Clear Sky Science · en
Genetic polymorphisms in DNA repair gene XRCC1 and the risk of diabetic polyneuropathy
Why tiny DNA changes matter for people with diabetes
For many people with type 2 diabetes, the first warning sign of trouble is not high blood sugar on a lab report, but tingling, burning, or numbness in their feet and hands. This condition, called diabetic polyneuropathy, can lead to pain, loss of balance, and even limb amputation. Yet not all people with diabetes develop it, even when their sugar levels look similar. This study asked a simple question with big consequences: could small inherited differences in a DNA repair gene help explain who is most at risk, and could modern data science help doctors spot those people earlier?
A closer look at nerve damage in diabetes
Diabetic polyneuropathy arises when long nerves, especially in the legs and feet, are slowly damaged over years of high blood sugar, poor blood flow, and chronic “rusting” of cells by reactive oxygen molecules. These molecules can chip away at key cell components, including DNA. Our cells are not helpless: they carry repair tools that constantly patch this damage. One of these tools is a protein made by a gene called XRCC1, which helps fix single-strand breaks in DNA. If this repair system works poorly, nerve cells may be less able to survive the constant stress of diabetes, making nerve damage more likely and more severe.
What the researchers tested in Egyptian patients
The team studied 732 adults in Egypt: 503 with type 2 diabetes and 229 healthy volunteers. Among those with diabetes, roughly half already had nerve damage and half did not. The scientists focused on two common “spelling differences” in the XRCC1 gene, known as Arg399Gln and Arg194Trp. These tiny changes, called single nucleotide polymorphisms, swap one building block of the XRCC1 protein for another and may alter how efficiently it repairs DNA. Using standard genetic tests on blood samples, the researchers determined which versions of these two sites each person carried, and then compared these patterns with the presence and severity of nerve problems.
Genetic patterns linked to higher nerve risk
The results showed a clear pattern. People with diabetes who carried the less common versions of XRCC1—called the A form at position 399 and the T form at position 194—were more likely to have nerve damage than those with the common forms. In particular, individuals with two A copies at position 399 had several-fold higher odds of neuropathy than those with two G copies, even after taking age, sex, and smoking into account. Similarly, having at least one T at position 194 raised the risk. When the two positions were considered together as haplotypes, one combination (A–T) stood out as especially risky for nerve disease, while other combinations appeared to protect against nerve complications despite including one risky letter, hinting at more complex gene–gene interactions.
How machine learning sharpened the picture
To move beyond simple comparisons, the investigators turned to machine learning, a branch of artificial intelligence that can sift through many variables at once. Using Random Forest and XGBoost algorithms, they trained models on 80% of the data and tested them on the remainder. These models highlighted not only the XRCC1 variants, but also traditional clinical factors—such as longer diabetes duration, higher fasting blood sugar and “bad” LDL cholesterol, lower “good” HDL cholesterol, older age, and higher body mass index—as key predictors of neuropathy. An explanation tool called SHAP helped visualize how each factor pushed an individual patient’s risk up or down. The team also compared two bedside scoring systems for nerve damage, finding that the Toronto Clinical Neuropathy Score better captured disease severity than an older disability score.
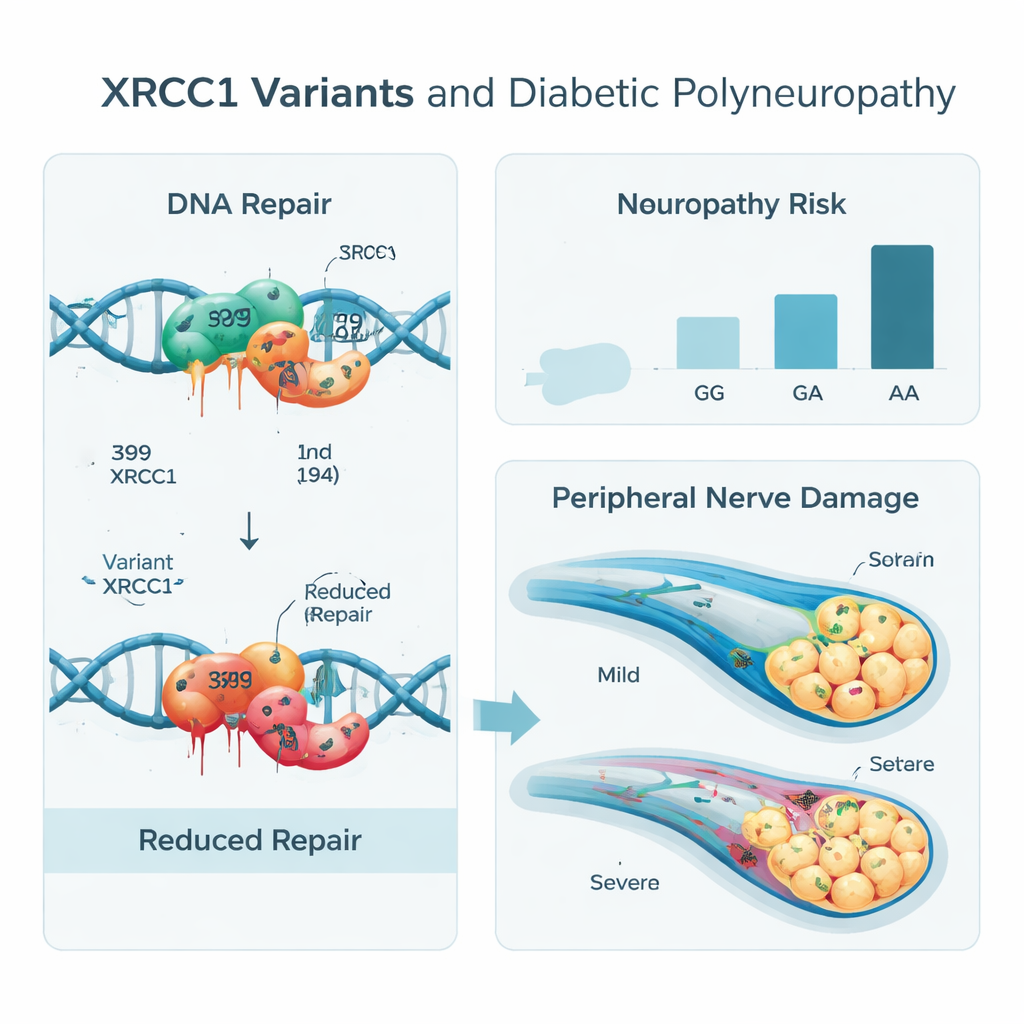
What this means for patients and prevention
In plain terms, the study suggests that some people with type 2 diabetes are born with a DNA repair toolkit that is less robust, making their nerves more vulnerable to the wear and tear of high blood sugar and abnormal blood fats. These inherited differences in the XRCC1 gene do not appear to cause diabetes itself, but they do seem to tilt the odds toward developing painful and disabling nerve problems once diabetes is present. By combining simple blood tests, clinical measurements, nerve scoring tools, and genetic information in smart computer models, doctors may one day be able to flag high-risk patients early and tailor follow-up and treatment—tight sugar and lipid control, lifestyle changes, and nerve-protective therapies—before serious nerve damage takes hold.
Citation: Hashim, N.A., El-Baz, H.A., Afya, Z.I.A. et al. Genetic polymorphisms in DNA repair gene XRCC1 and the risk of diabetic polyneuropathy. Sci Rep 16, 4815 (2026). https://doi.org/10.1038/s41598-026-35213-1
Keywords: diabetic neuropathy, DNA repair, XRCC1, genetic risk, machine learning