Clear Sky Science · en
Multistage treatment of industrial ethylene glycol (EG) effluent: integrating chemical extraction, coagulation/precipitation, and decolouration for enhanced wastewater remediation
Why Dirty Factory Water Matters to All of Us
Across many industries, a clear, sweet-tasting liquid called ethylene glycol keeps engines and machines from overheating or freezing. But when large amounts of this coolant end up in wastewater, it becomes a stubborn pollutant that is hard to remove and dangerous for rivers, lakes, and groundwater. This study explores a new, step-by-step way to clean ethylene-glycol–rich wastewater while also recovering some of the chemical for reuse, offering a practical path toward cleaner industry and safer water.
A Hidden Threat in Coolants and De‑Icers
Ethylene glycol is widely used in antifreeze, aircraft de‑icing fluids, and cooling systems in factories. It dissolves easily in water and carries a very high “chemical oxygen demand,” which means it can rob rivers and lakes of the oxygen fish and other life need to survive. Conventional treatment plants, especially those that rely mainly on microbes to break down pollution, often struggle with such strong and complex waste streams. The wastewater studied here came from a large desert industrial zone with many types of factories, producing a mix of ethylene glycol, oils, detergents, salts, and colored organic matter—far more complicated than the neat mixtures tested in many lab studies.
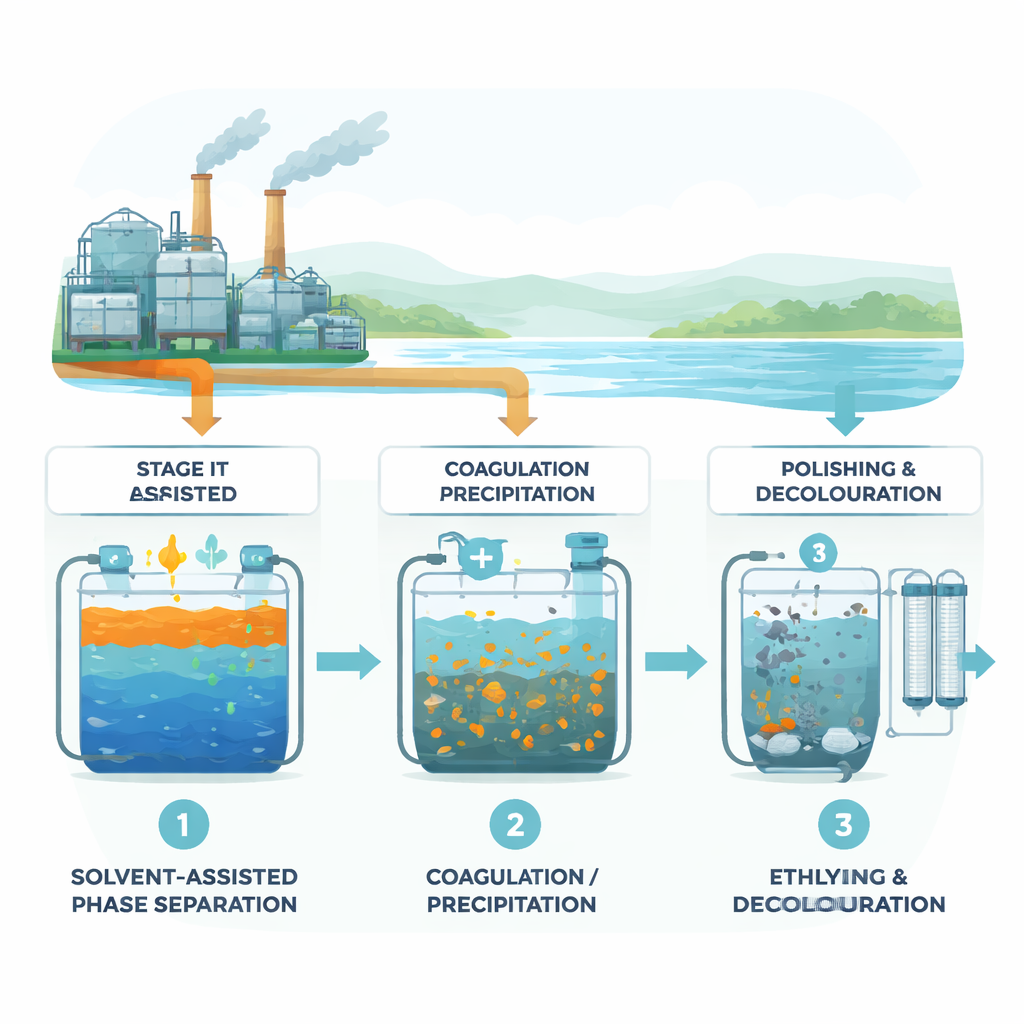
A Three‑Step Cleaning Line for Tough Wastewater
The researchers designed a multistage “treatment and recovery” line rather than a single magic step. First, they used a solvent called dichloromethane (DCM) in a phase‑separation step. Instead of plucking out pure ethylene glycol, DCM breaks up and pulls out clusters that contain glycol tangled with other organic materials and surfactants. This alone removed about three‑quarters of the overall organic load and captured an ethylene‑glycol–rich fraction that could potentially be refined and reused. Next, they added a coagulant—ferric chloride proved best—to make tiny suspended particles and cloudy material stick together and settle out. Finally, the partly cleaned water passed through a polishing stage built from nano‑sized aluminum particles and conventional filter media, which stripped out remaining color and much of the leftover dissolved pollution.
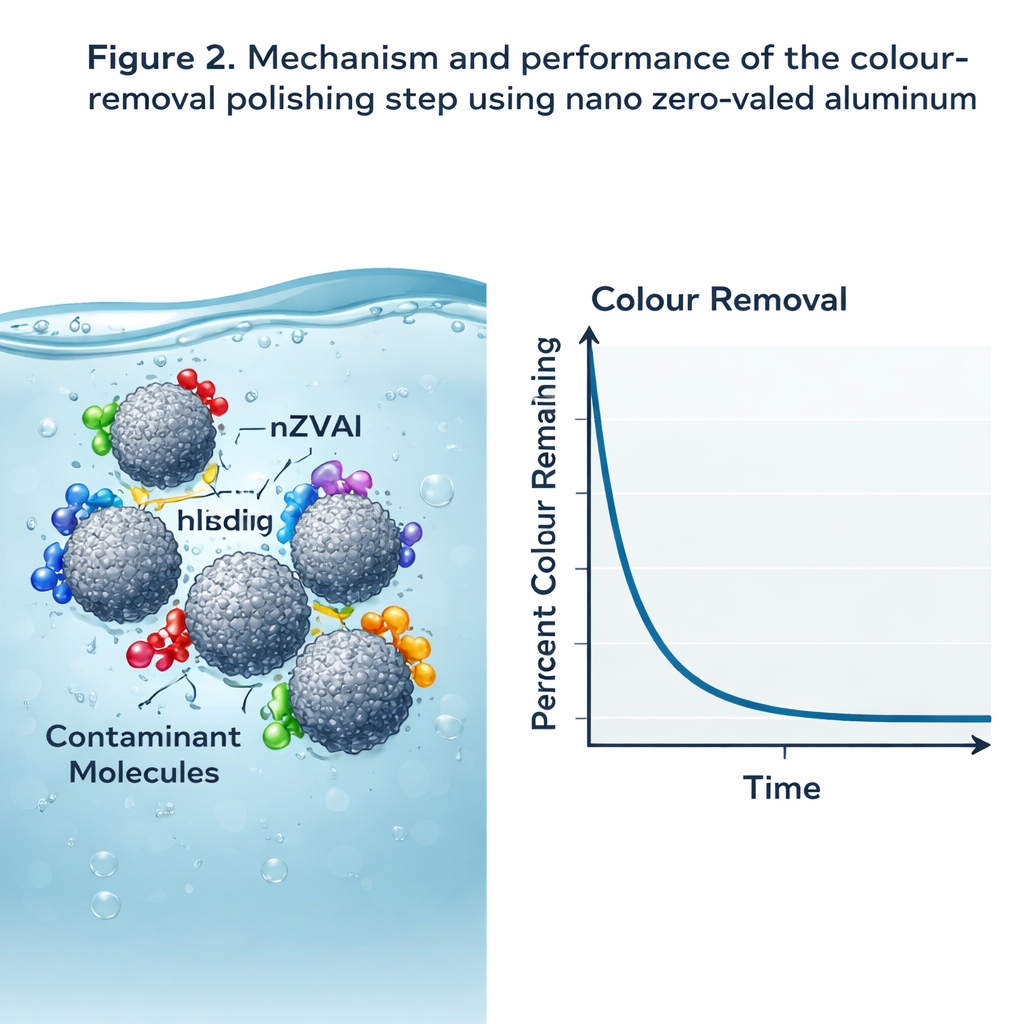
Nanoparticles as a Finishing Filter
At the heart of the polishing step are nano zero‑valent aluminum (nZVAl) particles, tiny grains of reactive metal with a huge surface area. These particles act like powerful microscopic sponges for colored and dissolved organic compounds. In carefully controlled tests, the team tuned the pH, dose, mixing speed, and contact time to get the best performance. They found that a modest amount of nZVAl, used at a pH close to where its surface is neutrally charged, removed more than 90% of the color in minutes, and a final filter bed containing nZVAl together with other media brought color removal to 100%. By tracking how quickly color disappeared from the water, the authors showed that the process followed a complex, multi‑step pattern rather than a simple one‑shot reaction, reflecting the varied surfaces and binding sites on the nanoparticles.
From Pilot Plant to Real‑World Impact
Importantly, the system was not just tested in flasks but in a pilot‑scale plant treating real industrial wastewater. By the end of the treatment train, levels of organic pollution, suspended solids, oil, and most metals fell below local discharge limits for sewers, and the once‑brown water became clear. A common advanced oxidation alternative, based on Fenton chemistry, did not perform as well on this complex mixture and created large amounts of iron‑rich sludge. The multistage system, in contrast, kept sludge volumes moderate and reduced chemical use by doing the heavy lifting in the first separation step.
Cleaner Water Without Breaking the Bank
To see whether such a system could be realistic for industry, the team estimated operating costs per cubic meter of treated water. When they accounted for solvent recycling and a modest credit for partially recovered ethylene glycol, the net treatment cost came out similar to existing methods for high‑strength industrial wastewater. In other words, factories could greatly improve their environmental performance—cutting color and organic pollution to low levels and conserving a valuable chemical—without facing crippling new expenses. For a layperson, the main takeaway is straightforward: by combining smart separation, traditional chemistry, and nanotechnology, it is possible to turn some of the dirtiest industrial water into a much safer stream, while nudging industry toward a more circular, less wasteful use of resources.
Citation: Mahmoud, A.S., Khamis, E., Mahmoud, M.S. et al. Multistage treatment of industrial ethylene glycol (EG) effluent: integrating chemical extraction, coagulation/precipitation, and decolouration for enhanced wastewater remediation. Sci Rep 16, 4088 (2026). https://doi.org/10.1038/s41598-026-35153-w
Keywords: ethylene glycol wastewater, industrial effluent treatment, nanoparticle polishing, solvent-based separation, circular economy