Clear Sky Science · en
The novel alumina/CQDs nanocomposites for modifying optical and structural properties of alumina nanostructure
Why tiny particles can reshape everyday materials
From water filters to electronics, aluminum oxide—better known as alumina—is a workhorse material. This study explores what happens when alumina is blended with glowing carbon "dots" only a few billionths of a meter across. The result is a new nanocomposite whose structure and light-handling abilities can be tuned simply by how it is made and heated, opening doors to smarter coatings, better water treatment and sensitive chemical sensors.

Building a new kind of nano mixture
The researchers set out to marry two well-known nanoscale ingredients: alumina nanoparticles, valued for their strength and large surface area, and carbon quantum dots, tiny carbon particles that can absorb and emit light. They first created a liquid rich in carbon quantum dots from a common substance, citric acid, using a simple heating and mixing step. This glowing solution was then added directly into a standard recipe for making alumina, so that the carbon dots formed and became embedded as the alumina particles precipitated from water. The resulting powder, called AQD, was studied as made, and again after a two-hour heat treatment at 550 °C, producing a second sample called CAQD.
Seeing and sizing the glowing carbon dots
Before looking at the final composite, the team carefully examined the carbon quantum dots in the starting liquid. Under ultraviolet light the solution shines green-blue, a hallmark of such dots. Measurements of the emitted light showed two main colors: visible green and a stronger near‑infrared glow, consistent with earlier work on carbon dots that contain tiny graphitic regions and surface defects. Electron microscope images revealed that the dots are roughly spherical, only about 2.5 nanometers across—so small that their size directly controls the color they emit. Additional tests confirmed that the dots are mostly carbon and oxygen, with a largely disordered, carbon‑rich structure decorated with oxygen‑containing chemical groups, features that are known to support strong and tunable optical behavior.
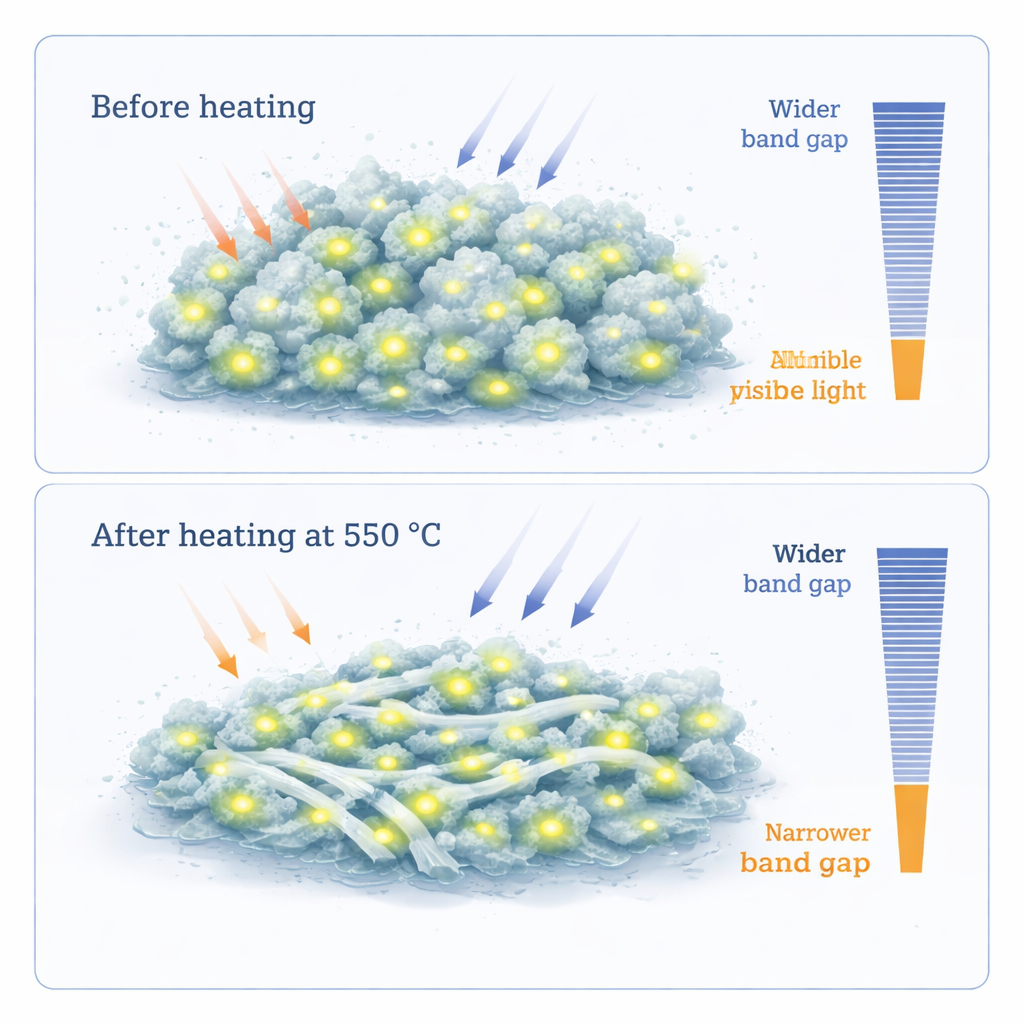
How heating reshapes structure at the nanoscale
Once the carbon‑dot‑loaded alumina powders were made, the team used a suite of techniques to see how their internal structure changed with heat. Infrared and Raman spectroscopy showed the fingerprints of both alumina bonds and carbon-related groups, while X‑ray diffraction revealed that the as‑made composite is mostly amorphous—its atoms lacking long‑range order. After heating to 550 °C, the alumina regions partly crystallize and some of the carbon is burned away, but a significant fraction of carbon remains, now more firmly embedded. Electron microscope images show both small, nearly spherical particles and thin, strand‑like structures, with average sizes on the order of 8–12 nanometers. Heating causes the particles to grow slightly and the strands to lengthen, yet the overall distribution stays narrow and uniform.
Light reflection, band gaps and internal surface area
The optical tests reveal one of the most striking outcomes. Both the as‑made and heated composites reflect a large fraction of light from the near‑ultraviolet through the entire visible and into the near‑infrared range (about 300–1200 nanometers), making them excellent broad‑band reflectors. At the same time, careful analysis of the reflected light shows that adding carbon dots narrows the material’s effective "band gap"—the energy needed for electrons to jump and carry current under illumination. In the as‑made sample, extra low‑energy transitions appear, linked to electronic states introduced by the carbon dots and their defects, while the heated sample settles into a slightly wider but still reduced band gap compared with pure alumina. Gas-adsorption measurements further show that both versions of the composite are highly porous, with extremely large internal surface areas (over 200 square meters per gram) and pores in the nanometer range, ideal for trapping molecules or hosting reactions.
Where these designer particles might be used
Put simply, the study demonstrates a straightforward way to weave light‑responsive carbon dots into a robust alumina framework, then fine‑tune the result with heat. For non‑experts, the key message is that this recipe yields a white, highly porous powder that strongly reflects light over a broad range yet has its electronic properties adjusted by its carbon content. Such a combination—large internal surface, controllable light absorption and strong reflectance—makes these alumina/carbon‑dot nanocomposites promising for cleaner water through photocatalytic treatment, optical coatings that manage heat and glare, and chemical or gas sensors that respond more readily to their environment. The work shows how tweaking matter at the scale of billionths of a meter can quietly upgrade materials that underpin many technologies we rely on every day.
Citation: Gholizadeh, Z., Aliannezhadi, M., Ghominejad, M. et al. The novel alumina/CQDs nanocomposites for modifying optical and structural properties of alumina nanostructure. Sci Rep 16, 4837 (2026). https://doi.org/10.1038/s41598-026-35063-x
Keywords: alumina nanocomposite, carbon quantum dots, photocatalytic water treatment, optical reflector materials, high surface area nanoparticles