Clear Sky Science · en
Integrated proteomics and metabolomics reveal phytosesquiterpene lactones inhibit TNBC cell activity by depleting ATP synthesis and reprogramming primary metabolism
Plant Compounds That Starve Aggressive Breast Cancer
Triple‑negative breast cancer is one of the hardest forms of breast cancer to treat because it lacks the usual hormone “handles” that many drugs grab onto. This study explores two molecules extracted from a medicinal plant that appear to shut down the internal power stations of these cancer cells, cutting off their energy supply while sparing normal breast cells. Understanding how this works could open the door to gentler, more targeted treatments for patients who currently have limited options.
Why Targeting Cancer’s Power Source Matters
Every cell depends on tiny structures called mitochondria to make ATP, the cell’s basic fuel. Cancer cells, especially aggressive ones, are often unusually dependent on their mitochondria to support rapid growth, spread, and survival under stress. The researchers focused on triple‑negative breast cancer cells, which account for about 15–20% of breast cancers and tend to relapse and metastasize more often than other types. They studied a natural molecule, deoxyelephantopin (DET), and its refined cousin DETD‑35, both sesquiterpene lactones from the medicinal plant Elephantopus. Earlier work showed these compounds trigger oxidative stress and unusual forms of cell death in cancer cells, but how they disturb the cells’ energy factories was not well understood.
How the Plant Molecules Disrupt Cancer Cell Mitochondria
Using human triple‑negative breast cancer cells grown in the lab, the team found that DET and DETD‑35 rapidly increased the production of reactive oxygen species, a type of chemically reactive “exhaust” generated during energy production. In response, the cells boosted certain protective enzymes, but not enough to restore balance. The compounds also forced open a channel in the mitochondrial membrane known as the permeability transition pore, a change linked to swelling, loss of membrane charge, and the early steps of cell death. Within just a few hours, ATP levels inside the cancer cells dropped sharply. When the researchers added an antioxidant beforehand, these damaging effects were largely reversed, showing that oxidative stress was a key part of the process.
Mapping the Damage to Proteins and Metabolism
To understand the big picture, the scientists combined two powerful “omics” approaches. They cataloged thousands of mitochondrial proteins and measured many small metabolites in treated vs. untreated cells. This integrated view revealed that DET and DETD‑35 disturbed proteins involved in oxidative phosphorylation, the main pathway mitochondria use to make ATP, and activated death‑related signaling. At the same time, core metabolic routes that handle amino acids, fats, and building blocks for DNA were re‑wired. Certain lipids that help shape the inner mitochondrial membrane, as well as molecules tied to antioxidant defenses and sugar breakdown, shifted in ways consistent with stressed, energy‑starved cancer cells. Crucially, these widespread changes were not seen in normal mammary cells exposed to the same doses, suggesting a degree of selectivity for tumor cells.
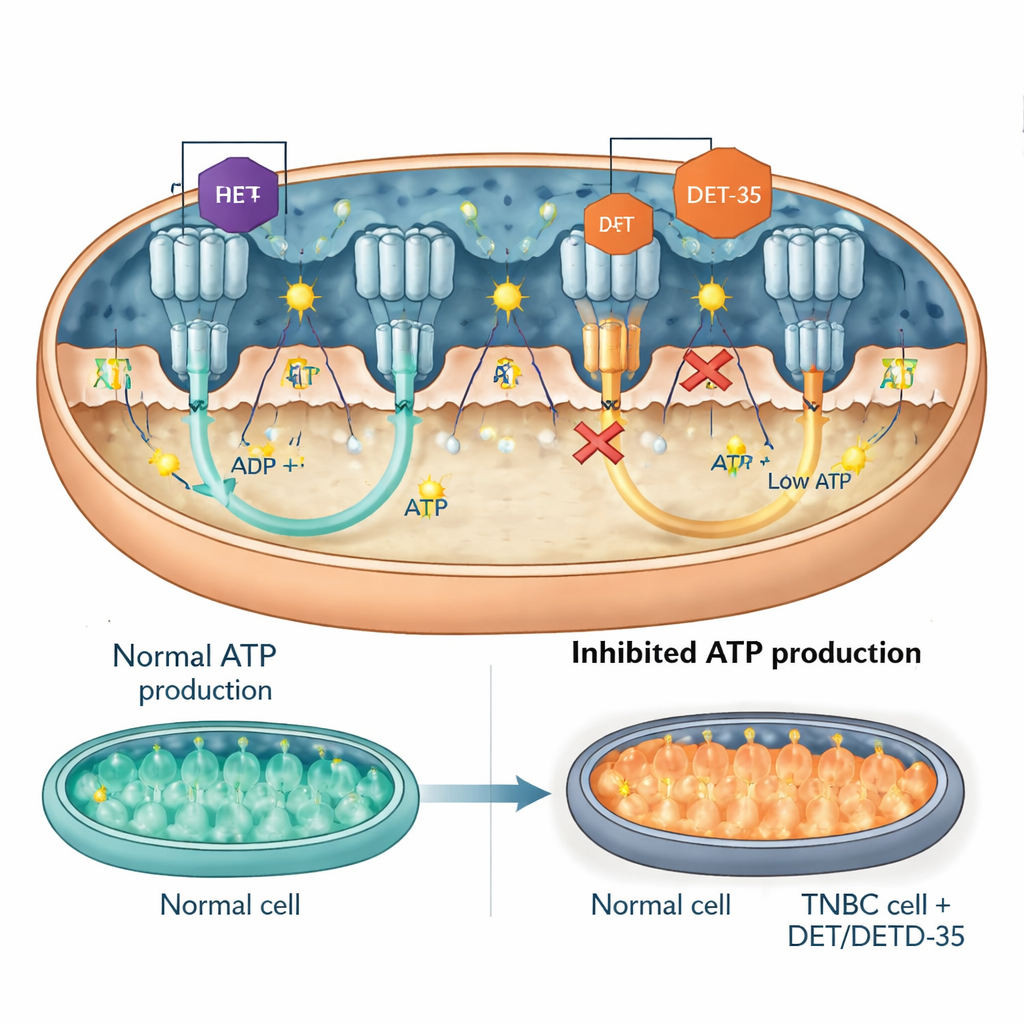
A Key Signaling Protein and the ATP Turbine Itself
The team then homed in on specific players within these disrupted networks. One was PRKCA, a signaling protein that can move into mitochondria and influence both energy output and stress responses. DET and DETD‑35 increased PRKCA levels in cancer cells. When researchers used genetic tools to dial PRKCA down, the drugs became less toxic: cancer cells were more likely to survive, produced more ATP, and showed fewer signs of mitochondrial malfunction and apoptosis. Another focus was ATP synthase, the molecular “turbine” that spins to create ATP. Measurements showed that DET and DETD‑35 directly reduced ATP synthase activity in cancer‑cell mitochondria. Computer docking models suggested that both compounds lodge at critical interfaces of the enzyme, the same general regions where known ATP synthase‑blocking drugs bind, physically hindering its motion and cutting ATP output. In mice bearing human triple‑negative breast tumors, treatment with either compound lowered the levels of ATP synthase components in the tumor tissue, supporting the cell‑culture findings.
What This Could Mean for Future Cancer Treatments
Overall, the study paints a coherent picture: these plant‑derived compounds push triple‑negative breast cancer cells into an energy crisis by overloading them with oxidative stress, prying open mitochondrial pores, and directly jamming the ATP‑making machinery. As ATP dwindles and metabolism is scrambled, the cancer cells lose their ability to grow and survive, while normal breast cells remain largely unharmed under the same conditions. To a lay reader, the takeaway is that scientists may be able to exploit cancer’s heavy dependence on its internal power stations, using carefully designed molecules—some inspired by plants—to drain the batteries of tumor cells without pulling the plug on healthy tissue. More work is needed before such agents could reach the clinic, but this integrated view of proteins, metabolites, and energy flow offers a promising roadmap.
Citation: Shiau, JY., Huang, HJ., Nakagawa-Goto, K. et al. Integrated proteomics and metabolomics reveal phytosesquiterpene lactones inhibit TNBC cell activity by depleting ATP synthesis and reprogramming primary metabolism. Sci Rep 16, 5264 (2026). https://doi.org/10.1038/s41598-026-35194-1
Keywords: triple-negative breast cancer, mitochondria, ATP synthase, natural products, cancer metabolism