Clear Sky Science · en
Extracellular vesicle-derived miR-760 as a novel promising candidate biomarker differentiating stable RRMS from SPMS
Why this research matters for people with MS
Multiple sclerosis (MS) can follow very different paths. Some people have clear attacks with periods of recovery (relapsing-remitting MS, or RRMS), while others gradually get worse over time (secondary progressive MS, or SPMS). Today, doctors mainly rely on symptoms and MRI scans to tell these forms apart, and there is no simple blood test that shows when a patient is moving from one stage to the other. This study explores whether tiny packets released by cells into the blood, carrying small genetic regulators called microRNAs, could offer a new, minimally invasive way to distinguish stable RRMS from SPMS and help guide treatment decisions.
Tiny messengers traveling in the blood
Our cells constantly shed microscopic, membrane-bound bubbles known as extracellular vesicles into the bloodstream. These vesicles carry proteins, fats, and genetic material from their cells of origin, acting as messengers that reflect what is happening inside the body. In this study, researchers collected blood from three groups: people with stable RRMS, people with SPMS, and healthy volunteers. They isolated extracellular vesicles from plasma and confirmed their size, shape, and typical surface markers using electron microscopy, light scattering, and flow cytometry. This ensured that what they were measuring really came from vesicles, not from stray cell fragments or free-floating molecules.
Searching for a molecular signature
The team focused on microRNAs, very short strands of RNA that fine-tune the activity of many genes at once. Because they are protected inside vesicles, these microRNAs are relatively stable in blood. Using RNA sequencing and follow-up PCR tests, the researchers measured levels of selected vesicle-associated microRNAs known or suspected to be involved in inflammation and nerve damage. They also measured a broad panel of immune-signaling proteins (cytokines), as well as markers of nerve and support-cell injury, in the same blood samples. Comparing the groups, they found that four vesicle-derived microRNAs—miR-760, miR-98-5p, miR-301a-3p, and miR-223-3p—showed clear differences between stable RRMS and SPMS, while many inflammatory molecules were higher overall in MS than in healthy people.
A standout candidate: miR-760
Among these tiny regulators, miR-760 emerged as especially informative. In patients with stable RRMS, its levels in extracellular vesicles were markedly lower than in healthy controls. In SPMS, however, miR-760 levels rose back toward normal. When the researchers built statistical models to see which combination of markers best separated RRMS from SPMS, miR-760 consistently carried the strongest weight. A model that combined miR-760 with another microRNA (miR-146a-5p) and a growth factor linked to nerve support (basic fibroblast growth factor, or FGF basic) correctly distinguished the two MS forms with high accuracy. The study’s analyses of gene networks suggest that miR-760 may be connected to pathways involved in immune signaling, myelin repair, and protection of nerve cells, hinting that it might be more than just a passive marker.
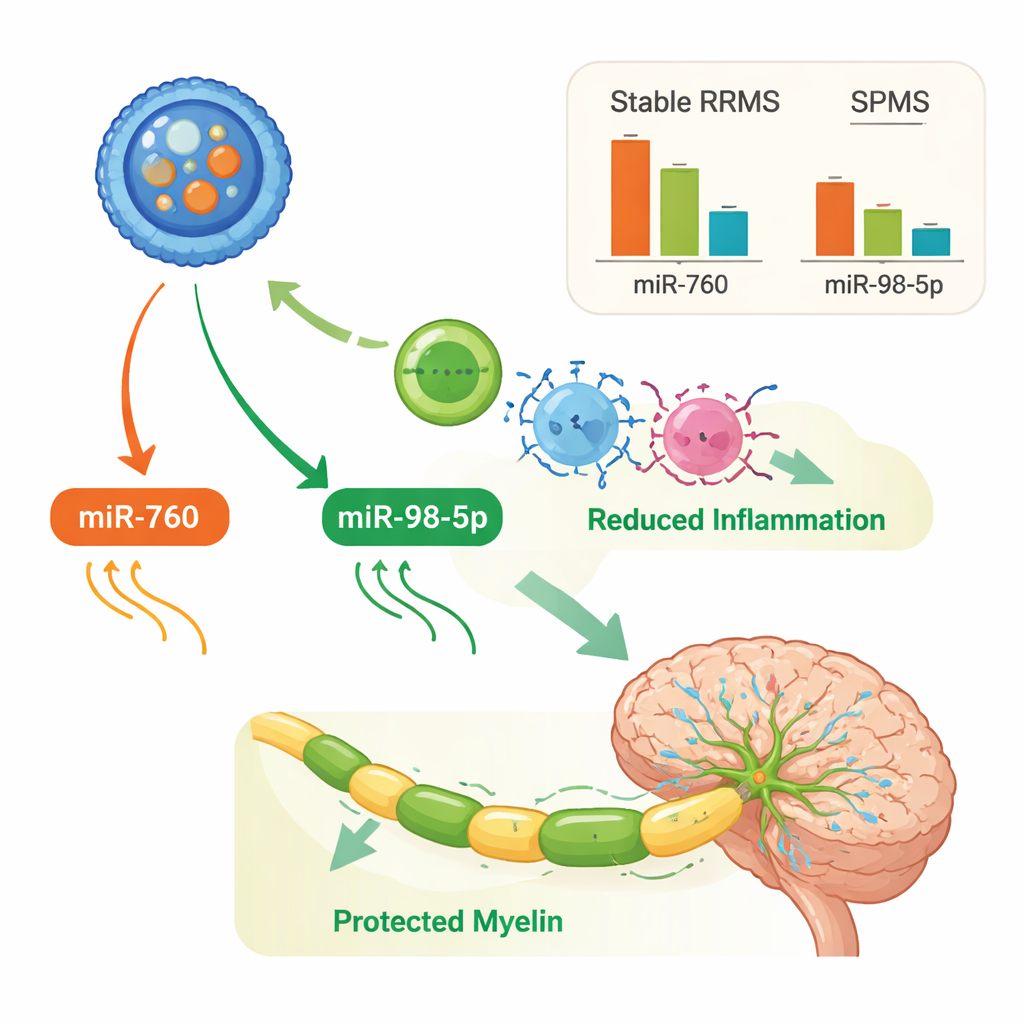
Different immune patterns in progressive disease
The researchers also investigated miR-98-5p, another microRNA that was boosted in both RRMS and SPMS compared with healthy people, and especially high in SPMS. Prior work links miR-98-5p to the protection of the blood–brain barrier and the calming of certain aggressive immune cells. In this study, network and pathway analyses suggested that the set of genes controlled by miR-98-5p and the other key microRNAs touches many processes relevant to MS, including inflammation, disposal of damaged proteins, and pathways shared with other brain diseases such as Alzheimer’s and ALS. Interestingly, some relationships between microRNAs and immune signals—such as inverse links between miR-760 or miR-98-5p and inflammatory cytokines IL-4 and IL-17—appeared only in SPMS, pointing to a shift in how immune regulation is wired as the disease becomes progressive.
What this could mean for future care
For people living with MS, the practical promise of this work is a future in which a simple blood draw might help show whether their disease is staying in a relapsing-remitting phase or drifting into a more steadily worsening course. The authors propose vesicle-derived microRNAs—especially miR-760 and miR-98-5p—as promising building blocks for such tests, particularly when combined with protein markers like FGF basic. At the same time, they stress that these findings are early: the study involved a modest number of patients from a single center, and the biological roles of these microRNAs in MS need to be confirmed experimentally. Larger, independent and long-term studies will be required before doctors can rely on these signals in everyday practice, but the results mark an important step toward more precise, blood-based tools for tracking the course of MS.
Citation: Wasilewska, K., Dziedzic, A., Anandan, S. et al. Extracellular vesicle-derived miR-760 as a novel promising candidate biomarker differentiating stable RRMS from SPMS. Sci Rep 16, 5208 (2026). https://doi.org/10.1038/s41598-026-35189-y
Keywords: multiple sclerosis, biomarkers, extracellular vesicles, microRNA, disease progression