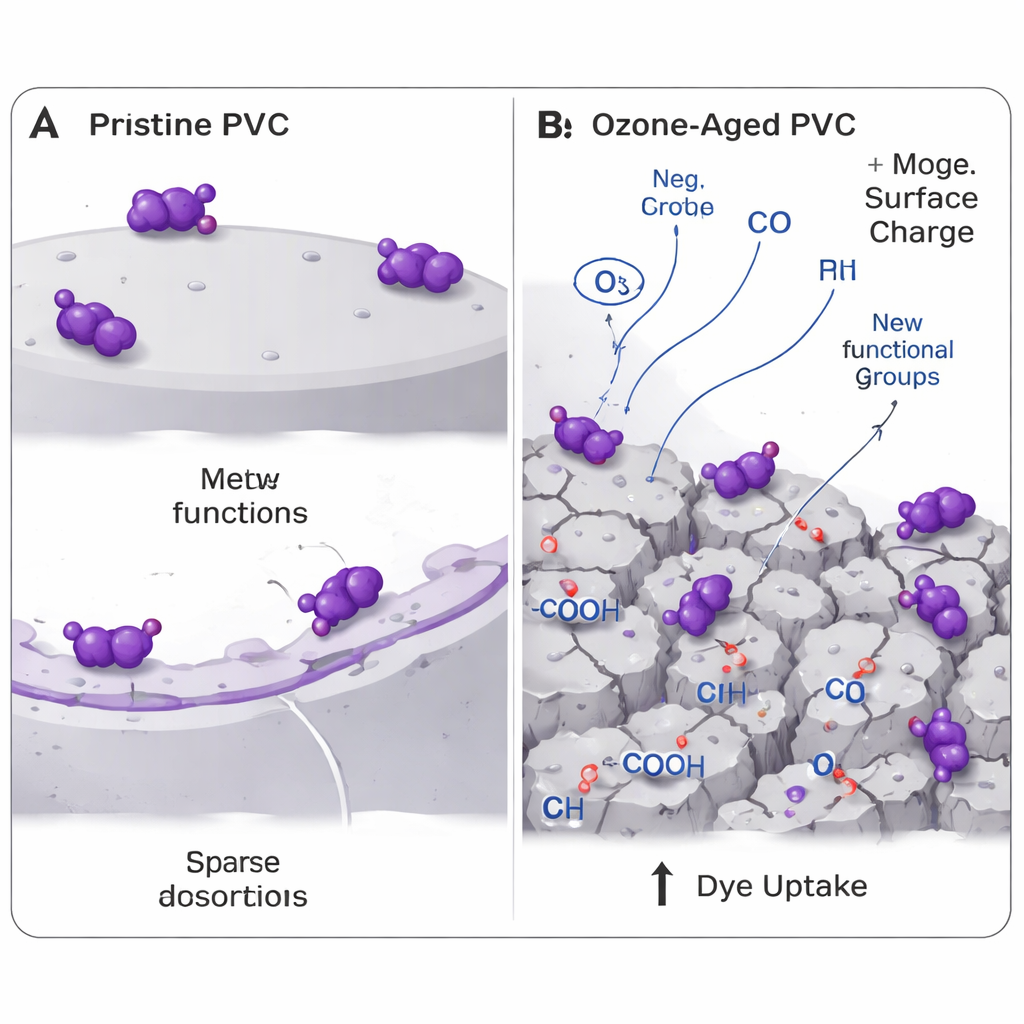
To recreate aging, the researchers suspended PVC microplastic particles in water and bubbled ozone through the mixture for an hour. They then compared the “virgin” and “ozone-aged” particles using a suite of techniques that examine chemical bonds, surface shape, and particle size. Infrared measurements showed that ozone stripped away some chlorine-based groups typical of PVC and introduced more oxygen-bearing groups such as carbonyl and carboxyl, chemical features that can grab onto charged molecules like dyes. Electron microscope images revealed that once-smooth particles developed cracks, pits, and pores, while size measurements showed a modest drop in average particle diameter, pointing to surface erosion and fragmentation. The electrical charge at the particle surface became more negative, a key change because crystal violet is positively charged.
How aging boosts the dye-carrying power of plastics
These physical and chemical makeovers had a clear effect on how the plastics interacted with crystal violet in water. When both virgin and aged PVC were mixed with dye solutions, the untreated particles removed about half of the dye, while the ozone-aged ones removed more than three-quarters. In other words, ozone turned PVC from a relatively indifferent surface into a much stickier one for this pollutant. Mathematical models of how the dye attached over time suggested that the process was dominated by relatively weak, reversible interactions spread over the surface, rather than the formation of strong chemical bonds. At equilibrium, the data fit best with a model in which dye molecules form a single layer on a limited number of similar sites, with a maximum capacity of about 5.55 milligrams of dye per gram of aged plastic—modest compared with high-tech filters, but significant for a pollutant that can cling to trillions of particles.
Water conditions that change the story
The team also tested how typical water conditions affected dye uptake by aged PVC. Under acidic conditions, the plastics removed the most crystal violet; at neutral pH the performance dipped slightly, and in alkaline water it fell sharply, reflecting shifts in surface charge and competition from hydroxide ions. Increasing the saltiness of the water, measured as electrical conductivity, also reduced dye adsorption, as dissolved ions crowded out dye molecules at the surface and weakened electrical attraction. Temperature had a more complex effect: the highest dye removal occurred at cold conditions (4 °C), dropped at room temperature, then partly rebounded at warmer levels, suggesting a tug-of-war between how strongly the dye prefers the surface and how quickly molecules move and collide.
What this means for pollution and cleanup
Although the ozone-aged PVC did not match engineered materials like activated carbon in raw capacity, the results point to an unsettling conclusion. Because microplastics are widespread, long-lived, and easily transported, even a modest ability to bind pollutants matters when multiplied across countless particles. Ozone, used to purify water and kill germs, can unintentionally turn PVC microplastics into more effective carriers for toxic dyes like crystal violet. These altered particles may ferry pollutants through treatment plants and natural waters, and into organisms that swallow them, releasing their chemical cargo in new environments. The study suggests that managing microplastic pollution—and the ways we treat our water—must account not only for the plastics we can see, but also for how invisible aging processes change what those plastics can carry.
Citation: Esmaeili Nasrabadi, A., Babaei, N., Bonyadi, Z. et al. Enhanced crystal violet adsorption from aqueous solutions on ozone-aged polyvinyl chloride microplastics. Sci Rep 16, 4859 (2026). https://doi.org/10.1038/s41598-026-35218-w
Keywords: microplastics, polyvinyl chloride, ozonation, crystal violet dye, water pollution