Clear Sky Science · en
The sequential use of IVF disposable devices doesn’t cause cumulative toxicity unless a defective device compromises the entire procedure
Why Everyday Lab Tools Matter for Making Babies
Most people think of in vitro fertilization (IVF) in terms of hormones, egg retrieval, and embryo selection. Few realize that success also depends on the dozens of plastic items—tubes, dishes, straws, and catheters—that eggs, sperm, and embryos touch along the way. This study asks a simple but important question: when all these disposable tools are used one after another in a normal IVF cycle, do their small chemical leaks add up to harm embryos, or is the danger mainly from rarely spotted defective devices?
The Hidden Chemistry Around IVF Embryos
Modern IVF aims to mimic the human body as closely as possible, controlling temperature, acidity, and oxygen levels so that embryos can grow safely. Yet every step relies on plastic consumables that can release tiny amounts of chemicals into the surrounding liquid. These substances may come from the plastic itself, from additives that improve flexibility or clarity, or from residues left by manufacturing and sterilization. Prior work showed that some combinations of tools used together can reduce sperm quality, raising concerns that embryos—arguably even more fragile—might also suffer from a build-up of contaminants over an entire IVF procedure.
Putting IVF Devices Through a Mouse Safety Test
To explore this, the researchers recreated ten typical IVF “paths,” from sperm collection and preparation through fertilization, embryo culture, freezing, thawing, and transfer. Each path used 7 to 25 different disposable items in the same order and for the same time and temperatures as in real clinics. Instead of exposing human embryos, they passed culture medium through the devices and then used that medium to grow mouse embryos in a standard safety test called the Mouse Embryo Assay. Fresh fertilized mouse eggs were followed for five days, checking whether they divided normally, reached the blastocyst stage (a hollow ball of cells ready to implant), and how many cells each blastocyst contained—a sensitive measure of embryo health.
When One Bad Device Spoils the Whole Chain
Most combinations of devices behaved well: embryos grown in media exposed to eight of the ten device paths developed just as well as in clean control media. However, two combinations clearly stood out. One included high-security sperm freezing straws, and the other focused on embryo transfer using a specific catheter model. In these two cases, fewer embryos reached the advanced blastocyst stage, and those that did had fewer cells, signaling stress or damage. To pinpoint the culprit, the team retested each component alone and in repeated batches. The same types of sperm straw and catheter consistently showed toxicity, especially when used under routine conditions such as wetting the cotton plug in the straw or pre-warming the catheter—steps that likely increase the release of volatile chemicals or hormone‑like compounds from the materials.
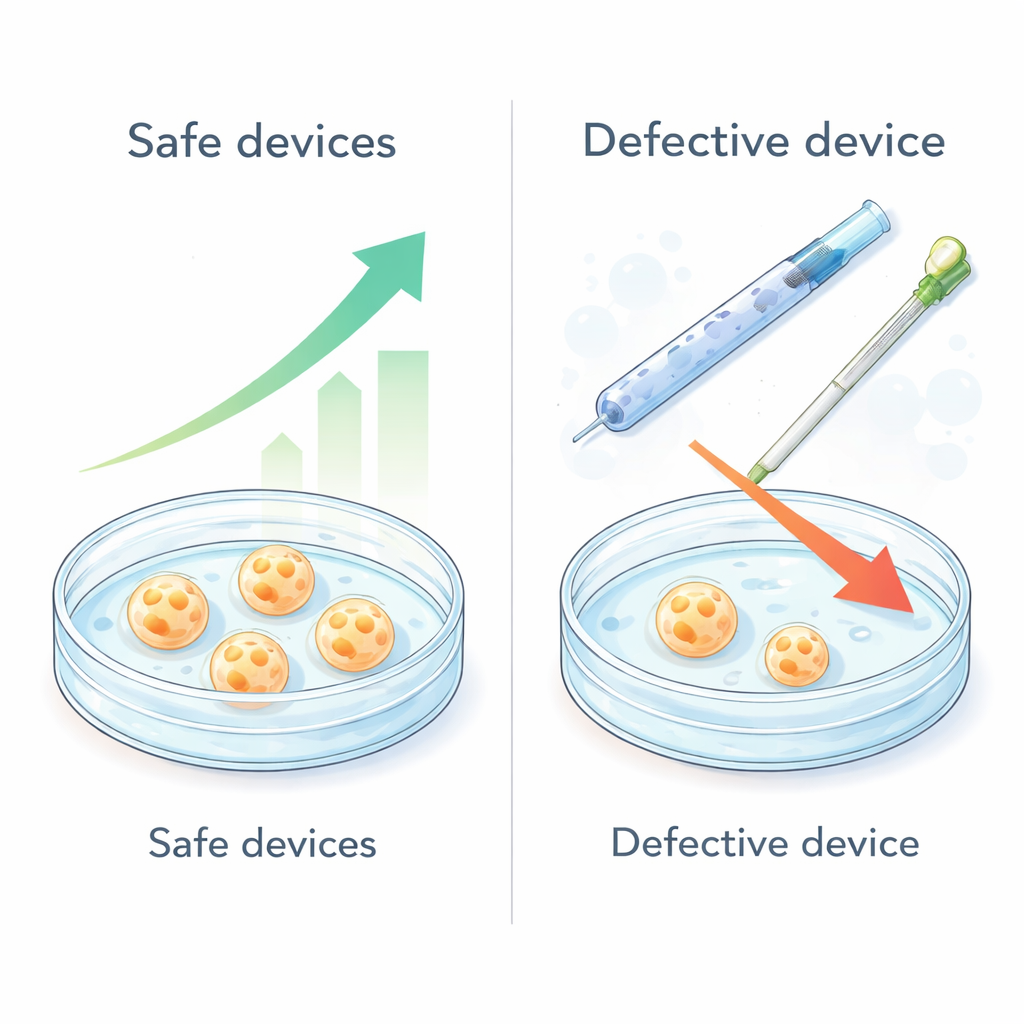
Why Safety Labels Are Not the Final Word
Strikingly, the problematic straws and catheters had already passed manufacturers’ internal safety checks and carried the required regulatory markings. The mismatch appears to stem from how the standard mouse assay is run. Different companies may use different mouse strains, culture conditions, or endpoints, some of which are less sensitive to subtle toxicity. The present study used a more demanding approach, including detailed blastocyst cell counts, and found “false negatives” that earlier tests missed. The work also reminds us that plastics can release complex mixtures—including volatile organic compounds and bisphenols—that may be harmful even at low levels, especially to early embryos whose development and long‑term health can be shaped by tiny epigenetic changes.
What This Means for Patients and Clinics
For people undergoing IVF, the reassuring news is that simply using many disposable devices in sequence does not automatically create a toxic environment. The danger arises when a single defective or poorly tested item is introduced, at which point it can compromise the entire procedure. For clinics and regulators, the message is more urgent: do not rely solely on manufacturers’ certificates. Each laboratory should verify new lots of critical devices with sensitive embryo‑based tests, and authorities should push for harmonized, more stringent testing protocols. By tightening quality control on the “invisible” tools of IVF, we can better protect embryos, reduce failed cycles linked to hidden toxicity, and give patients a safer, more reliable path to parenthood.
Citation: Delaroche, L., Besnard, L., Bazin, F. et al. The sequential use of IVF disposable devices doesn’t cause cumulative toxicity unless a defective device compromises the entire procedure. Sci Rep 16, 5491 (2026). https://doi.org/10.1038/s41598-026-35128-x
Keywords: IVF safety, embryotoxicity, laboratory plastics, mouse embryo assay, disposable medical devices