Clear Sky Science · en
Study on CO2/CH4 displacement process in shale microscale models with adsorption/desorption behavior by lattice Boltzmann method
Turning a Climate Problem into a Helpful Tool
Burning fossil fuels releases carbon dioxide (CO2), the main driver of climate change. At the same time, much of the world’s natural gas is locked in dense rocks called shale, where it is difficult to extract. This study explores a technology that tries to tackle both issues at once: using CO2 to push methane (the main component of natural gas) out of shale while trapping the CO2 underground. By peering into the rock at the scale of billionths of a meter, the authors show how injected CO2 can free methane from the rock’s tiny pores and improve gas recovery, while potentially storing CO2 in the process.
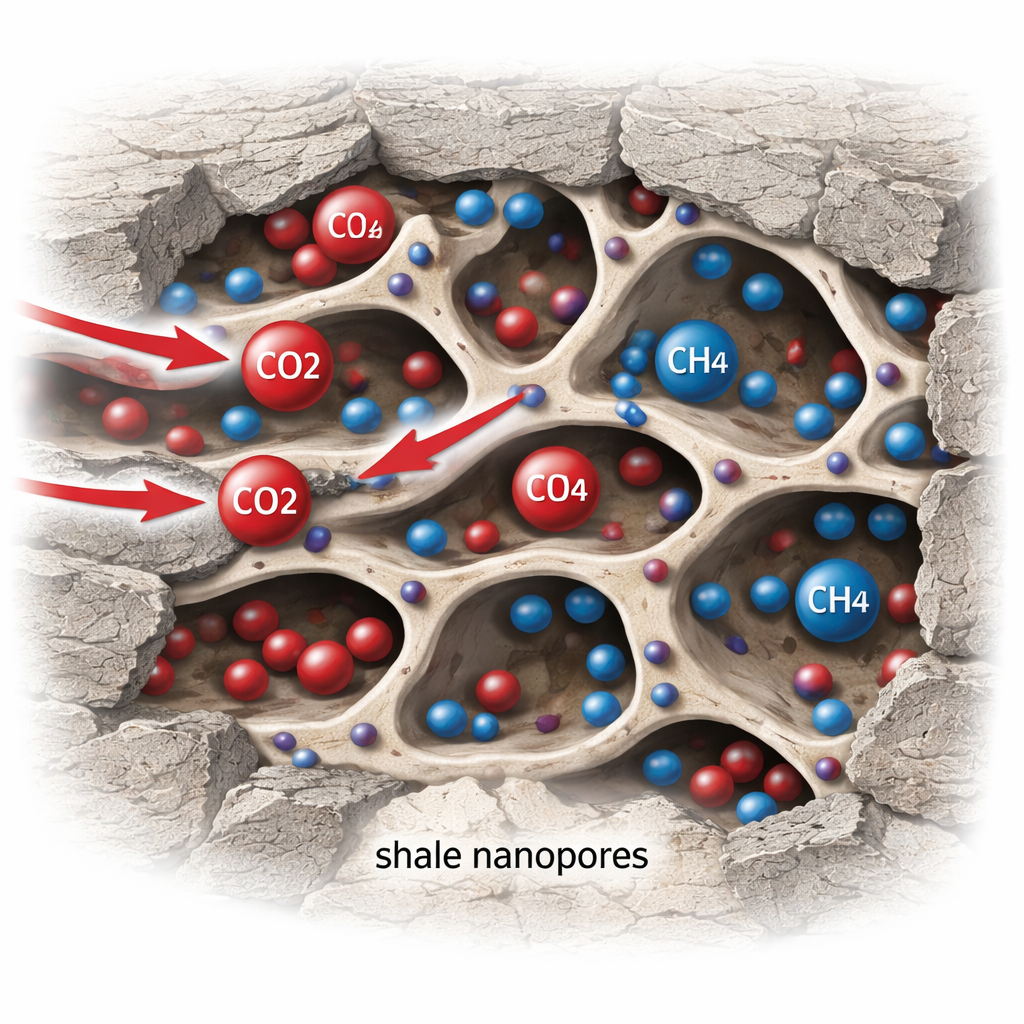
Gas in Tiny Pores Beneath Our Feet
Shale rocks are riddled with nanoscale pores—spaces so small that a human hair would look enormous by comparison. These pores act as both storage tanks for methane and potential hiding places for CO2. Inside them, gas exists in two main forms: as free molecules moving through the pore spaces, and as molecules that cling to the rock surfaces in a thin layer. In such cramped conditions, gas does not flow like water in a pipe; instead, movement is controlled by a mix of sticking, unsticking, and slow diffusion. To understand whether CO2 can realistically displace methane from these pores, it is essential to model not just how gases flow, but also how they compete to attach to and detach from the pore walls.
A Virtual Microscope for Gas Flow
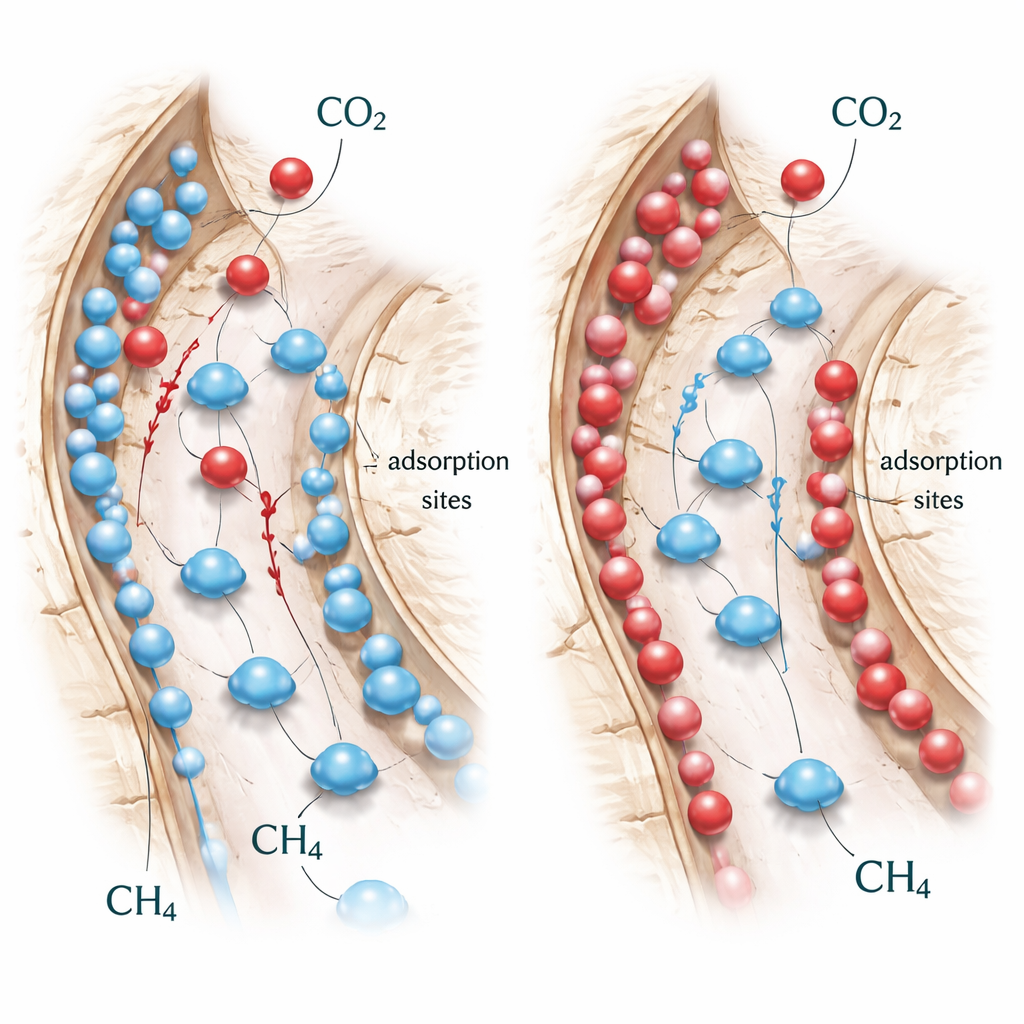
Directly watching how gases move inside shale at this tiny scale is extremely difficult in the lab, so the researchers turned to a numerical tool called the lattice Boltzmann method. This method treats fluids as many small packets moving and colliding on a grid, allowing computers to reconstruct how gas flows through complex networks of pores. The team first built a mathematical description of how two gases—CO2 and methane (CH4)—compete for the same surface spots in a single molecular layer. Their model captures both adsorption (molecules sticking to the rock) and desorption (molecules leaving the surface), and how these processes respond to gas concentration and pressure. They then wove this competition model into lattice Boltzmann simulations of gas flow and diffusion inside simplified but realistic shale-like pore structures.
Watching CO2 Push Methane Out
Using this virtual rock, the authors simulated what happens when CO2-rich gas is injected into a pore system initially saturated with methane. In a single-particle test case, CO2 entering from one side quickly attaches to the “upstream” face of the grain, sharply increasing its adsorption rate. At the same time, methane already on the surface is forced to let go and diffuse into the nearby gas, then drift downstream with the flow. Over time, the methane content inside the particle steadily falls to nearly zero, while CO2 content rises until both adsorption and desorption balance out. The study identifies two stages in this process: an early competitive stage when both gases are rapidly exchanging places, followed by a slower approach to an equilibrium where CO2 remains on the surface and methane has largely departed.
How Injection Strength and Rock Structure Matter
The simulations show that the amount of CO2 in the injected gas strongly controls how quickly and how completely methane is displaced. With no CO2 injection, methane desorbs only slowly. As the CO2 concentration rises, methane is released faster, the CO2 layer builds up more quickly on the rock, and the system reaches equilibrium sooner. The structure of the rock also plays a key role. In porous models with more open space (higher porosity), gas can move and diffuse more easily, so CO2 sweeps through the pore network faster and displaces methane more efficiently. The study also finds that flow speeds vary sharply in different parts of the pore network, and that regions rich in CO2 tend to be poor in methane, both in the flowing gas and on the solid surfaces, highlighting a clear one-for-one replacement pattern.
What This Means for Energy and Climate
For non-specialists, the bottom line is that this work provides a detailed picture of how CO2 can physically push methane out of shale at the microscopic level. The model suggests that injecting CO2 at higher concentrations into suitable shale formations could both boost natural gas production and promote long-term storage of CO2 by binding it to the rock’s inner surfaces. While real reservoirs are more complex than any computer model, these results strengthen the scientific basis for CO2-enhanced shale gas recovery as a dual-purpose technology: one that taps hard-to-reach gas resources while also helping to keep carbon dioxide out of the atmosphere.
Citation: Zhang, Y., Xu, Y., Chen, X. et al. Study on CO2/CH4 displacement process in shale microscale models with adsorption/desorption behavior by lattice Boltzmann method. Sci Rep 16, 5033 (2026). https://doi.org/10.1038/s41598-026-35062-y
Keywords: shale gas, carbon dioxide storage, enhanced gas recovery, methane displacement, porous media modeling