Clear Sky Science · en
Identify and characterize a carbapenem-resistant Salmonella enteritidis phage depolymerase Dpo52
Why this research matters to everyday health
Food poisoning from Salmonella is familiar to many people, but a growing number of these bacteria are now resistant to our strongest "last resort" antibiotics, called carbapenems. Once Salmonella settles into a slimy, protective layer known as a biofilm, it becomes even harder to kill. This study explores an alternative weapon: a virus that infects bacteria and a special enzyme it carries, called Dpo52, that can strip away Salmonella’s protective coating and help stop these dangerous germs from settling in.
A growing problem on our plates
Salmonella enteritidis is one of the most common causes of foodborne illness worldwide, frequently linked to contaminated eggs, poultry, and other animal products. Doctors typically rely on antibiotics to clear these infections, but many Salmonella strains have learned to evade multiple drugs, including carbapenems that are usually reserved for severe cases. One key reason is that the bacteria build biofilms—slimy communities that stick to surfaces such as the gut lining, food-processing equipment, or food itself. Inside these biofilms, bacteria are shielded from antibiotics and from the immune system, turning a routine infection into a much harder problem.
Turning bacterial viruses into helpers
The researchers focused on bacteriophages, or phages for short—viruses that specifically infect bacteria. They isolated a phage from hospital sewage in China that attacks carbapenem-resistant S. enteritidis and named it vB_Sen_S3P. This phage could infect 22 out of 30 patient-derived Salmonella isolates, including the most drug-resistant ones. Electron microscope images revealed a classic phage structure with a geometric head and short tail, and growth tests showed it multiplies rapidly, releasing thousands of new viral particles from each infected bacterium. Genetic sequencing confirmed that this phage lacks known antibiotic resistance or virulence genes, making it a promising candidate for safe therapeutic use.
The special enzyme that peels off the slime
Inside the phage’s DNA, the team identified a gene called ORF52 that likely encodes a depolymerase—an enzyme that cuts apart the long sugar chains forming the bacterial capsule and biofilm matrix. They cloned this gene into laboratory E. coli, produced the protein, and named it Dpo52. Structural predictions suggested that one part of Dpo52 recognizes and binds to Salmonella’s surface, while another part works as a molecular “cutter” for the extracellular sugars. In lab tests, droplets of purified Dpo52 placed on lawns of Salmonella created clear halo zones—evidence that the enzyme was chewing away the protective material around the cells without actually killing the bacteria directly.
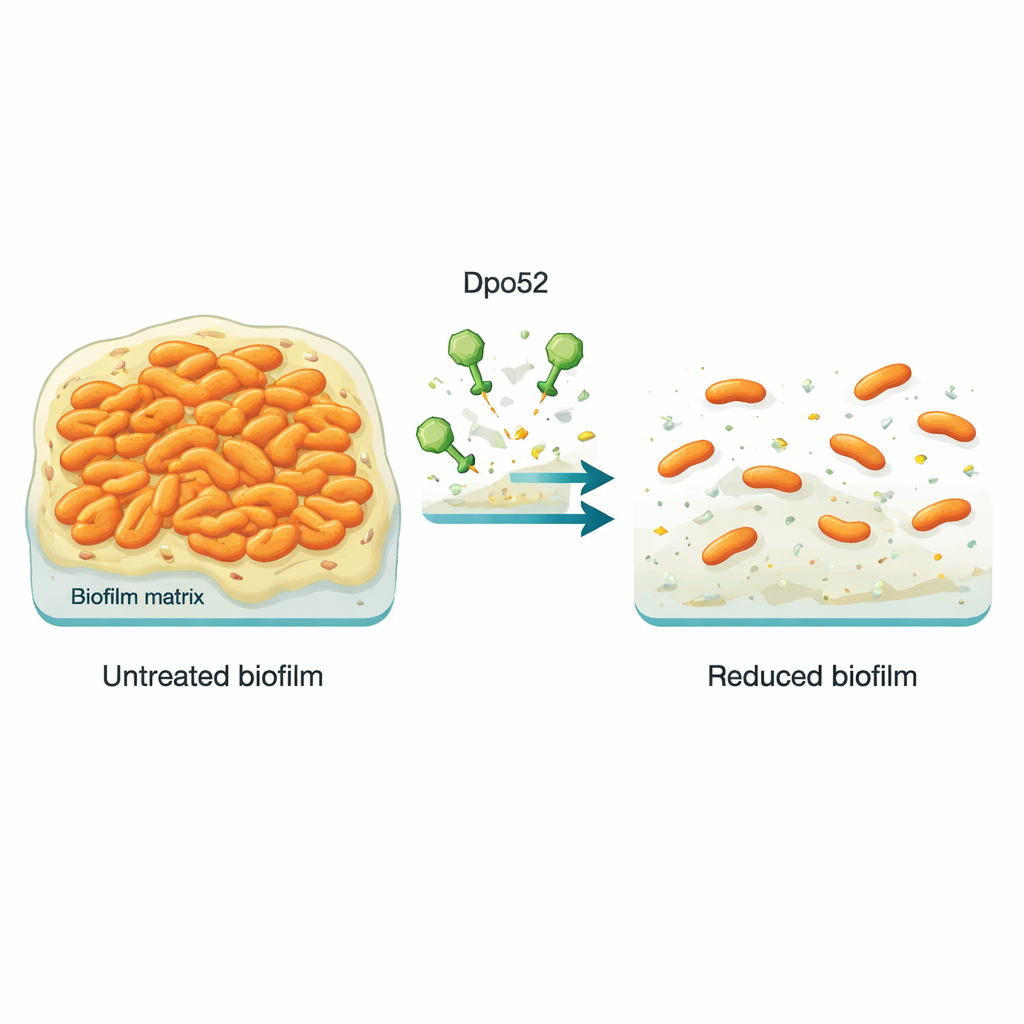
Stopping biofilm before it takes hold
To see whether Dpo52 could stop biofilms from forming, the scientists grew two carbapenem-resistant Salmonella strains in small plastic wells, with and without different doses of the enzyme. After incubation, they stained the wells to measure how much sticky biofilm had accumulated. Wells treated with moderate to high amounts of Dpo52 had far less staining, showing that the enzyme strongly reduced biofilm formation in a dose-dependent manner. However, when biofilms were allowed to mature first and then treated, Dpo52 could no longer break them apart, likely because the dense, multi-layered structure blocked the enzyme from reaching all of the sugar matrix.
Safety, stability, and future use
Dpo52 proved to be robust: it remained active across a wide range of temperatures from refrigerator-cold to 60 °C and from moderately acidic to alkaline conditions. Importantly, tests on human immune cells (macrophage-like THP‑1 cells) showed no detectable toxicity, even at high doses. Microscopy confirmed that Dpo52 removed the pale capsule surrounding Salmonella cells, consistent with its role in digesting extracellular polysaccharides. Taken together, these traits suggest Dpo52 could be added to surfaces, foods, or phage-based therapies as a targeted tool to keep drug-resistant Salmonella from establishing hard-to-treat biofilms.
What this means for fighting tough infections
For lay readers, the key message is that this work identifies a highly specific, non-toxic enzyme that helps strip away the protective “slime coat” of dangerous, drug-resistant Salmonella before it can dig in. While Dpo52 does not dissolve fully mature biofilms, it shows strong potential as a preventive measure—used alone or alongside phages and antibiotics—to keep food and medical environments safer. As scientists refine such enzymes and broaden their activity, they could become an important part of our future toolkit against antibiotic-resistant infections.
Citation: Li, W., Yuan, M., Che, J. et al. Identify and characterize a carbapenem-resistant Salmonella enteritidis phage depolymerase Dpo52. Sci Rep 16, 4906 (2026). https://doi.org/10.1038/s41598-026-35081-9
Keywords: Salmonella, antibiotic resistance, bacteriophage therapy, biofilms, depolymerase enzymes