Clear Sky Science · en
Enhanced electrochemical behavior of Co3O4-modified MoNi-layered double hydroxide nanocomposites for pseudocapacitive applications
Powering Future Gadgets
From electric cars to wearable electronics, our lives increasingly depend on devices that need to charge quickly yet run for a long time. Supercapacitors are one class of energy storage device that can gulp down charge in seconds, but they typically store less energy than batteries. This paper explores a new recipe for the heart of a supercapacitor—the electrode—in order to store more energy without giving up fast charging or long life, bringing us closer to slimmer phones, more responsive electric vehicles, and steadier renewable power systems.
Why Supercapacitors Need Better Materials
Unlike everyday batteries, which rely on slow chemical changes, supercapacitors store energy mostly on their surfaces. That makes them excellent for quick bursts of power and for surviving tens of thousands of charge–discharge cycles. The catch is that today’s commercial supercapacitors usually hold less energy per unit weight than batteries, limiting their use when space and mass are at a premium. To overcome this, scientists turn to so‑called “pseudocapacitive” materials, which add fast, reversible chemical reactions on top of simple surface charging. The challenge is to find materials that offer many active reaction sites, let ions slip in and out easily, and remain stable over years of use.
Building a Three-Metal Electrode
The authors focus on a family of substances called layered double hydroxides, or LDHs. These are stack‑like structures made of positively charged metal layers separated by water and charge‑balancing ions. LDHs naturally offer a large internal surface area and many chemical sites where energy‑storing reactions can occur. In this work, the team creates an LDH that combines nickel and molybdenum (MoNi‑LDH), then decorates it with a small amount of cobalt oxide (Co3O4). The result is a hybrid material in which nickel, molybdenum, and cobalt can all take part in rapid redox reactions—the electron‑swapping processes that underlie pseudocapacitance.
From Powder to Porous Network
To assemble these ingredients, the researchers use a water‑based process called hydrothermal synthesis. First, they grow Co3O4 as fine, wire‑like crystals. Next, they prepare the MoNi‑LDH as nearly spherical particles. Finally, they combine the cobalt oxide with the LDH solution and heat it so that the nanowires attach to and penetrate the spheres. Microscopy images show that the underlying LDH spheres largely keep their shape while becoming threaded with Co3O4 wires. Measurements of gas adsorption confirm that this composite has more surface area and a richer mix of pore sizes than either material alone, giving ions more channels to enter, move, and react. Chemical tests also verify that nickel, molybdenum, cobalt, and oxygen are cleanly incorporated into the structure.
Testing Charge Storage Performance
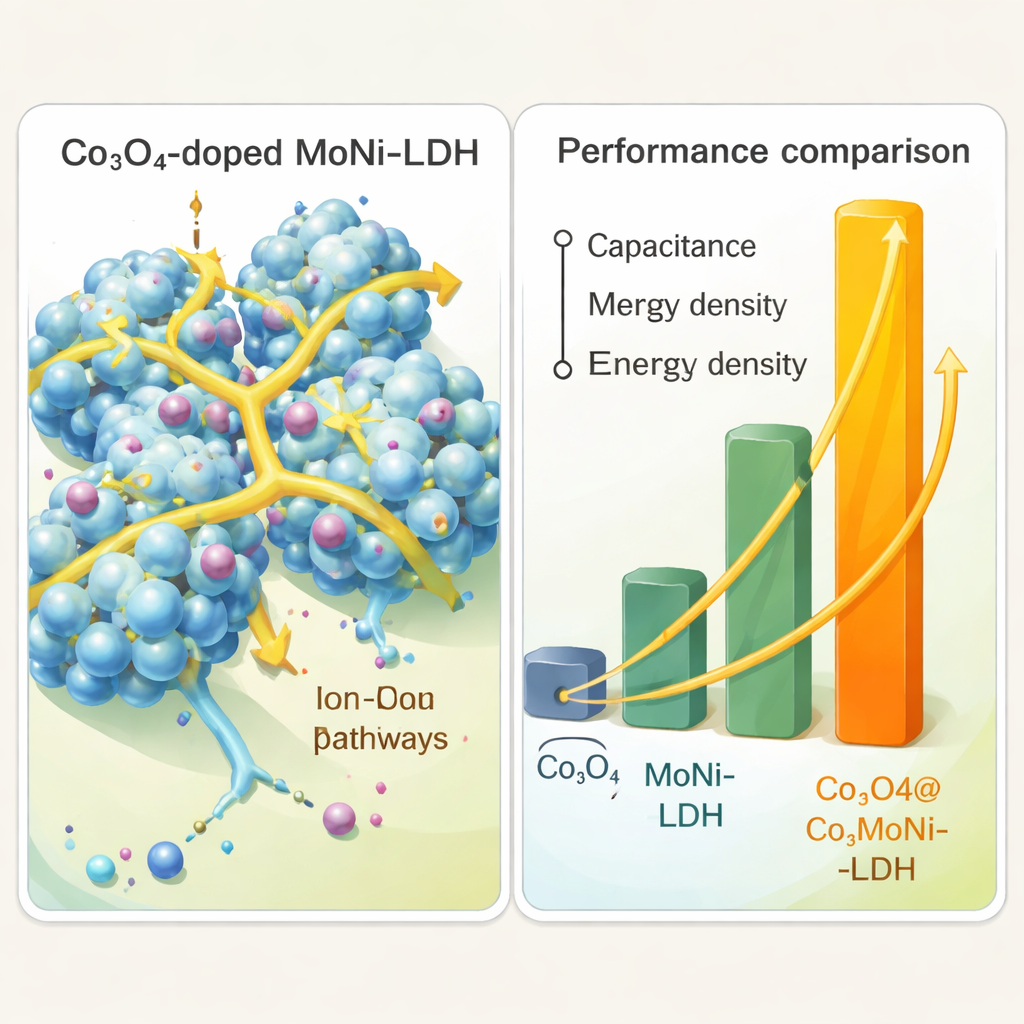
The team then builds simple two‑electrode test cells and measures how much charge the different materials can store and how fast they can deliver it. Compared with plain Co3O4 or plain MoNi‑LDH, the combined Co3O4@MoNi‑LDH electrode shows far larger electrical signals in cyclic tests, a sign of more active reactions. In constant‑current charge–discharge experiments, the composite reaches a specific capacitance of about 466 farads per gram at a moderate current—a figure roughly seven times higher than cobalt oxide alone and more than double that of the nickel‑molybdenum LDH. The energy density, a measure of how much usable energy can be drawn from a given mass, also jumps dramatically, surpassing 165 watt‑hours per kilogram under the test conditions. Even after 5000 rapid cycles, most of the original performance remains, showing that the material is durable.
Why the Mix Works So Well
Electrical measurements that probe internal resistance help explain the gains. The composite electrode offers lower resistance to both electrons and ions than the individual ingredients, meaning charges can move more freely through the material and its liquid electrolyte. The intertwined nanowires keep the LDH layers from clumping together, preserving open paths for ion flow. At the same time, cobalt, nickel, and molybdenum each contribute their own redox reactions, multiplying the number of sites where charge can be stored. This combination of a porous, well‑connected structure and multiple active metals is what gives the hybrid its edge.
What This Means for Everyday Technology
For non‑specialists, the main message is that carefully mixing and shaping familiar metals at the nanoscale can transform how well an energy storage device works. The Co3O4@MoNi‑LDH electrode introduced here stores far more energy than earlier versions while still charging quickly and standing up to repeated use. Although this is still laboratory‑scale work, the relatively simple, water‑based manufacturing method suggests that such materials could one day be produced in bulk. If that happens, we could see supercapacitors taking on a larger role alongside batteries in electric vehicles, portable electronics, and the smooth operation of solar and wind power grids.
Citation: Oroujzadeh, R., Rostami, S., Mirzaei-Saatlo, M. et al. Enhanced electrochemical behavior of Co3O4-modified MoNi-layered double hydroxide nanocomposites for pseudocapacitive applications. Sci Rep 16, 5517 (2026). https://doi.org/10.1038/s41598-026-35216-y
Keywords: supercapacitors, energy storage, nanocomposites, electrode materials, pseudocapacitance