Clear Sky Science · en
REAC RGN-AR treatment modulates adipogenic differentiation in adipose tissue-derived stem cells
Why reshaping fat cells matters
Most of us think of body fat simply as extra weight, but fat tissue is a busy organ that talks to the rest of the body and can either protect or harm our health. In obesity and related diseases such as type 2 diabetes, fat cells often become enlarged, inflamed, and metabolically harmful. This study explores a new, noninvasive technology that gently uses radio waves to steer fat-derived stem cells away from forming unhealthy “storage” fat cells and toward a more active, calorie-burning type of cell. Understanding and harnessing this switch could open fresh paths for treating metabolic disorders and improving tissue repair.
A new way to talk to cells
Our bodies contain stem cells within fat tissue that can renew themselves and turn into several cell types, including fat, bone, and cartilage. These adipose tissue-derived stem cells (ADSCs) are easy to obtain from routine surgery and are already being explored for regenerative therapies. The challenge is that, especially in people with metabolic problems, these cells tend to become ordinary white fat cells that store large amounts of fat and can fuel low-grade inflammation. The researchers tested a technology called Radio Electric Asymmetric Conveyer (REAC), in a specific protocol they call TO RGN-AR, designed to subtly adjust the cells’ own electrical activity. The goal was to see whether this bioelectrical “tuning” could preserve the stem cells’ youthful flexibility while discouraging them from turning into unhealthy fat cells.
Keeping cells youthful and flexible
In laboratory experiments, ADSCs were isolated from small samples of human fat taken from adults without obesity or diabetes. The cells were then grown in dishes under conditions that normally push them to become fat cells. Some dishes received the REAC treatment for 72 hours via tiny probes placed in the culture medium, while others served as untreated controls. When the scientists later examined gene activity, they found that REAC exposure increased the levels of key “stemness” genes—Oct-4, Sox2, and Nanog—that help cells remain versatile and capable of self-renewal. Remarkably, this protective effect persisted even when the cells were exposed to a strong fat-inducing cocktail, suggesting that the radio electric treatment helped the cells resist a one-way commitment to becoming ordinary fat.
Steering away from storage fat
The team also measured genes that drive the formation of white fat cells, such as PPAR-γ, LPL, and ACOT2, which are involved in fat storage and lipid processing. In cells exposed to differentiation medium alone, these genes were strongly switched on, consistent with a shift toward large, triglyceride-packed fat cells. In contrast, REAC-treated cells showed a marked drop in these adipogenic markers without any loss of cell survival or growth. At the same time, a gene linked to heat-generating and metabolically active fat, UCP1, was increased. This pattern suggests that REAC does not simply block cell growth; instead, it redirects the developmental program away from building more storage fat and toward a more energy-burning identity.
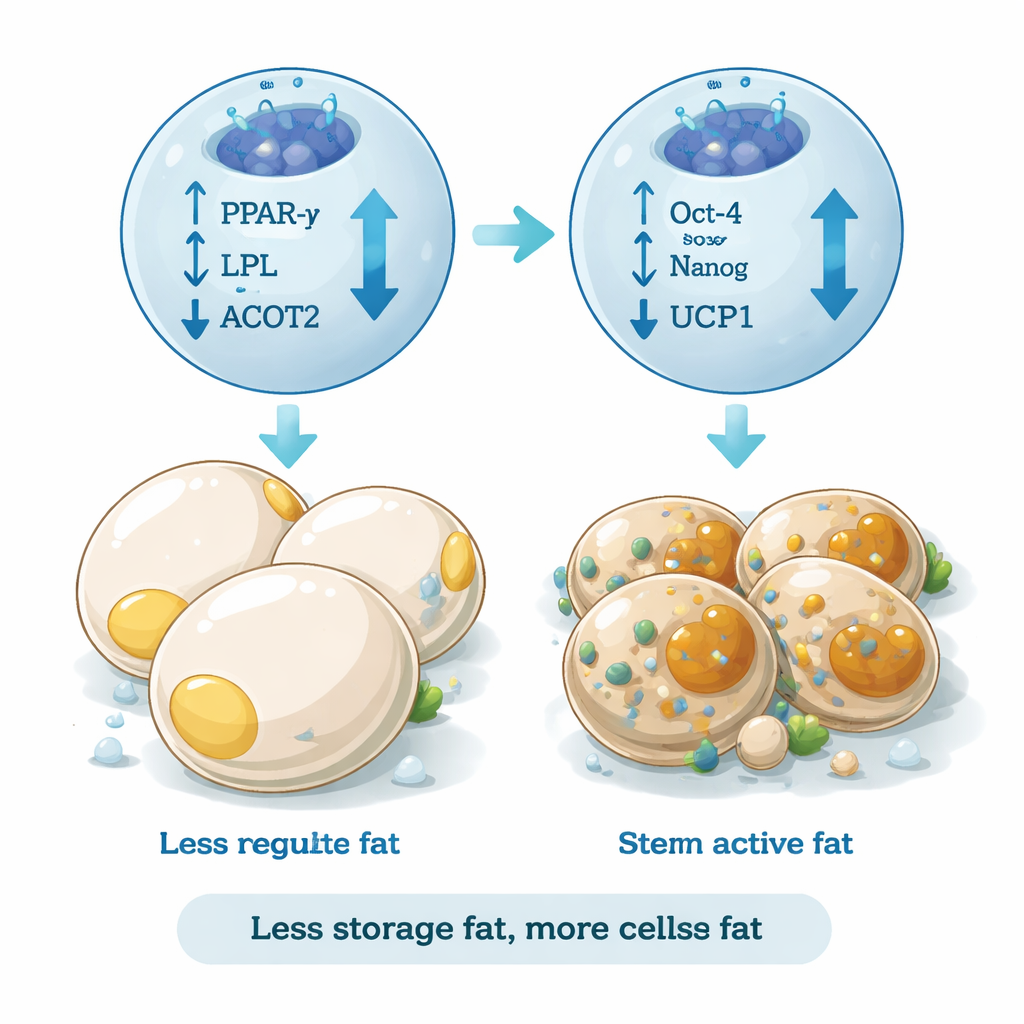
Encouraging “good” fat traits
Beyond gene activity, the researchers examined the actual appearance and surface markers of the cells. Using fluorescent labeling, they tracked proteins that distinguish different fat cell types. Cells left to differentiate on their own expressed high levels of ASC-1, a marker of classic white fat. With REAC treatment, ASC-1 levels dropped, while markers associated with beige and brown fat, including TMEM26 and PAT2, became more prominent. Beige fat cells are of particular interest because they can burn calories to produce heat and tend to release anti-inflammatory signals. Under the microscope, REAC-treated cultures also contained fewer mature, lipid-filled fat cells and more cells that retained a stem-like shape, further indicating a shift toward a healthier, more flexible fat profile.
What this could mean for future therapies
Altogether, the findings suggest that REAC TO RGN-AR can gently “coach” fat-derived stem cells to stay more youthful, avoid turning into harmful storage fat cells, and instead adopt features of beige, metabolically active fat. While these experiments were done in dishes and not in patients, they point to a promising strategy: using finely tuned electrical cues to restore balance in fat tissue, reduce chronic inflammation, and support better metabolic health. If future studies in animals and humans confirm these effects, such noninvasive treatments could complement diet, exercise, and medications in tackling obesity-related diseases and could also improve the performance of stem-cell-based therapies in regenerative medicine.
Citation: Cruciani, S., Rinaldi, S., Fontani, V. et al. REAC RGN-AR treatment modulates adipogenic differentiation in adipose tissue-derived stem cells. Sci Rep 16, 4860 (2026). https://doi.org/10.1038/s41598-026-35204-2
Keywords: adipose stem cells, bioelectrical modulation, beige fat, metabolic disorders, regenerative medicine