Clear Sky Science · en
Dual-specificity phosphatase 6 interferes with the repressive activity of forkhead box O1 towards CYP4A11 that mediates lipid accumulation in the liver
Why fat in the liver matters
Too much fat in the liver, often called fatty liver disease, is becoming increasingly common with modern diets rich in sugar and fat. While many people have heard of cholesterol or blood sugar, fewer know that tiny molecular switches inside liver cells help decide whether fat is burned or stored. This study looks at three such switches—proteins called DUSP6, FOXO1, and CYP4A11—and uncovers how their tug‑of‑war can push liver cells toward storing excess fat. Understanding this hidden control system could point the way to new treatments for fatty liver disease and related metabolic problems.
A hidden traffic controller inside liver cells
The researchers focused on a protein called DUSP6, which is known mainly for turning off a cell‑signaling route called the ERK pathway. Past work showed that mice lacking DUSP6 were surprisingly resistant to diet‑induced fatty liver, and their livers had lower levels of CYP4A enzymes, which are involved in fat processing. Here, the team asked what DUSP6 does in human liver‑derived cells and how it might affect a human version of these enzymes called CYP4A11. They used two standard liver cancer cell lines, HepG2 and HuH‑7, as stand‑ins for human liver cells and loaded them with a mixture of fatty acids (palmitic and oleic acid) to mimic an unhealthy, high‑fat environment. 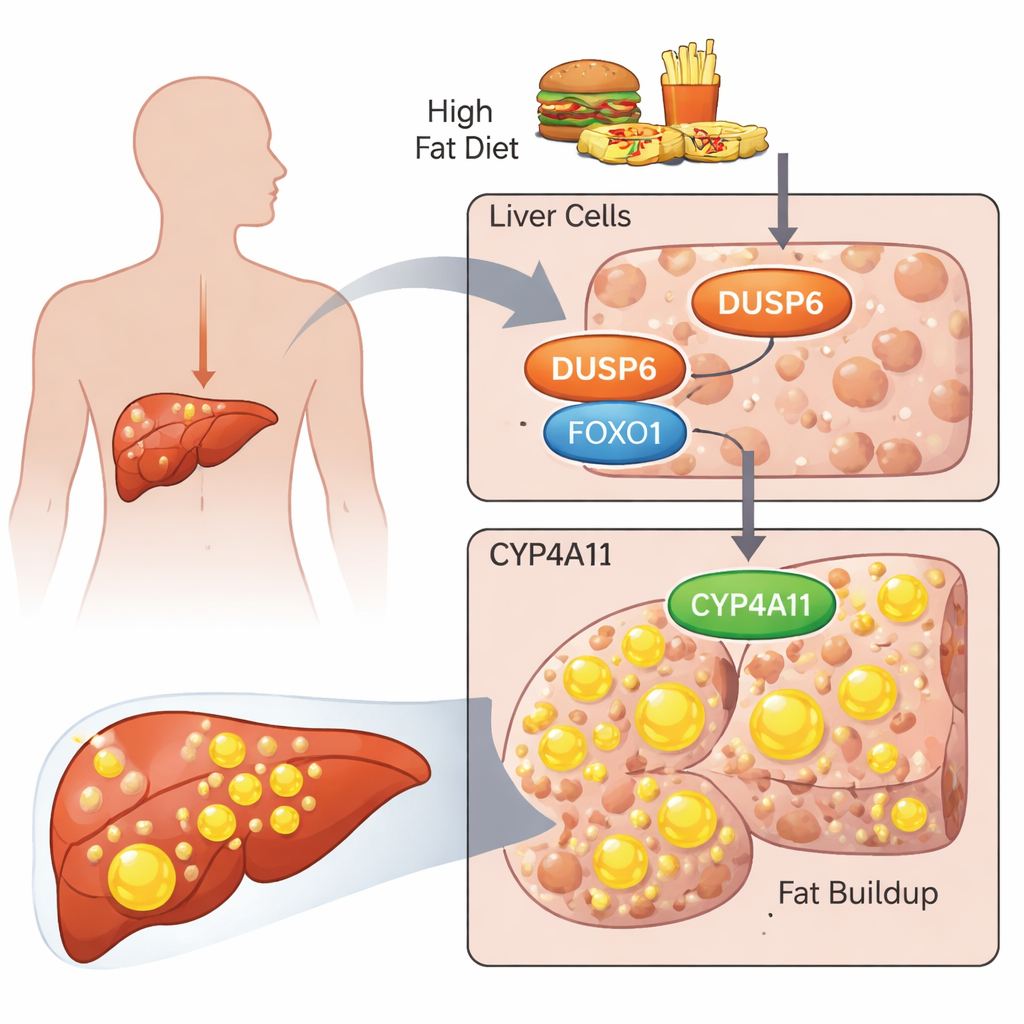
From fatty acids to a fat‑stuffed cell
When the cells were challenged with these fatty acids, they visibly accumulated fat droplets, which the scientists stained and measured. At the same time, levels of DUSP6 and CYP4A11 rose, along with activated forms of two major signaling proteins, AKT and FOXO1. Knocking down DUSP6 with targeted RNA molecules reversed this pattern: the cells stored less fat, CYP4A11 levels fell, but activated ERK, AKT, and FOXO1 all increased. Blocking the ERK pathway alone actually made fat buildup worse, whereas blocking AKT alone reduced it. When both ERK and AKT were inhibited together, fat accumulation dropped, and CYP4A11 levels declined. These experiments linked CYP4A11 tightly to how much fat builds up and suggested that AKT activity is especially important for driving fat storage.
A brake on fat‑promoting genes
Another key player, FOXO1, is a transcription factor—a protein that can enter the cell nucleus and turn genes on or off. FOXO1 is known to act as a brake on several members of the same enzyme family as CYP4A11. The team found that reducing FOXO1 levels caused the cells to hoard more fat and increased CYP4A11, while directly boosting CYP4A11 made the cells fattier still. In contrast, forcing cells to produce more FOXO1 reduced both CYP4A11 and fat accumulation. A drug that blocks CYP4A11 activity (HET0016) cut down the extra fat caused by low FOXO1, supporting the idea that CYP4A11 is not just a bystander but a driver of lipid buildup. Chemical tests showed that HET0016 lowered 20‑HETE, a product made by CYP4A11 that is linked to oxidative stress and inflammation, further tying this pathway to liver damage.
How DUSP6 disarms the brake
To understand how FOXO1 controls CYP4A11, the scientists examined the DNA region in front of the CYP4A11 gene and found sequences where FOXO1 can bind. Using a chromatin immunoprecipitation assay, they showed that FOXO1—specifically its non‑phosphorylated form—binds directly to the CYP4A11 promoter, consistent with a repressive, braking role on this gene. Co‑immunoprecipitation experiments revealed that DUSP6 physically interacts with FOXO1 but not with its phosphorylated version. Fractionation and microscopy studies showed that DUSP6 sits in the cell’s cytoplasm and, when present, keeps more FOXO1 out of the nucleus. When DUSP6 was knocked down, more FOXO1 moved into the nucleus, where it could bind DNA and suppress CYP4A11. In effect, DUSP6 acts like an anchor, sequestering FOXO1 in the cytoplasm and preventing it from doing its job as a brake on CYP4A11. 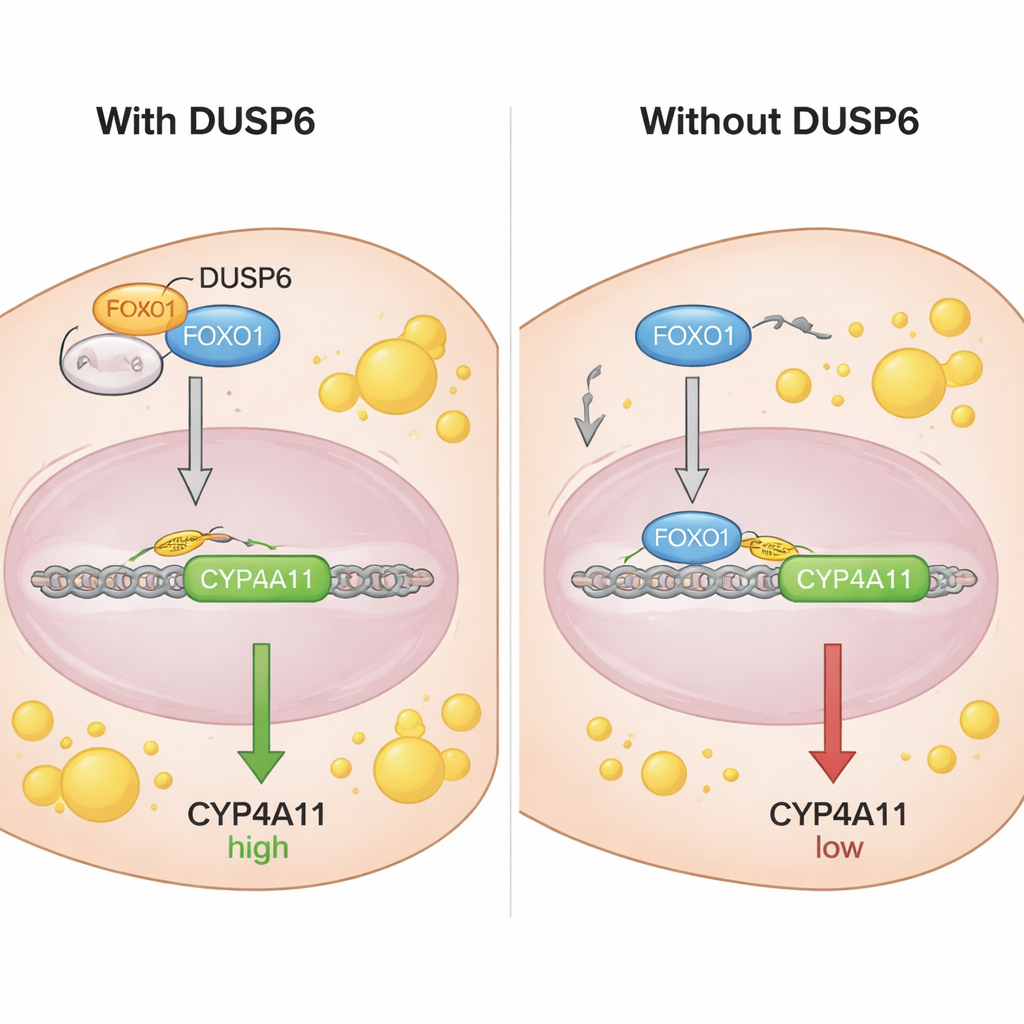
What this means for fatty liver and beyond
Put simply, this work maps out a chain of events inside liver cells: DUSP6 holds FOXO1 in the cytoplasm, which weakens FOXO1’s ability to shut down the CYP4A11 gene. With FOXO1 sidelined, CYP4A11 becomes more active, helping push the cell toward storing more fat and producing molecules like 20‑HETE that may promote oxidative stress and inflammation. While these findings come from cultured liver‑derived cells, not healthy human liver tissue, they highlight DUSP6 and CYP4A11 as potential new targets for treating metabolic dysfunction‑associated steatotic liver disease. Therapies that reduce DUSP6 activity or dampen CYP4A11’s effects could, in theory, restore FOXO1’s braking function and help keep liver fat in check.
Citation: Kimura, M., Saiki, Y., Iwata, K. et al. Dual-specificity phosphatase 6 interferes with the repressive activity of forkhead box O1 towards CYP4A11 that mediates lipid accumulation in the liver. Sci Rep 16, 4795 (2026). https://doi.org/10.1038/s41598-026-35118-z
Keywords: fatty liver disease, DUSP6, FOXO1, CYP4A11, liver metabolism