Clear Sky Science · en
Disruption of NLRP3 inflammasome assembly via ligand-induced remodeling of pyrin domain interfaces
Why calming runaway inflammation matters
Many chronic diseases—from arthritis and gout to heart disease and neurodegeneration—are fueled by an overactive immune alarm system inside our cells. A key part of this alarm, called the NLRP3 inflammasome, helps us fight infections but can also drive damaging inflammation when it misfires. This study describes a new small molecule, named E9, that targets an early step in NLRP3 activation, offering a fresh angle for designing medicines that cool harmful inflammation without shutting down the immune system entirely.
A cellular fire alarm with a dark side
When our bodies sense danger, immune cells assemble the NLRP3 inflammasome, a large protein complex that acts like a molecular fire alarm. Once formed, this structure switches on an enzyme called caspase-1, which in turn activates powerful inflammatory messengers, including the cytokines IL-1β and IL-18. These signals are vital for fighting infections and clearing damaged tissue. But if the inflammasome stays switched on for too long or turns on at the wrong time, it contributes to chronic inflammatory conditions affecting the joints, blood vessels, brain, and other organs. That is why researchers worldwide are searching for precise ways to control NLRP3 without broadly suppressing immunity.
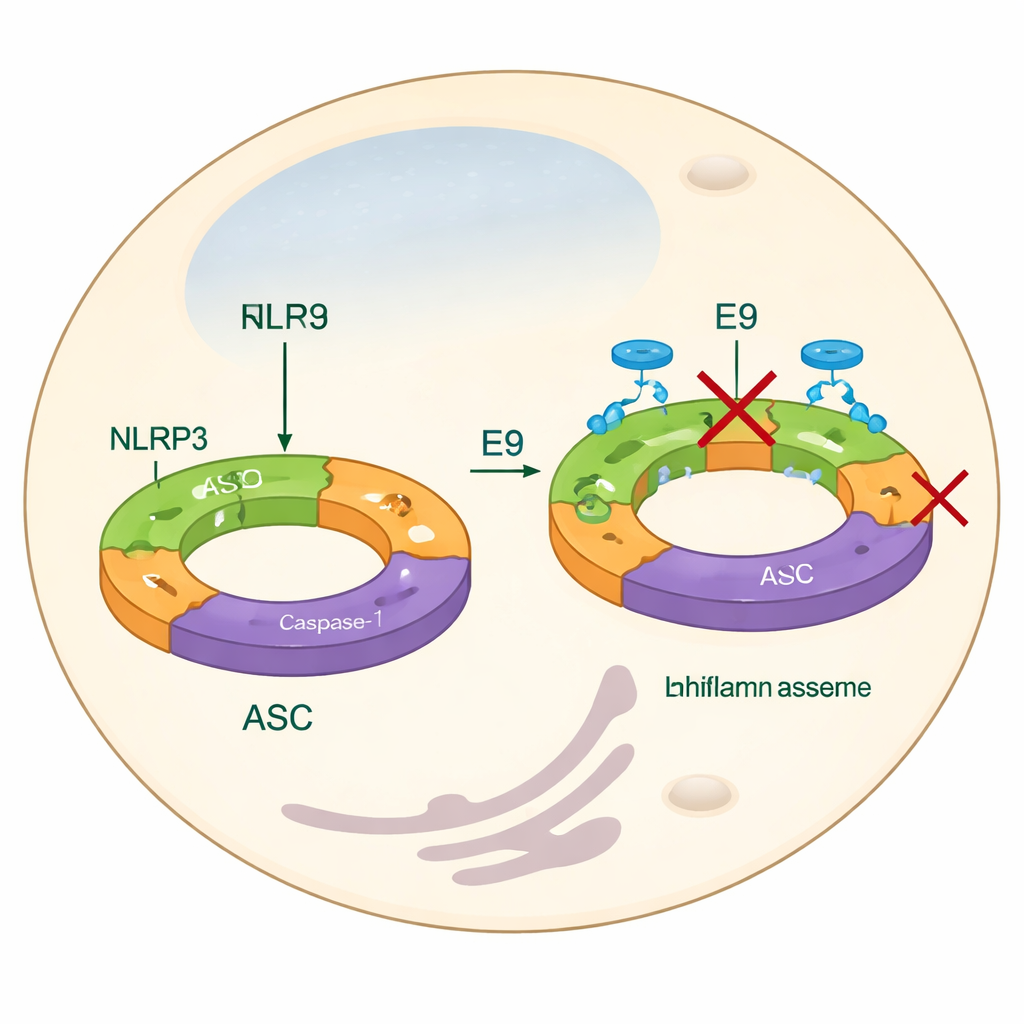
Looking beyond the usual drug targets
Most current strategies for taming NLRP3 focus on blocking its end products—like IL-1β—or on interfering with a central engine-like domain that uses energy molecules such as ATP. While these approaches can work, they may miss the earliest assembly steps and do not fully address how NLRP3 proteins join together to form a functional machine. In this work, the authors instead home in on the pyrin domain, a part of NLRP3 that helps individual units snap together into long filaments. These filaments then recruit another adaptor protein, ASC, which clumps into bright “specks” inside cells—a visual hallmark of inflammasome activation. By finding chemicals that prevent pyrin domains from fitting together properly, the team aims to stop the inflammasome from forming in the first place.
Finding a molecule that breaks the chain
The researchers screened a library of 1500 newly synthesized small molecules using a clever light-based test. They fused split pieces of a luciferase enzyme to the NLRP3 pyrin domain; when two pyrin domains came together, the enzyme fragments reassembled and produced light. Compounds that blocked pyrin–pyrin contact caused the light signal to drop. From this screen they identified four promising candidates, with one—E9—standing out for its strong and consistent inhibition. Follow-up measurements using a technique called microscale thermophoresis showed that E9 bound tightly to the pyrin domain at very low (submicromolar) concentrations, confirming that its effect was not a random artifact but the result of a specific interaction.
Seeing the effect inside living cells
To test whether E9 could also work in a living system, the team turned to human immune-like cells engineered to make a fluorescent version of ASC. When these cells are stimulated with bacterial components and a second trigger, NLRP3 switches on and ASC proteins cluster into bright specks that can be counted under the microscope. In the presence of E9, the number of these specks dropped markedly, comparable to or better than some known NLRP3 blockers. This showed that interfering with pyrin domain contacts is enough to reduce inflammasome assembly and the early steps of inflammatory cell death, at least under laboratory conditions.
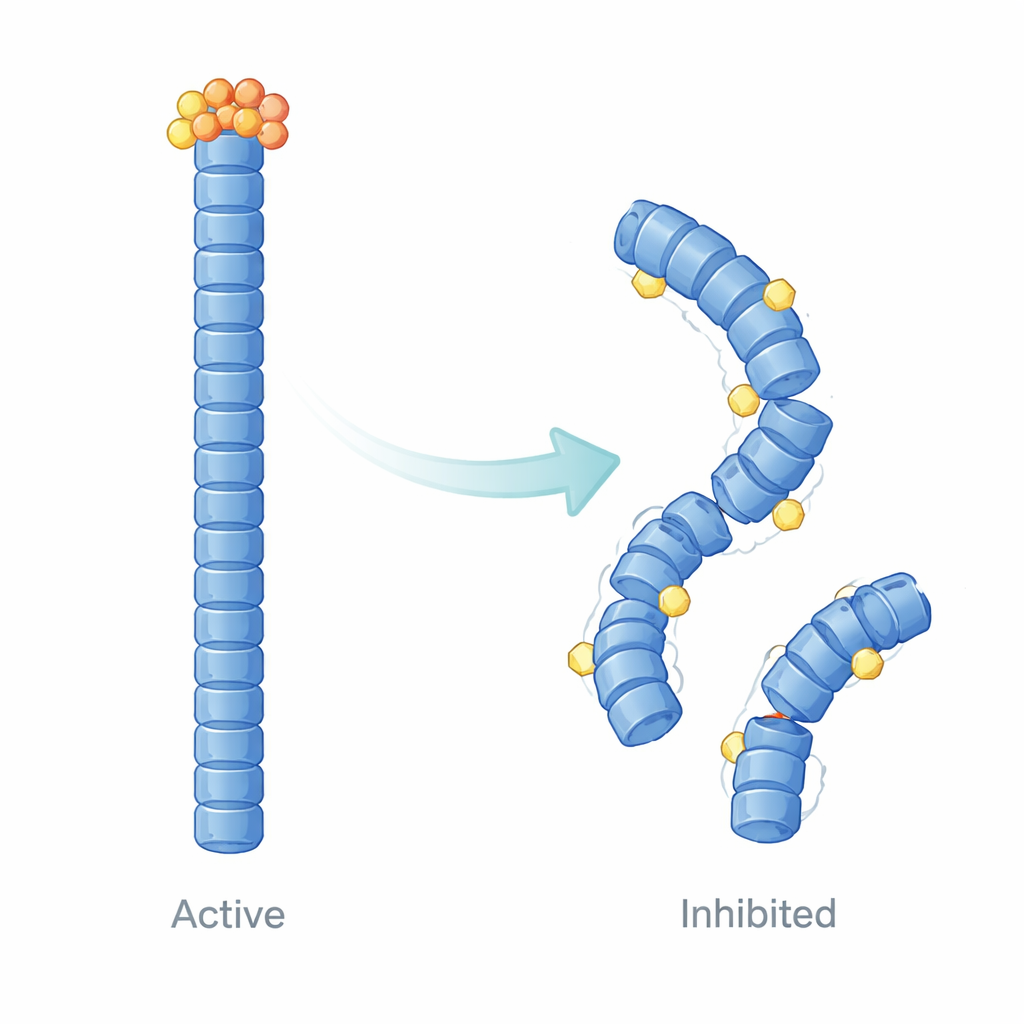
How E9 reshapes the inflammasome scaffold
Beyond simply measuring binding, the authors used computer-based structural modeling and molecular dynamics simulations to visualize how E9 changes the behavior of NLRP3 filaments over time. Their calculations suggest that E9 lodges at specific contact surfaces where pyrin domains touch, subtly loosening and reshaping the filament. Instead of a uniformly stable, cooperative structure, the filament becomes uneven: regions near the bound E9 grow more flexible and lose their precise alignment, while other parts become dynamically isolated. These shifts dampen the large-scale motions and tight communication needed for efficient ASC recruitment, providing a physical explanation for the reduced speck formation seen in cells.
What this could mean for future therapies
Taken together, the findings position E9 as a prototype for a new class of inflammasome-targeting drugs that act by disrupting the interfaces between NLRP3 building blocks rather than by blocking its energy use or final products. Although the current work is limited to test-tube assays, computer simulations, and cultured cells—and further studies in animals and humans are needed—it demonstrates that carefully designed small molecules can remodel the inflammasome scaffold itself. For patients with diseases driven by runaway inflammation, such interface-focused drugs could someday offer more precise control, dialing down harmful immune activation while leaving essential defenses largely intact.
Citation: Khosravifard, S., Hosseinkhani, S., Bakhtiary, N. et al. Disruption of NLRP3 inflammasome assembly via ligand-induced remodeling of pyrin domain interfaces. Sci Rep 16, 4951 (2026). https://doi.org/10.1038/s41598-026-35050-2
Keywords: NLRP3 inflammasome, pyrin domain, small molecule inhibitor, innate immunity, pyroptosis