Clear Sky Science · en
Oleoylethanolamide supplementation improves mood and reduces fatigue in veterans with GWI in a 15-week randomized, double-blind, placebo-controlled exploratory clinical trial
Why this study matters to everyday readers
Many veterans from the 1990–1991 Gulf War still live with a puzzling condition called Gulf War Illness, marked by deep tiredness, low mood, pain, and thinking problems. There are no approved treatments. This study tested whether a naturally occurring fat-like molecule, oleoylethanolamide (OEA) — already sold as a dietary supplement — could safely help veterans feel less exhausted and improve their mood and day‑to‑day functioning.
A lingering illness after war service
Roughly one in three veterans from the first Gulf War is affected by Gulf War Illness, a chronic cluster of symptoms that include fatigue, mood disturbance, pain, and cognitive difficulties. These problems likely stem from a mix of disruptions in energy production, metabolism, and immune activity. Despite how common and disabling the condition is, there are still no medicines specifically approved to treat it, leaving many veterans without effective options and motivating researchers to explore safe, biology‑based supplements.
A gut‑brain messenger under the spotlight
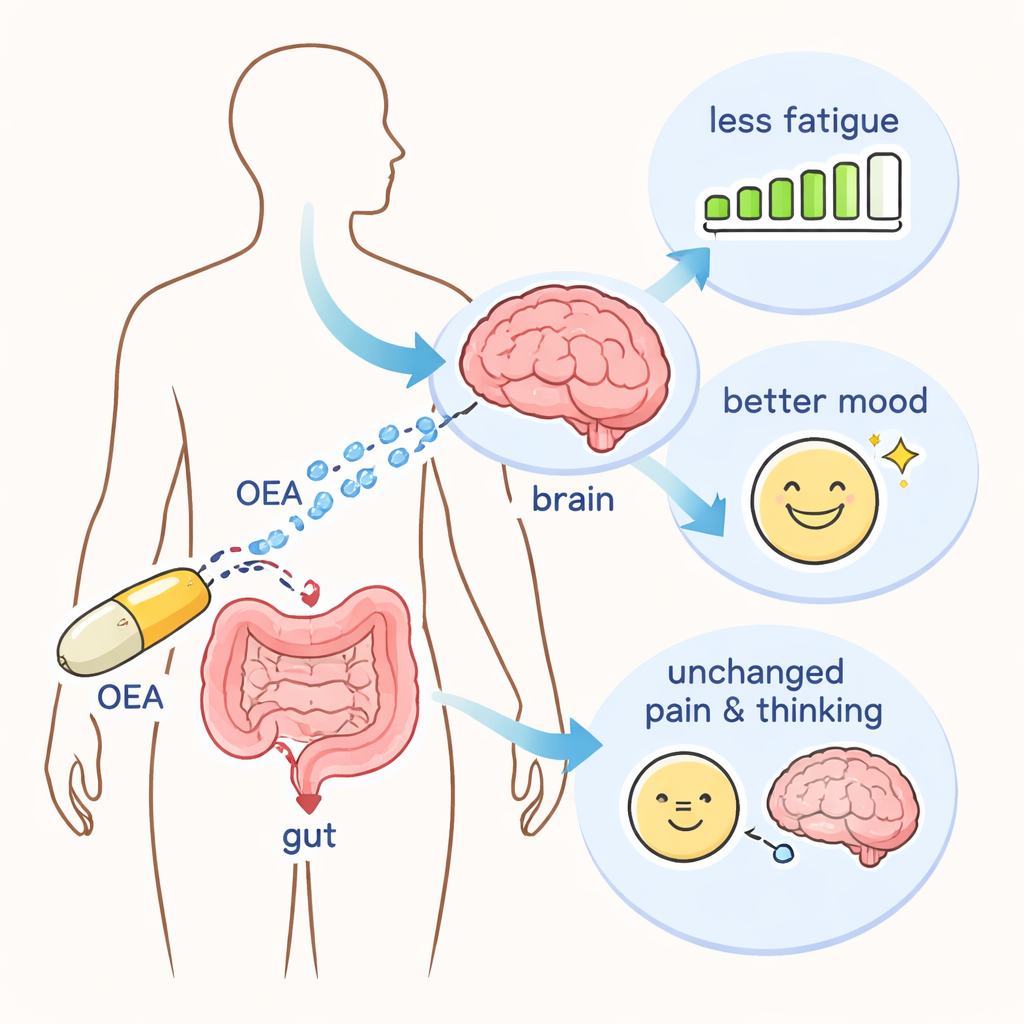
OEA is a small molecule made in the lining of the small intestine when we eat fat. It acts on a protein called PPAR‑alpha that helps regulate how the body uses energy and fats. Beyond appetite and weight control, OEA also talks to the “gut‑brain” nerve pathway, influencing brain circuits involved in energy, mood, and inflammation. Animal studies — including a mouse model exposed to chemicals linked to Gulf War Illness — suggested that OEA can reduce fatigue‑like behavior and improve mood and thinking, while calming brain inflammation. These hints led the researchers to ask whether OEA might bring similar benefits to veterans.
How the trial was run
The team conducted a 15‑week, randomized, double‑blind, placebo‑controlled clinical trial at a single site in Florida. Fifty‑two Gulf War veterans with a well‑defined chronic multisymptom illness were randomly assigned to receive either 200 milligrams of OEA twice a day or a matching placebo for 10 weeks. For the final 5 weeks, everyone received OEA in an open‑label extension. Throughout the study, participants completed standard questionnaires on fatigue, mood, pain, and quality of life, and took computerized and paper‑and‑pencil tests of memory and other thinking skills. Blood samples were used to measure OEA levels and several inflammatory and metabolic markers. Neither the veterans nor the study staff knew who was receiving OEA during the initial 10 weeks.
What changed — and what did not
Veterans taking OEA reported meaningful drops in overall fatigue compared with those on placebo, including improvements in general and mental fatigue and in feeling less limited and more motivated. On a widely used mood scale, OEA users had lower total mood disturbance, with particular reductions in tension, anger, confusion, and fatigue‑related mood complaints. On a veteran‑focused quality‑of‑life survey, those on OEA showed better energy, emotional well‑being, and social functioning over time. However, OEA did not improve performance on cognitive tests or reduce pain scores on a short pain questionnaire, suggesting that its main benefits in this trial were on energy and emotional health rather than on pain or thinking abilities.
Safety and what blood tests revealed
OEA was well tolerated. The number and types of side effects were similar in the OEA and placebo groups, and no serious safety problems emerged. Common complaints, such as mild digestive issues or muscle aches, occurred in both groups and were mostly rated as mild or possibly unrelated to the supplement. One participant in the OEA group and one in the placebo group reported a worsening of post‑traumatic stress symptoms; investigators judged these as unlikely to be caused by OEA. Blood measurements confirmed that OEA levels rose in those taking the supplement, but there were no clear changes in other related molecules, blood fats, or inflammatory markers during the short study window.
What this means for veterans and future research
For veterans living with long‑lasting fatigue and mood problems after Gulf War service, this exploratory trial offers cautious optimism. Over 15 weeks, OEA supplementation appeared safe and was linked to reduced fatigue and improved mood and daily functioning, even though it did not ease pain or sharpen thinking in this small study. Because the trial was modest in size and length, the authors stress that larger, longer studies are essential before OEA can be recommended as a proven treatment. Future research will need to pin down the best dose, clarify how OEA works in the body and brain, and identify which veterans are most likely to benefit.
Citation: Abdullah, L., Keegan, A.P., Hoffmann, M. et al. Oleoylethanolamide supplementation improves mood and reduces fatigue in veterans with GWI in a 15-week randomized, double-blind, placebo-controlled exploratory clinical trial. Sci Rep 16, 4933 (2026). https://doi.org/10.1038/s41598-026-35168-3
Keywords: Gulf War Illness, veterans, fatigue, mood, oleoylethanolamide