Clear Sky Science · en
Crystal structure of Echinococcus multilocularis cystatin B reveals a novel feature in classical stefins
Why a liver parasite’s secret weapon matters
Alveolar echinococcosis is a mouthful of a name for a tiny tapeworm infection that behaves a lot like a slow-growing cancer in the liver. It is often deadly, and current drugs work poorly and can have serious side effects. This study looks at one particular molecule made by the parasite, a protein called EmCystatin-B, to understand how it might help the worm evade our defenses—and how it could someday be turned into a target for new treatments.
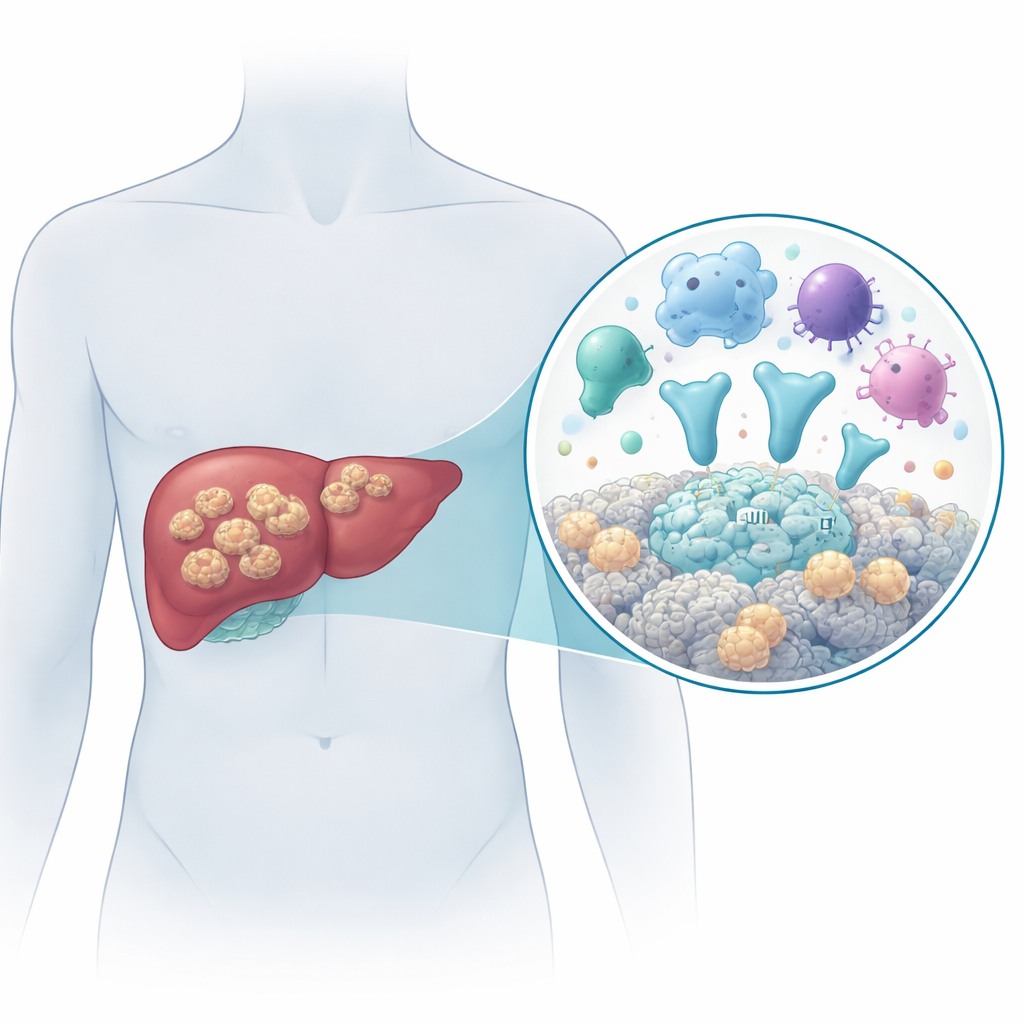
A dangerous tapeworm that grows like a tumor
The disease is caused by the larval stage of the tapeworm Echinococcus multilocularis. People become infected by accidentally swallowing parasite eggs, for example in contaminated food or water. Once inside the body, the larvae settle mainly in the liver and grow as many tiny fluid-filled sacs that infiltrate surrounding tissue, spread to nearby organs such as the spleen, and sometimes reach distant sites like the lungs or brain. Without effective treatment, more than 9 out of 10 patients die within 10 to 15 years. Surgery combined with long-term antiparasitic drugs is currently the best option, but early diagnosis is difficult and the medicines do not reliably cure the infection. That makes it crucial to understand how the parasite interacts with the human body at the molecular level.
A parasite protein that disarms cutting enzymes
Many animals, including humans, produce a family of proteins called cystatins that act as safety locks on powerful digestive enzymes known as cysteine proteases. These enzymes can chop up other proteins and are important for processes like breaking down worn-out cell parts and activating immune responses. Parasites have learned to make their own cystatin-like proteins to interfere with the host’s enzymes and immune system. In this work, the researchers identified a cystatin-like protein from E. multilocularis and named it EmCystatin-B. They found that the gene for EmCystatin-B is much more active in the parasite’s mature larval heads (protoscoleces) than in the surrounding cyst tissue, and that the protein is present in the cytoplasm and nucleus of the parasite’s cells, especially in dividing “stem-like” germinative cells. Laboratory tests showed that EmCystatin-B can strongly block the activity of human cathepsin B, an important cysteine protease involved in immunity and tissue remodeling.
A familiar shape with an unexpected twist
To see exactly how EmCystatin-B is built, the team crystallized the purified protein and used X‑ray crystallography to solve its three‑dimensional structure at near‑atomic resolution. Like other members of the cystatin family, each EmCystatin-B unit has a characteristic shape: a central helical segment backed by several flat, ribbon-like strands, forming a wedge that normally fits into the active site of a protease. Based on its sequence and lack of an obvious signal peptide for secretion, EmCystatin-B belongs to the “stefin” subgroup of cystatins, which are usually small, intracellular proteins that do not contain stabilizing disulfide bonds. Surprisingly, the structure revealed that although each EmCystatin-B unit has the expected fold, the way these units associate with each other is unusual and species‑specific compared with the human counterpart, cystatin B.
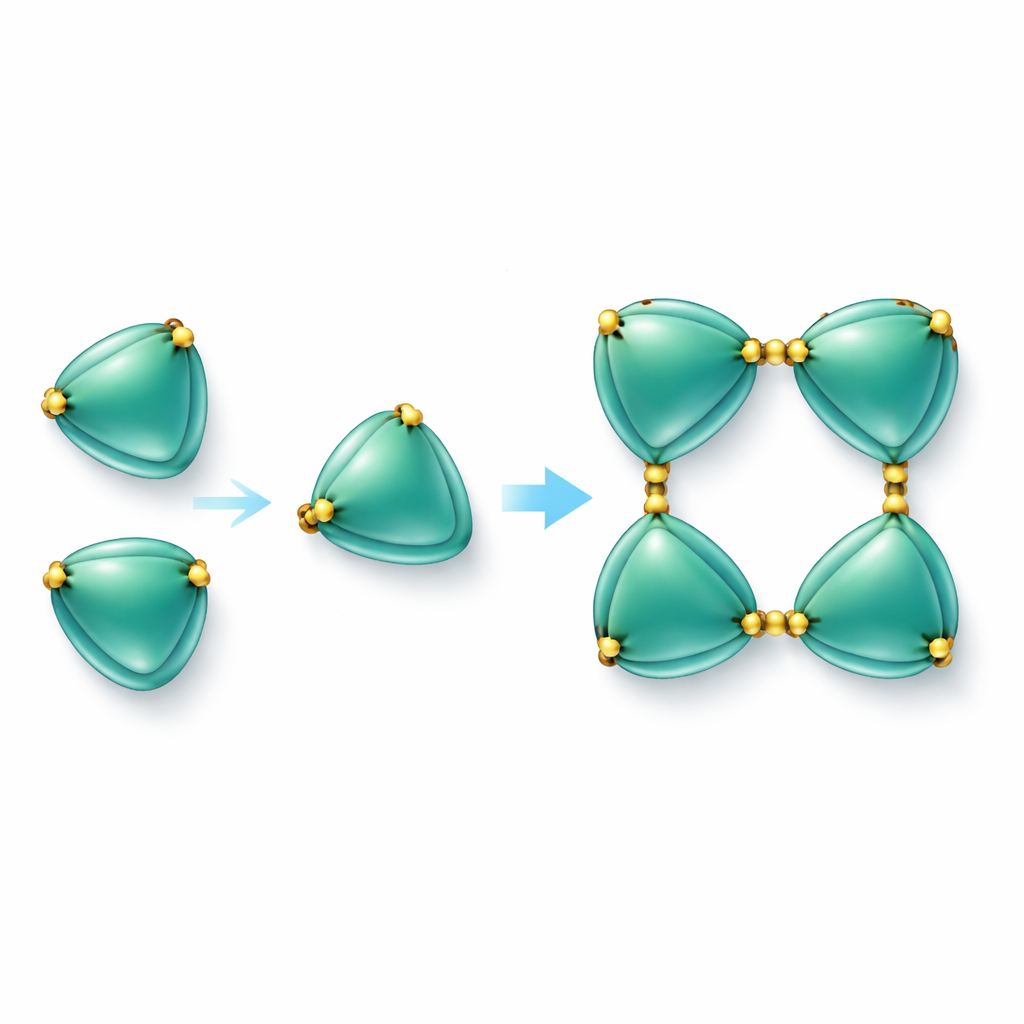
Building protein chains with sulfur links
The most striking finding was that EmCystatin-B molecules do not simply float around as single units. Instead, they can join together in a stepwise fashion to form dimers (pairs) and then tetramers (groups of four). Two different mechanisms help form the initial dimers. In one, a part of one molecule swings over and completes the structure of its neighbor, a phenomenon known as domain swapping. In the other, pairs of molecules are tied together by disulfide bridges—strong chemical links between sulfur-containing amino acids (cysteines). In EmCystatin-B, cysteine at position 4 in one molecule bonds to cysteine at position 76 in a partner molecule. Four such cross-links then stabilize the tetramer. This use of multiple intermolecular disulfide bridges is completely new for classical stefins, which typically lack such bonds. When the researchers changed these key cysteines to another amino acid, the protein could no longer assemble efficiently into higher-order complexes and its ability to inhibit cathepsin B dropped sharply.
What this means for fighting infection
To a non-specialist, the message is that the parasite has evolved a specially wired version of a common protein family, using extra chemical “rivets” to lock several inhibitor units together. This oligomeric form appears important for shutting down a host enzyme that would otherwise help control the infection. By uncovering the precise architecture of EmCystatin-B and its unique disulfide-linked assemblies, the study expands our understanding of how cystatin proteins can be tuned across species. In the long run, these insights could guide the design of drugs that either block EmCystatin-B’s protective effect on the parasite or mimic its immune-calming properties to treat inflammatory diseases.
Citation: Hong, W., Cheng, Z., Xu, Z. et al. Crystal structure of Echinococcus multilocularis cystatin B reveals a novel feature in classical stefins. Sci Rep 16, 5114 (2026). https://doi.org/10.1038/s41598-026-35060-0
Keywords: alveolar echinococcosis, Echinococcus multilocularis, cystatin B, protein structure, immune modulation