Clear Sky Science · en
Graphitic carbon nitride–reduced graphene oxide (g-C3N4@r-GO) nanocomposites for photocatalytic hydrogen production by water splitting and high-performance electrochemical supercapacitors
Clean Energy from Everyday Elements
Hydrogen fuel and fast, rechargeable energy storage are often presented as two separate technological challenges. This study shows how both can be tackled at once using a single, metal‑free material made from abundant elements like carbon and nitrogen. By carefully blending a light‑absorbing yellow powder (graphitic carbon nitride) with ultra‑thin carbon sheets (reduced graphene oxide), the researchers created a "two‑in‑one" material that can use sunlight to split water into hydrogen and also act as a high‑performance supercapacitor for storing electrical energy.
Building a Smarter Sponge for Light and Charge
At the heart of the work is a composite called g‑C3N4@r‑GO, where graphitic carbon nitride (g‑C3N4) is combined with sheets of graphene oxide that have been chemically reduced to carry electricity better. On their own, g‑C3N4 absorbs light but conducts electricity poorly, while graphene‑based materials conduct well but do not split water efficiently. By stacking these two in intimate contact, the team creates a kind of electronic p–n junction – a built‑in electric field that helps separate the positive and negative charges generated when light hits the material. They tested two gentle reducing agents, vitamin C (ascorbic acid) and sodium borohydride, to tune how conductive and well‑connected the graphene sheets become. 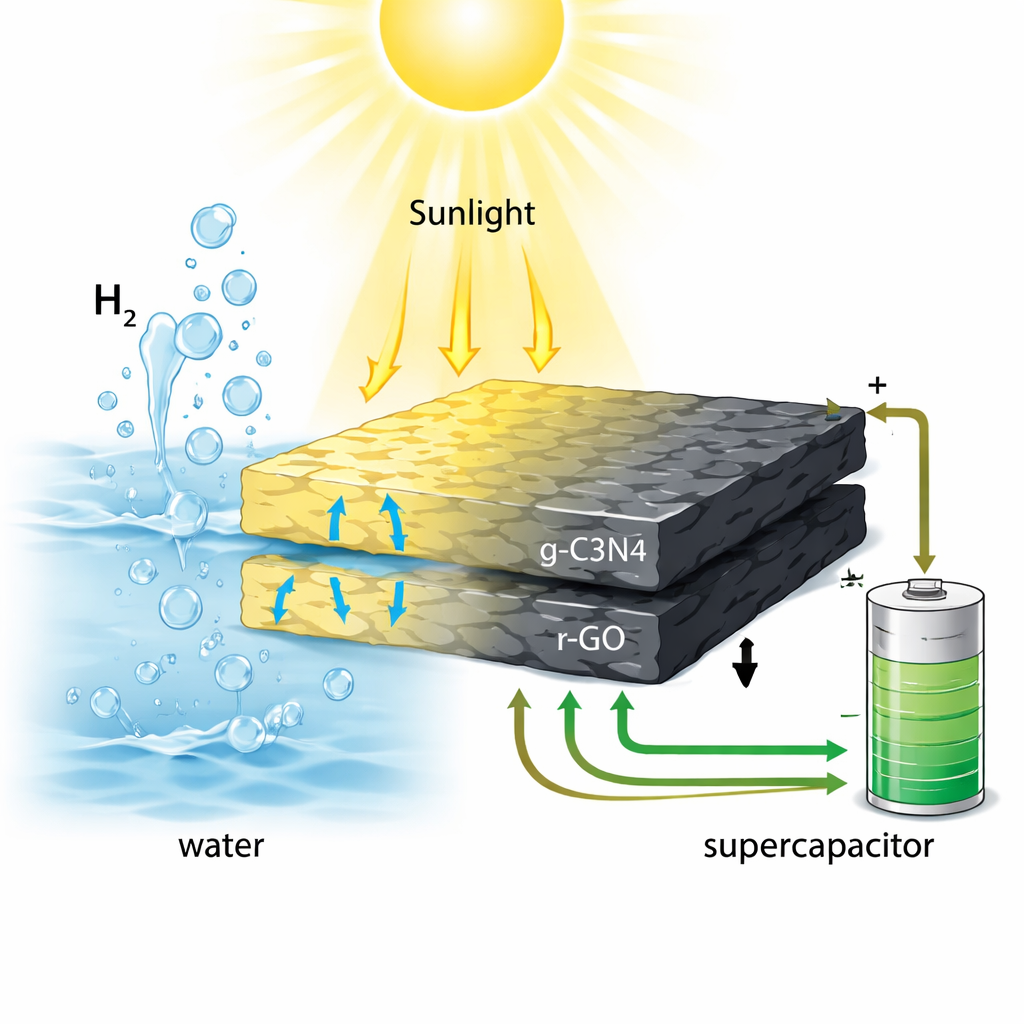
Peering into the Nanoscale Architecture
To understand why one composite outperformed the rest, the authors used a suite of structural and optical tools. Electron microscope images revealed how the powders are built from stacked flakes and rod‑like particles; in one version, shallow pits appear that can trap and recombine charges instead of letting them do useful work. X‑ray diffraction showed how well‑ordered the atomic layers are, while infrared and ultraviolet‑visible spectroscopy revealed how chemical bonds and light‑absorbing features shift when g‑C3N4 is coupled to graphene. The best performer, made with ascorbic acid, had the smallest effective band gap (energy threshold for absorbing light) and signs of strong interaction between the two components, which favors both light harvesting and electron flow.
Turning Light and Water into Hydrogen Fuel
When the composites were placed in water containing a small amount of methanol and illuminated with a xenon lamp, they produced hydrogen gas at very different rates. Pure g‑C3N4 and graphene oxide on their own generated relatively little hydrogen. In contrast, the g‑C3N4@r‑GO material reduced with vitamin C produced 339.82 micromoles of hydrogen per hour per gram of catalyst, with an apparent quantum efficiency of 2.52% at 420 nanometers. That means more than five times more hydrogen than some of its counterparts under the same conditions. Tests over multiple cycles showed that the material retained nearly 90% of its hydrogen‑producing power after three runs, indicating good stability and recyclability without relying on expensive or toxic metals. 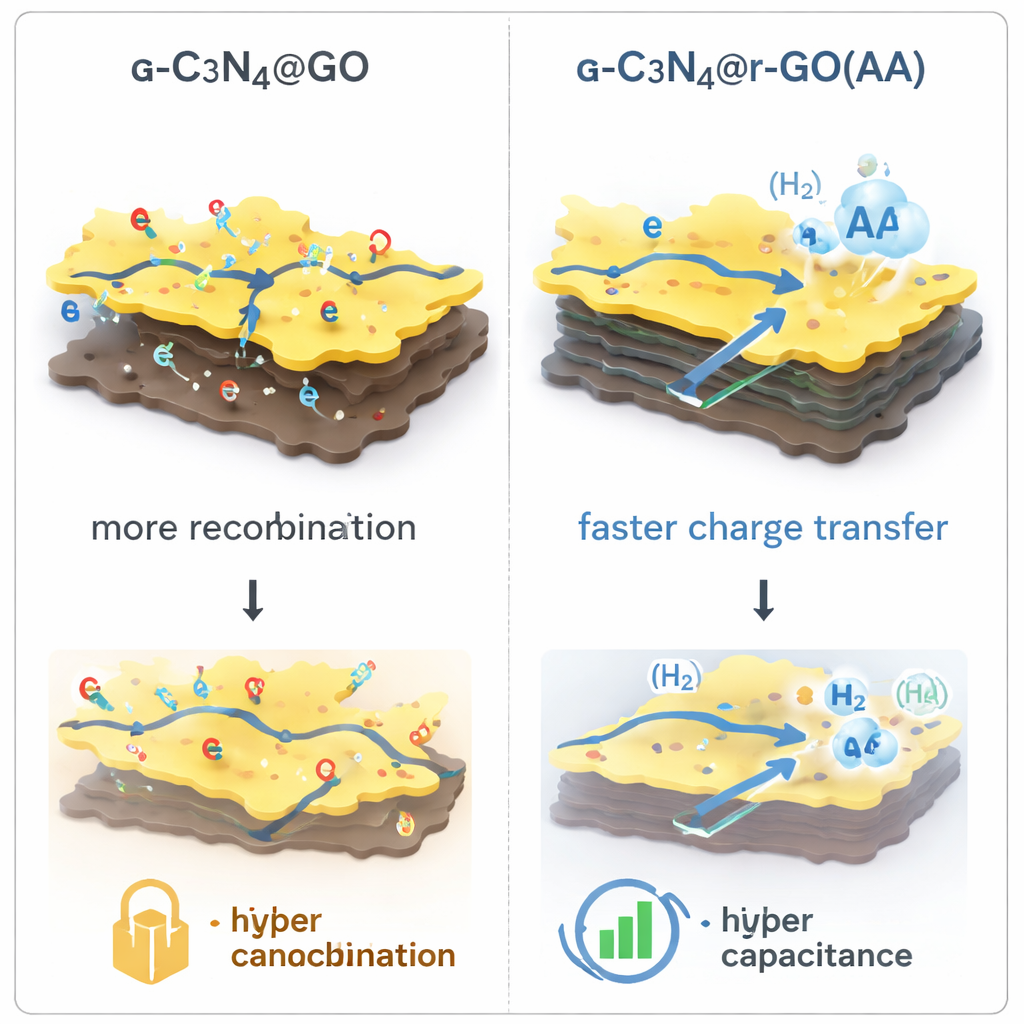
Acting as a High-Speed Energy Reservoir
The same composite was also pressed into electrodes and immersed in alkaline solution to test its performance as a supercapacitor – a device that stores and releases charge very quickly. Using standard electrochemical measurements, the researchers found that the g‑C3N4@r‑GO (ascorbic acid) electrode reached a specific capacitance of about 323 farads per gram at low scan rates, outperforming several related materials reported in the literature. Even after 5000 charge–discharge cycles at relatively high current, it kept almost 79% of its initial capacitance, showing that the structure can withstand repeated use. The graphene layers provide fast pathways for electrons, while nitrogen‑rich sites in the carbon nitride help store charge through reversible reactions with ions in the liquid.
Why This Matters for Future Energy Systems
For non‑specialists, the key message is that carefully designed carbon‑based materials can do double duty in a clean‑energy future: they can help generate hydrogen fuel from water using sunlight and also act as robust, fast‑charging energy storage devices. By avoiding precious or toxic metals and using mild chemistry such as vitamin C reduction, the study points toward cheaper, more sustainable routes to large‑scale hydrogen production and high‑power supercapacitors. While more work is needed on safety, scaling, and integration into real devices, these g‑C3N4@r‑GO composites move us closer to a practical, metal‑free toolkit for both making and storing renewable energy.
Citation: Nagar, O.P., Kameliya, M., Gurbani, N. et al. Graphitic carbon nitride–reduced graphene oxide (g-C3N4@r-GO) nanocomposites for photocatalytic hydrogen production by water splitting and high-performance electrochemical supercapacitors. Sci Rep 16, 5465 (2026). https://doi.org/10.1038/s41598-026-35069-5
Keywords: hydrogen production, water splitting, graphene composite, supercapacitor, solar energy