Clear Sky Science · en
Genetic diversity of Pseudomonas aeruginosa isolated from clinical samples with ISSR molecular marker in a tertiary care teaching hospital
Why hospital germs matter to all of us
Anyone who has spent time in a hospital—whether as a patient or visiting a loved one—depends on antibiotics to work when they are needed most. But some germs are becoming so tough that even our strongest drugs struggle to stop them. This article explores one such troublemaker, a bacterium called Pseudomonas aeruginosa, and shows how scientists in an Indian hospital mapped its hidden genetic variety to better understand why it is so hard to control.
A stubborn germ in modern hospitals
Pseudomonas aeruginosa is a slippery foe. It thrives in moist environments, from ventilator tubes to wound dressings, and preys especially on people whose defenses are already weakened by illness, burns or long hospital stays. It can cause serious lung, blood, urinary and wound infections. What makes it especially dangerous is its knack for resisting many antibiotics at once, turning routine infections into life‑threatening crises and driving up treatment costs and hospital stays worldwide.
Looking beneath the surface of infection
To see how diverse this germ really is inside one hospital, the researchers collected 100 bacterial samples from blood, urine, sputum and wound swabs taken during routine care at a large teaching hospital in eastern India. They focused on 18 strains that were resistant to multiple drugs and tested how each reacted to a wide panel of antibiotics. Worryingly, more than four out of five of the samples were resistant to key medicines such as cefoperazone, meropenem and imipenem—drugs often reserved as last‑line options when others fail. A few antibiotics, including some less commonly used ones, still worked better, hinting at remaining but narrowing treatment choices. 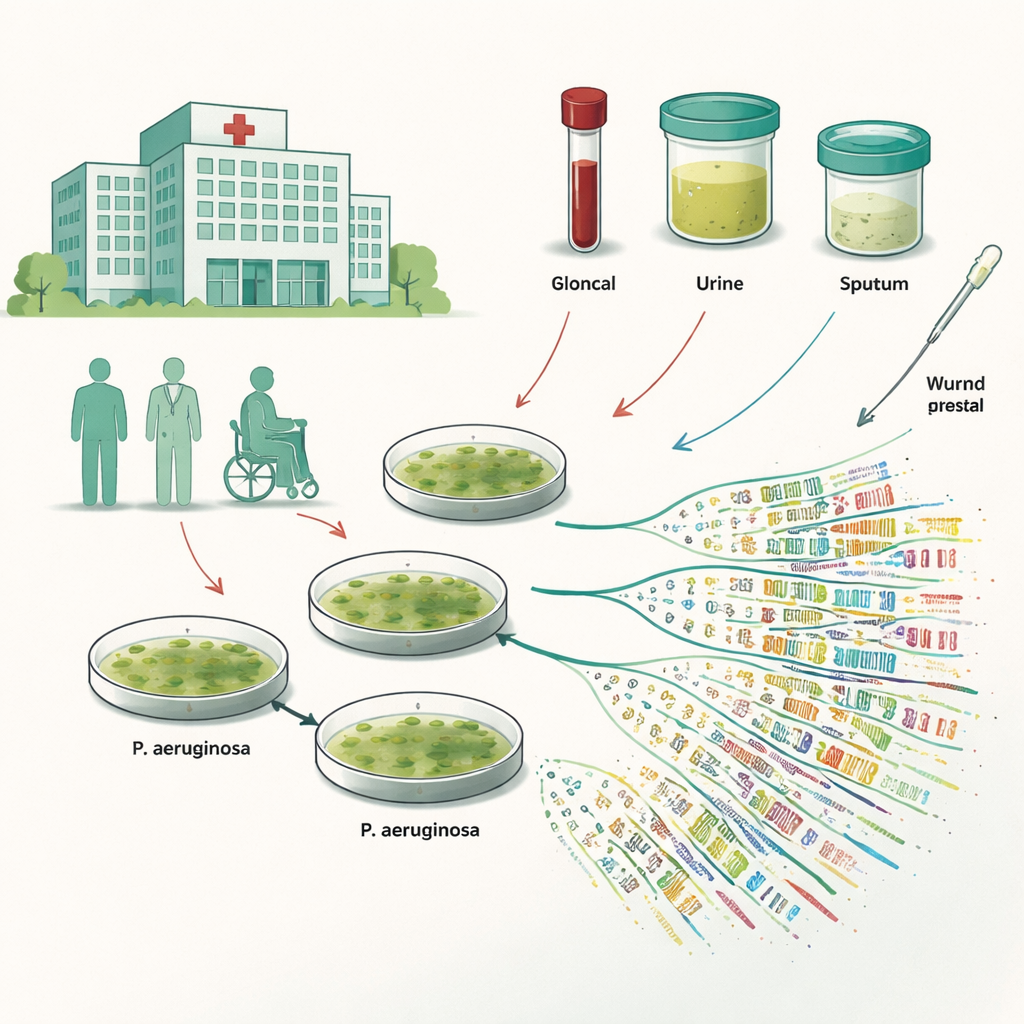
Reading the bacteria’s “bar codes”
Counting which drugs failed was only half the story. The team also wanted to know whether these infections came from one successful “super strain” spreading through the hospital, or from many unrelated lineages arriving and evolving independently. To do this, they used a DNA fingerprinting approach called ISSR, which highlights the stretches of genetic code lying between short repeated sequences. When run through a PCR machine and separated on a gel, these stretches form a pattern of bands that acts like a bar code for each strain. Using 17 informative primers, the researchers generated 95 distinct DNA bands and then compared the patterns across all 18 strains with computer tools that group similar fingerprints together.
Many distant cousins, not one superbug
The genetic comparisons revealed that the hospital was not dealing with a single runaway clone. Instead, the strains fell into several distinct clusters, with similarity scores ranging from quite close relatives to highly distant cousins. Some isolates that behaved similarly in drug tests turned out to be genetically different, while others that were related shared elements of resistance. Principal component plots and tree‑like diagrams reinforced the picture of multiple lineages coexisting in the same facility rather than one dominant strain sweeping through. This diversity likely arises as the bacteria swap genes, mutate and adapt under constant exposure to antibiotics and the human immune system. 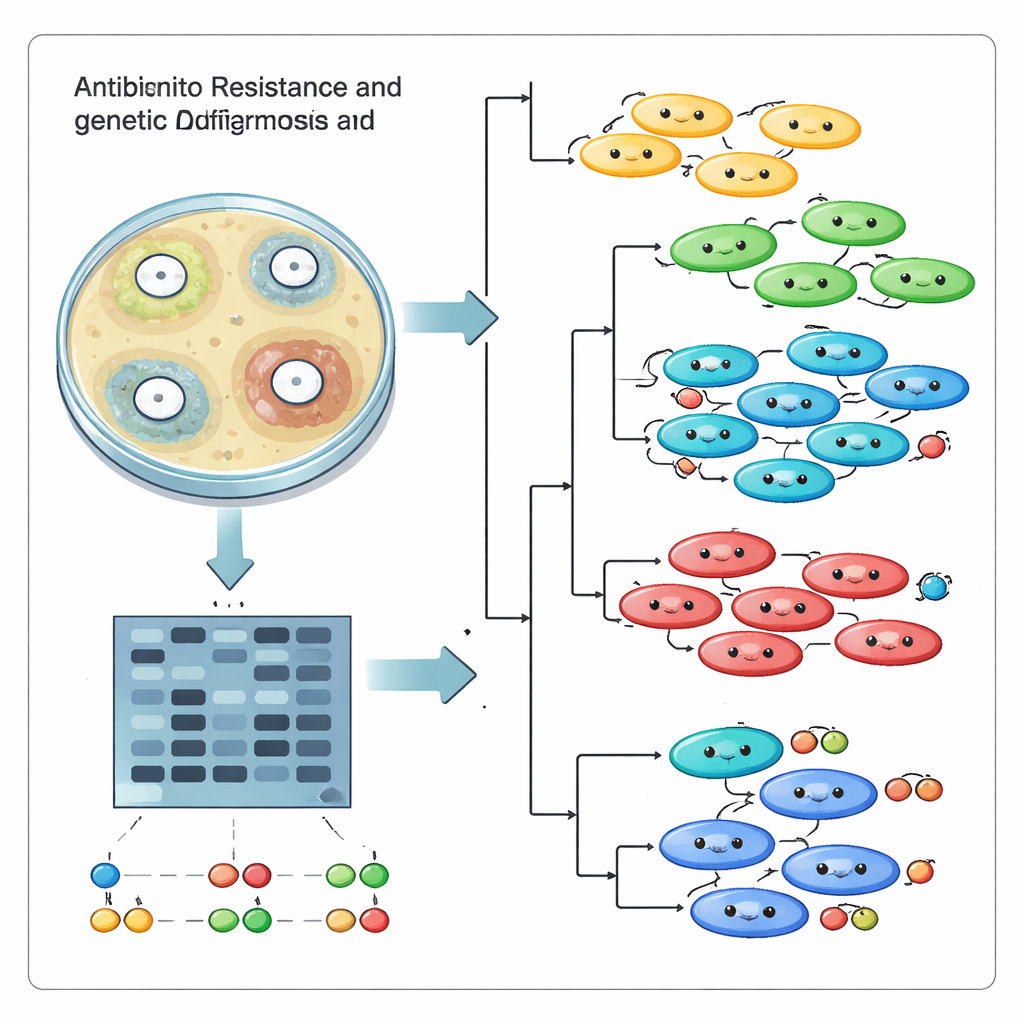
What this means for patients and hospitals
For hospitals, these findings carry a clear message: tracking only which antibiotics fail is not enough. Because genetically distinct strains can share similar resistance patterns—and closely related ones can behave differently—health‑care teams need both routine drug‑susceptibility testing and periodic genetic surveillance to see how the bacterial population is shifting over time. The ISSR approach used here is relatively simple and low‑cost, making it attractive for resource‑limited settings, although the authors stress that combining it with more detailed whole‑genome sequencing in future studies would provide a fuller picture.
A hidden landscape that demands vigilance
In plain terms, this study shows that within a single hospital, Pseudomonas aeruginosa is not one enemy but a crowd of related yet distinct troublemakers, many already armed against multiple antibiotics. By charting this hidden landscape of diversity, the researchers provide tools and insight that can help doctors choose more targeted treatments and infection‑control teams design smarter containment strategies. Continuous monitoring of these genetic patterns will be essential to stay a step ahead of this adaptable germ and to keep hospital care safe for patients.
Citation: Mishra, P., Sahoo, D. & Sahu, M.C. Genetic diversity of Pseudomonas aeruginosa isolated from clinical samples with ISSR molecular marker in a tertiary care teaching hospital. Sci Rep 16, 5315 (2026). https://doi.org/10.1038/s41598-026-35090-8
Keywords: Pseudomonas aeruginosa, antibiotic resistance, hospital infections, genetic diversity, molecular typing