Clear Sky Science · en
Bacteria from foods and gut microbiota produce methylglyoxal and this metabolite leads to the formation of bioactive 1-acetyl-β-carboline alkaloids
Why your gut’s chemistry lab matters
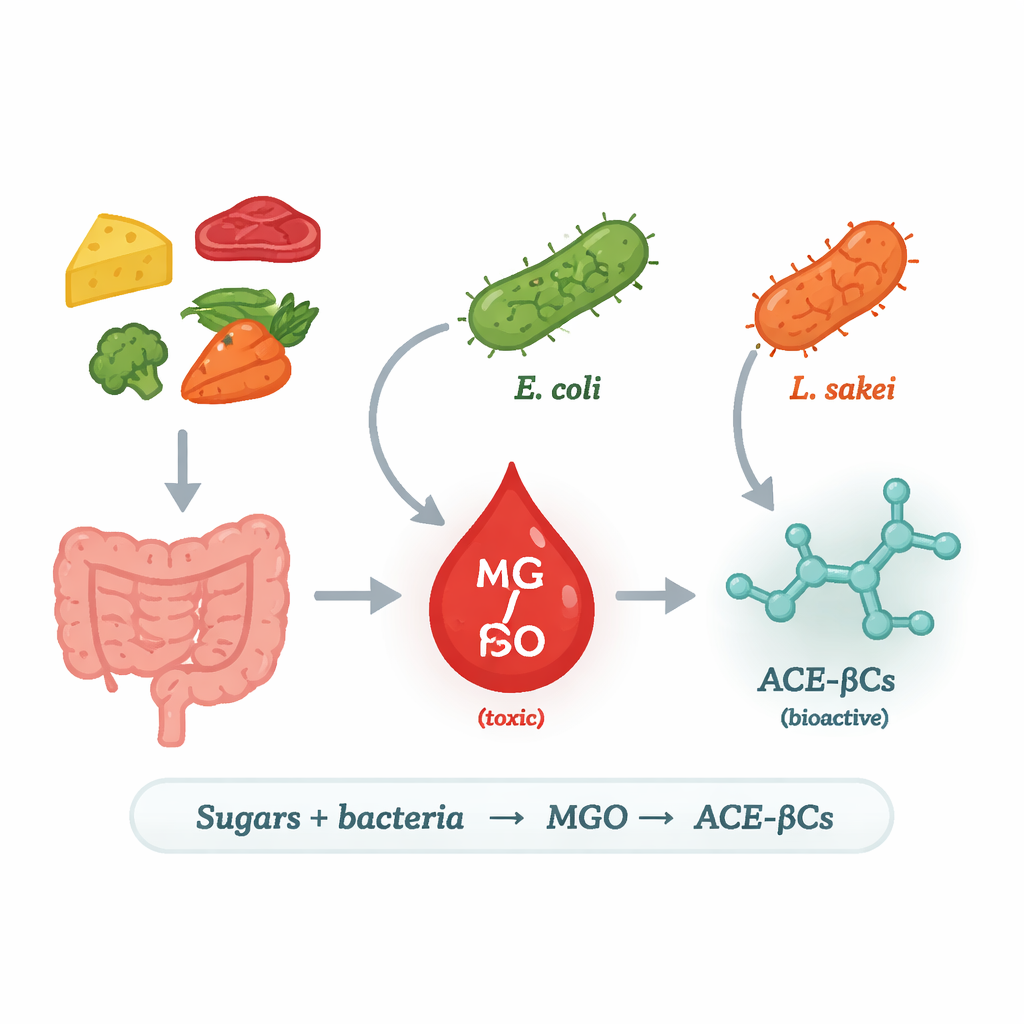
Inside our intestines and in many fermented foods, trillions of bacteria constantly transform the sugars we eat into a host of chemical by‑products. This study explores one such pathway in detail: how some common bacteria turn sugars into a highly reactive compound called methylglyoxal, and then into a family of bioactive molecules called 1‑acetyl‑β‑carbolines. Because these substances are linked both to disease processes and to potential new medicines, understanding when and how bacteria make them could reshape how we think about food, the microbiome, and health.
From everyday sugars to a risky molecule
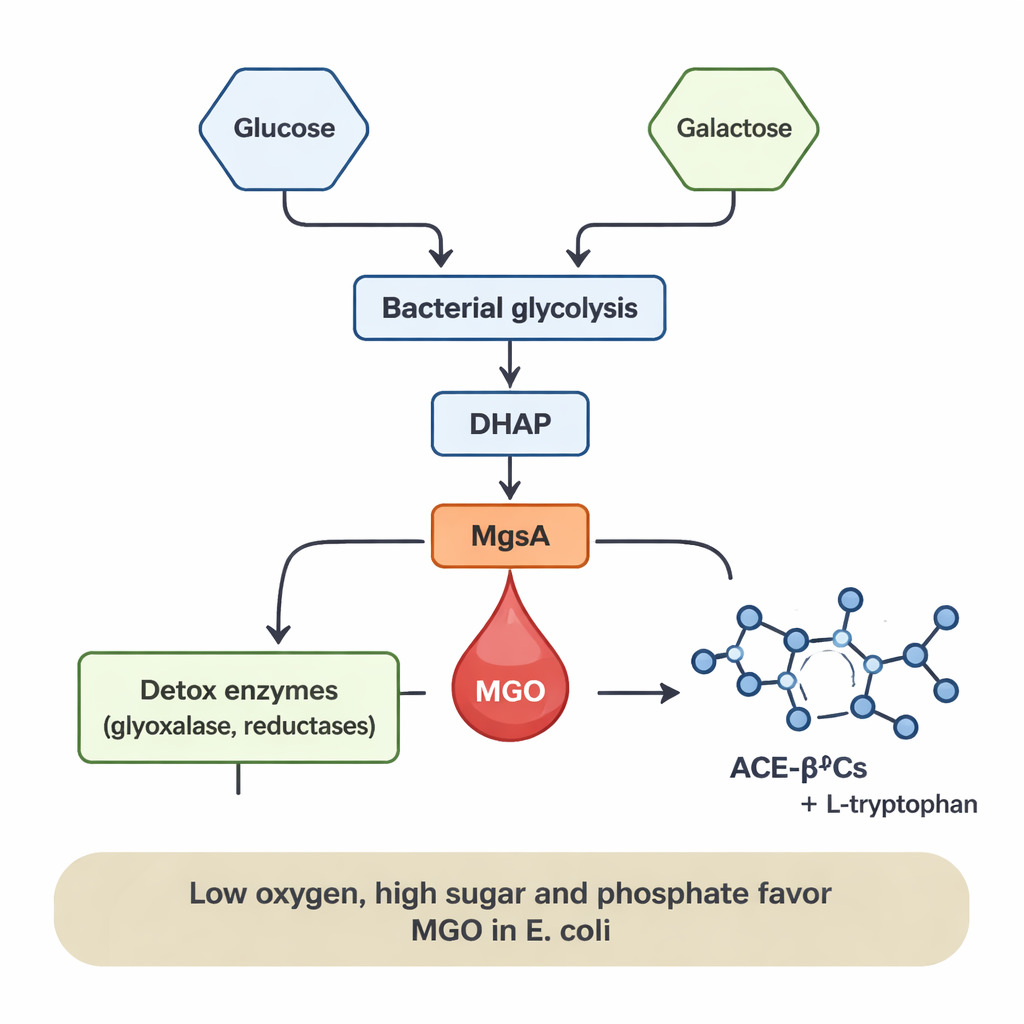
The researchers focused on methylglyoxal (MGO), a very reactive, toxic by‑product of sugar breakdown. In humans, excess MGO is associated with diabetes, heart disease, and neurodegeneration because it can damage proteins and DNA. Our cells normally keep MGO in check using detox systems, but they are not the only source: bacteria can also make MGO. In some microbes, an enzyme called methylglyoxal synthase (MgsA) converts a sugar‑processing intermediate directly into MGO, creating a “shortcut” in energy metabolism. The team asked which food and gut bacteria use this route, under what conditions, and what happens to the MGO they release.
Who are the bacterial producers?
To answer this, the scientists grew several strains of lactic acid bacteria often found in foods, along with a laboratory strain of Escherichia coli, in a controlled medium containing either glucose or galactose. They then tracked MGO and two related alkaloids in the culture liquid over time using high‑precision chromatography and mass spectrometry. Only E. coli and the meat‑associated species Lactilactobacillus sakei produced notable amounts of MGO and the alkaloids; other tested lactic acid bacteria did not. A genome search revealed the key difference: E. coli and L. sakei carry the gene for MgsA, whereas most of their lactic acid relatives do not. When the researchers inserted the mgsA gene from L. sakei into a non‑producing strain, that engineered bacterium began making both MGO and the alkaloids, firmly linking MgsA to this pathway.
How toxic MGO becomes bioactive alkaloids
The next question was what happens to the MGO once it is made. The team focused on 1‑acetyl‑β‑carboline alkaloids (ACE‑βCs), small molecules with reported antimicrobial, anti‑inflammatory and anticancer activities. They showed that when MGO‑producing bacteria were grown in the presence of the amino acid L‑tryptophan, levels of two ACE‑βCs—1‑acetyl‑β‑carboline and its 3‑carboxylic acid form—increased sharply. Adding extra MGO to the medium also boosted their formation. This indicates that MGO leaks out of the cells and reacts spontaneously with L‑tryptophan in the surrounding liquid, without any additional enzymes, to generate these bioactive compounds. In other words, the same chemistry that makes MGO dangerous also turns it into more complex molecules that may have useful effects.
What turns the dial up or down
MGO production was not fixed; it depended strongly on the bacteria’s diet and environment. In E. coli, MGO appeared only when glucose was present, and its levels climbed as glucose concentration rose. Adding phosphate, a common mineral nutrient, further amplified MGO formation by several‑fold, likely because it encourages build‑up of phosphorylated sugar intermediates that feed into the MgsA shortcut. Low‑oxygen (anaerobic) conditions—similar to those in the human gut—also favored MGO release, whereas high‑oxygen, well‑aerated conditions sharply reduced it even though the bacteria still grew well. In contrast, L. sakei mainly generated MGO when grown on galactose, and production increased with higher galactose levels. These patterns suggest that whenever sugar flow through bacterial metabolism is high and oxygen is limited, MGO can act as a “pressure valve,” relieving the strain of accumulating sugar intermediates.
What this could mean for food and health
The findings suggest that not all gut or food bacteria are equal in their ability to make MGO and ACE‑βCs. Species that carry the MgsA enzyme, such as certain L. sakei strains and E. coli, can be significant contributors, especially in sugar‑rich, low‑oxygen settings like the intestine or some fermentations. The MGO they excrete could add to the overall chemical burden on host tissues, but its conversion into ACE‑βC alkaloids may also partly “mop up” this reactive compound while generating molecules with their own biological effects. Although this study was done in laboratory cultures, it highlights a plausible route by which diet, microbiota composition, and bacterial metabolism together shape the balance between harmful and potentially beneficial chemicals in our bodies, and it points to ACE‑βCs as possible markers of bacterial MGO production in foods and the gut.
Citation: Herraiz, T., Sánchez-Arroyo, A., de las Rivas, B. et al. Bacteria from foods and gut microbiota produce methylglyoxal and this metabolite leads to the formation of bioactive 1-acetyl-β-carboline alkaloids. Sci Rep 16, 4905 (2026). https://doi.org/10.1038/s41598-026-35162-9
Keywords: gut microbiota, methylglyoxal, bacterial metabolism, food fermentation, beta-carboline alkaloids