Clear Sky Science · en
Rab43 mitigates the inflammatory response in acute lung injury via MyD88 ubiquitination
Why calming lung inflammation matters
When a severe infection or injury strikes the lungs, the body’s own immune response can become dangerously overactive. This runaway inflammation, known as acute lung injury and its most severe form acute respiratory distress syndrome, can leave patients in intensive care units struggling to breathe and carries a high risk of death. Today, doctors mainly provide supportive care such as oxygen or ventilators, because there are no precise treatments that dial down harmful inflammation without weakening essential defenses. This study uncovers a tiny cellular “traffic cop” protein called Rab43 that helps keep lung inflammation in check, pointing toward a new kind of targeted therapy.
A closer look at overworked lungs
In acute lung injury, infection or toxins damage the thin barrier between air sacs and blood vessels. Local immune cells, especially macrophages that patrol the air spaces, rush to fight off invaders by releasing chemical signals called cytokines. While these molecules help kill microbes, in excess they draw in swarms of white blood cells and leak-promoting fluids, filling the air sacs with fluid and making gas exchange difficult. Patients can develop low blood oxygen, widespread inflammation, and often need mechanical ventilation. Understanding how macrophages decide when to stop producing these inflammatory signals is therefore crucial for designing smarter treatments.
The tiny regulator called Rab43
Rab43 belongs to a large family of small switch-like proteins that control how cargos move inside cells. The researchers had previously found that Rab43 helps macrophages clear away dead cells in damaged lungs. In this new work, they focused on whether Rab43 also influences how strongly macrophages respond to bacterial components such as lipopolysaccharide, a molecule that powerfully triggers inflammation. In both mouse lung macrophages and lab-grown bone marrow–derived macrophages, exposure to lipopolysaccharide caused Rab43 levels to fall. This suggested that during acute lung injury, the natural brake provided by Rab43 might be lost just when it is needed most.
What happens when Rab43 is missing
To see Rab43’s impact in a living organism, the team engineered mice that lack Rab43 only in their myeloid cells, including macrophages. They then induced acute lung injury by delivering lipopolysaccharide directly into the airways. Compared with normal mice, Rab43-deficient animals developed more severe lung damage, with more fluid in the lungs, more infiltrating neutrophils, and higher levels of inflammatory cytokines such as TNF-α, IL‑6, and IL‑1β in the blood and lung fluid. These mice lost more weight, showed signs of persistent whole-body inflammation, and had a substantially higher death rate after a lethal lipopolysaccharide challenge. The findings indicate that Rab43 normally restrains macrophage-driven inflammation and limits collateral damage to lung tissue.
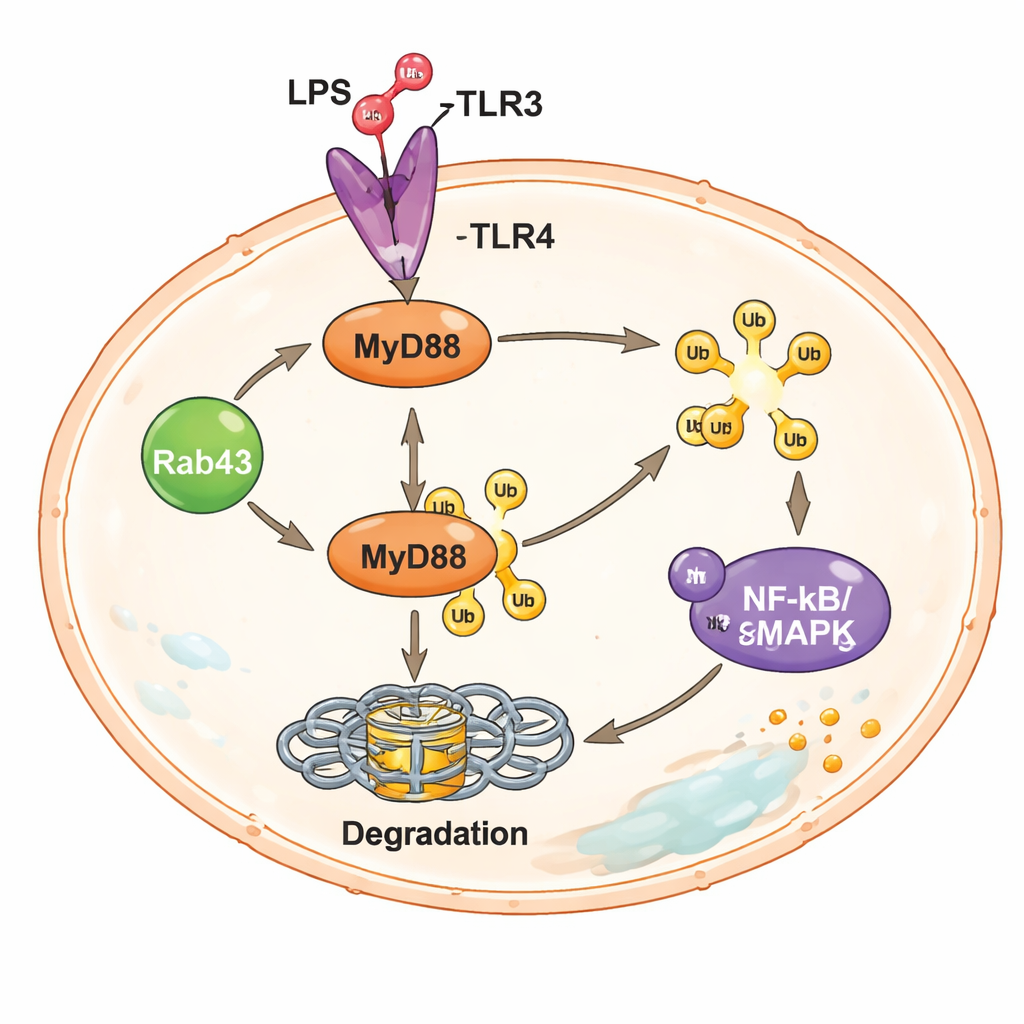
The signaling hub that Rab43 keeps in balance
Digging deeper, the scientists examined a key signaling adaptor inside macrophages called MyD88, which sits directly downstream of the lipopolysaccharide sensor on the cell surface. When activated, MyD88 launches powerful pathways, including NF‑κB and MAPK, that turn on genes for inflammatory cytokines. In macrophages lacking Rab43, MyD88 protein levels were markedly higher, and signals along these pathways were stronger, even though the amount of MyD88 genetic message (mRNA) did not change. This pointed to Rab43 influencing how quickly MyD88 protein is broken down rather than how much of it is produced. Indeed, the team showed that Rab43 promotes the attachment of tiny molecular “tags” called ubiquitin to MyD88, marking it for disposal by the cell’s protein recycling machinery. Without Rab43, MyD88 carried fewer of these tags, escaped timely degradation, and continued to drive inflammatory signaling.
How this discovery could guide future therapies
By linking Rab43 to the controlled removal of MyD88, the study reveals a built-in safety system that stops macrophage responses from overshooting after a threat is recognized. Rab43 boosts the expression of several enzymes that place or edit ubiquitin tags on MyD88, thereby ensuring that this powerful signaling hub does not stay switched on indefinitely. When Rab43 is missing or reduced, MyD88 lingers, inflammatory cascades run unchecked, and lung tissue suffers. Although treatments based on Rab43 are not yet available, these findings highlight Rab43 and the MyD88 degradation pathway as promising targets for future drugs designed to calm life-threatening lung inflammation while preserving the body’s ability to fight infection.
Citation: Wang, Y., Liu, X., Zhu, Z. et al. Rab43 mitigates the inflammatory response in acute lung injury via MyD88 ubiquitination. Sci Rep 16, 4843 (2026). https://doi.org/10.1038/s41598-026-35187-0
Keywords: acute lung injury, macrophages, inflammation, Rab43, MyD88