Clear Sky Science · en
In situ high temperature X-ray diffraction and dilatometric analysis of CGO–Cu composites for solid oxide devices
Keeping High-Temperature Power Devices from Cracking
Solid oxide fuel and electrolysis cells can turn fuels, steam and even carbon dioxide into useful energy and chemicals with remarkable efficiency—but only if their ceramic and metal parts expand and contract together as they heat and cool. This paper explores a new way to measure and predict how a promising copper–ceria composite anode grows and shrinks at high temperatures, helping engineers design devices that last longer and fail less often.
Why Matching Expansion Matters
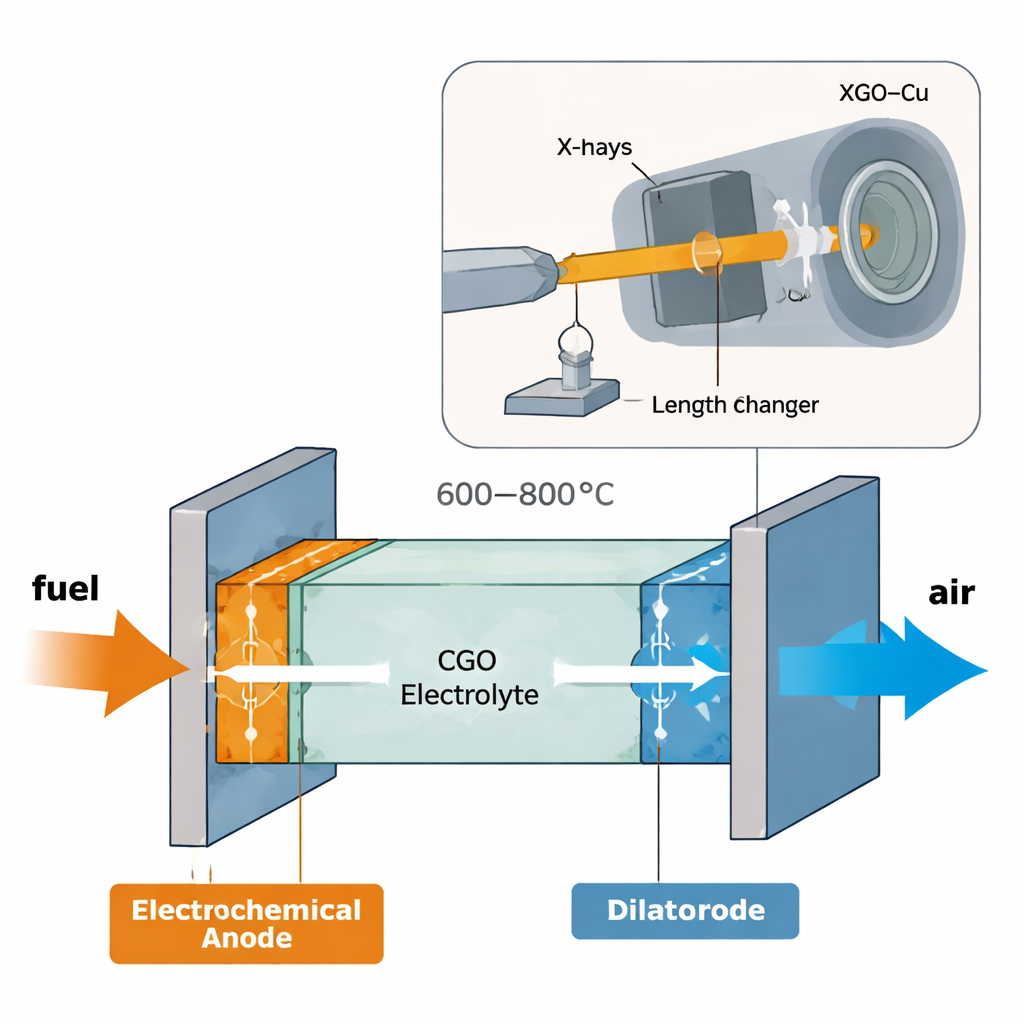
In solid oxide devices, electricity is generated or consumed in thin layers of ceramic and metal that must stay firmly bonded while operating at 600–800 °C. If one layer expands even slightly more than its neighbor, mechanical stress can build up, causing cracks or the electrode to peel away from the electrolyte. Traditional anodes based on nickel and zirconia are effective but vulnerable to carbon deposits and chemical damage when using real-world fuels. Copper–ceria composites offer a cleaner, cheaper alternative, but only if their thermal expansion closely matches that of ceria-based electrolytes. Understanding this match in realistic operating conditions is crucial for bringing more robust, lower-temperature solid oxide technologies to market.
A New Way to Watch Materials Breathe
The researchers focused on composites made from gadolinium-doped ceria (CGO), a fast oxygen-ion conductor, and copper, which provides electrical pathways. They prepared a series of CGO–Cu mixtures spanning roughly 40–70% ceria by volume, then shaped and treated them to form porous “cermet” bars similar to real anodes. Instead of testing thermal expansion and crystal structure separately, they combined two powerful methods in a single experiment: high‑energy synchrotron X‑ray diffraction to track atomic-scale lattice spacing in each phase, and dilatometry to measure the overall change in length of the whole bar during heating and cooling. This in situ setup let them watch both the microscopic and macroscopic “breathing” of the composite from room temperature up to 800 °C.
What Happens Inside the Composite
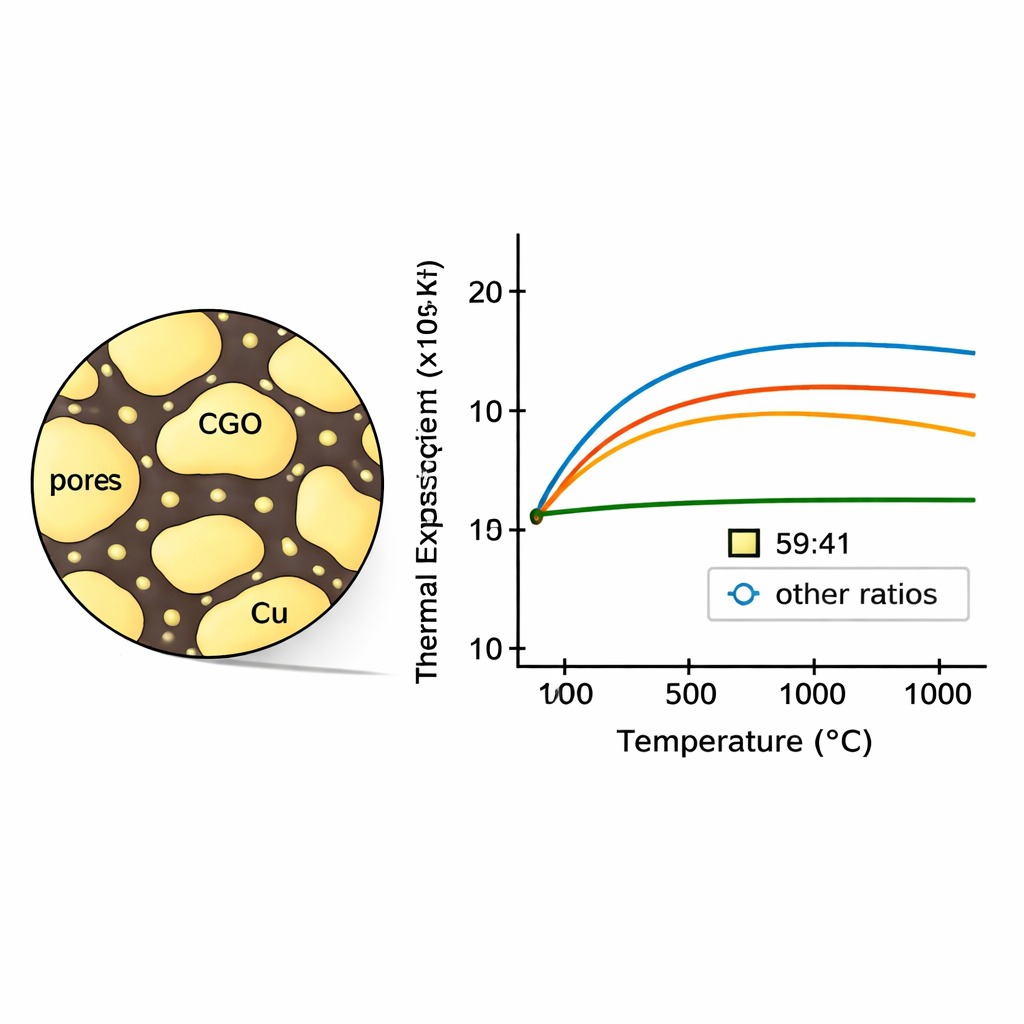
Imaging and composition analysis showed that copper does not remain as isolated particles. At high temperature and under reducing conditions, it becomes highly mobile, forming a continuous or semi‑continuous metallic network that wets the surfaces and grain boundaries of the CGO particles and fills pores. As copper content increased, overall porosity decreased and the material became denser, although the reduction of copper oxide to metal initially creates extra voids. X‑ray refinement revealed that both the CGO and Cu crystal lattices are slightly strained by their mutual constraints and that CGO grains become finer as the CGO fraction rises. These microstructural details—grain size, porosity and how the two phases interlock—strongly influence how the composite expands when heated.
Finding the Sweet Spot Composition
By extracting phase-specific expansion from the X-ray data and comparing it with the bulk expansion from dilatometry, the team showed that thermal expansion is not simply the average of the ceramic and metal values. At higher temperatures, additional sintering and pore closure, driven largely by mobile copper along grain boundaries, cause the composite to shrink slightly, bending the apparent expansion curves. Among all the tested mixtures, one stood out: the 59:41 CGO–Cu composite displayed an almost constant thermal expansion coefficient from room temperature to 800 °C, with minimal high-temperature shrinkage. Its overall expansion closely followed simple mixture rules, indicating that microstructural changes during heating were unusually small for this ratio.
What This Means for Future Energy Devices
For non-specialists, the key outcome is that the authors have identified both a promising material composition—CGO–Cu at 59:41 by volume—and a rapid, predictive measurement strategy. The combined X-ray and dilatometry approach reveals not only how much a composite expands, but also how its internal structure evolves while it does so. This makes it possible to engineer metal–ceramic electrodes that expand in lockstep with their electrolytes, reducing the risk of cracking and delamination. Such thermally stable, copper‑based cermets could help solid oxide fuel cells and electrolysis cells run reliably at lower temperatures, opening the door to more durable systems that convert fuels and greenhouse gases into electricity and valuable chemicals with less downtime and longer lifetimes.
Citation: Balaguer, M., Fabuel, M., Kriele, A. et al. In situ high temperature X-ray diffraction and dilatometric analysis of CGO–Cu composites for solid oxide devices. Sci Rep 16, 1315 (2026). https://doi.org/10.1038/s41598-026-35161-w
Keywords: solid oxide fuel cells, thermal expansion, cermet anodes, synchrotron X-ray diffraction, ceria copper composites