Clear Sky Science · en
Hepatitis virus-associated B cell non-Hodgkin’s lymphoma involves dysregulated epigenetic and RNA-mediated regulatory gene expression and altered snoRNA transcription
Why liver viruses matter to the immune system
Hepatitis B and D viruses are best known for damaging the liver, but growing evidence suggests they can also help trigger certain blood cancers. This study explores how chronic infection with these viruses might change the behavior of B cells—key defenders in our immune system—in ways that increase the risk of B cell non-Hodgkin’s lymphoma, a group of cancers that arise from these very cells.
Following patients where infections are common
To investigate these links, researchers studied patients in Mongolia, a country with some of the world’s highest rates of chronic hepatitis B and D. They collected blood samples from five groups: people with hepatitis B alone, people with both hepatitis B and D, patients who had both infection and B cell lymphoma, patients with lymphoma but no viral infection, and healthy volunteers. From these samples, they isolated peripheral B cells—the circulating B cells that patrol the bloodstream—and used RNA sequencing to read out which genes were turned on or off in each group.
Immune cells that look switched down
When the team compared gene activity patterns, they found that B cells from chronically infected patients carried a signature of broad immune dampening. Compared with B cells from healthy people and from lymphoma patients without infection, infected patients’ B cells showed reduced activity in gene networks linked to immune activation and response to inflammatory signals. Analyses of upstream regulators suggested that important immune stimulators, such as key cytokines and B cell receptor pathways, were effectively being held back, while several molecules that inhibit these signaling routes were more active. Overall, the data paint a picture of B cells that are less responsive and more suppressed during long-term hepatitis infection.
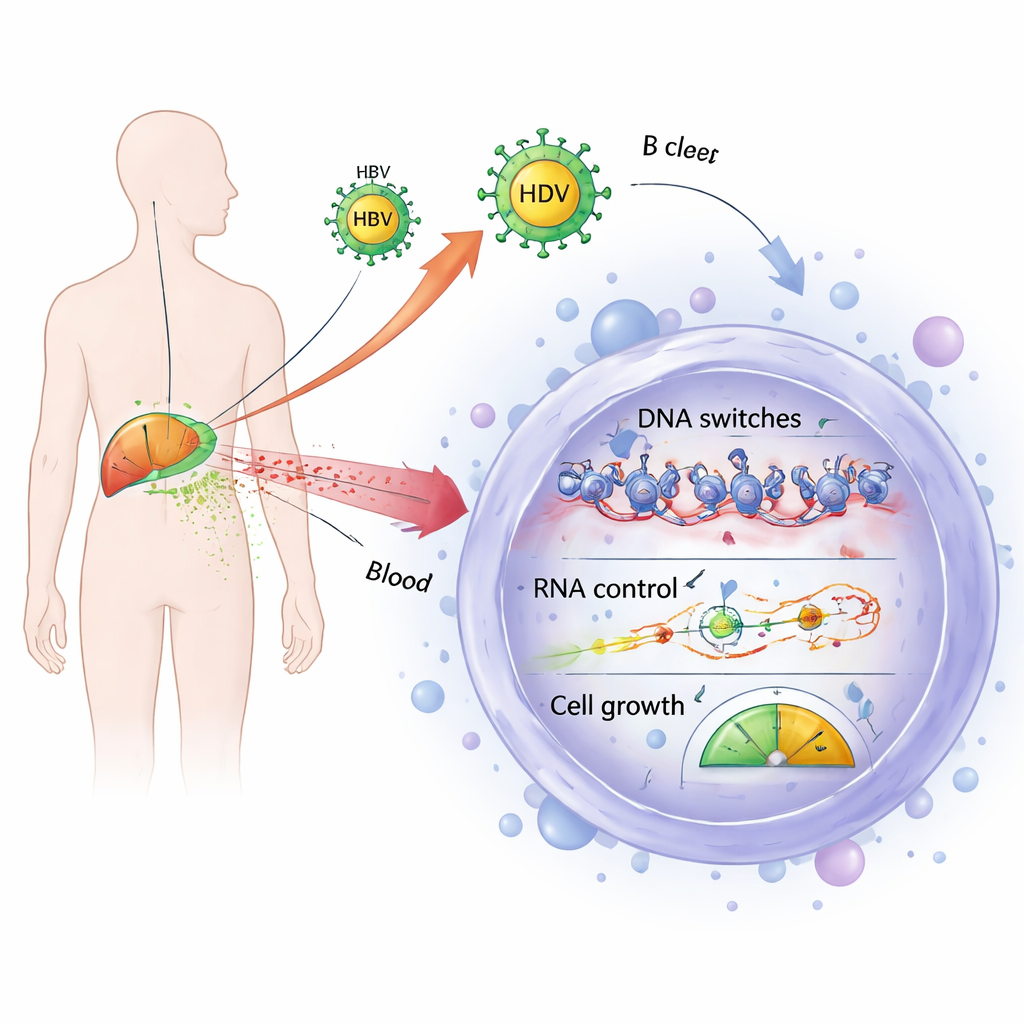
Shared gene control problems in infected and cancerous B cells
The researchers then focused on genes that were consistently more active in all disease groups—infected patients and lymphoma patients alike—compared with healthy donors. They identified 185 such genes and found that many are involved in controlling how DNA is packaged and read (epigenetic regulation) and how RNA messages are processed and silenced. This included multiple components of chromatin-remodeling complexes and enzymes that add or remove chemical marks from histone proteins, which shape access to genes. It also included key players in the machinery that microRNAs use to shut down target messages. Because normal B cell development depends on precise control of these systems, their overactivation in both virus-associated and unrelated lymphomas suggests a common pathway by which B cells may be nudged toward malignant transformation.
Unusual boosts in tiny RNAs inside infected B cells
One of the most striking findings was specific to virus-infected patients: a broad increase in small nucleolar RNAs, or snoRNAs, in their B cells. These tiny RNA molecules usually live in the cell’s nucleolus, where they help modify ribosomal RNA and build ribosomes, the factories that make proteins. In hepatitis B and D infection, these viruses are known to hijack nucleolar functions to support their own replication. Here, the team found 69 distinct snoRNAs whose activity changed, mostly increasing in infected B cells, especially in those with hepatitis B alone. Many of these snoRNAs have already been linked to cancers in other organs, sometimes acting like oncogenes or tumor suppressors, and some can directly alter the stability or translation of specific messenger RNAs.
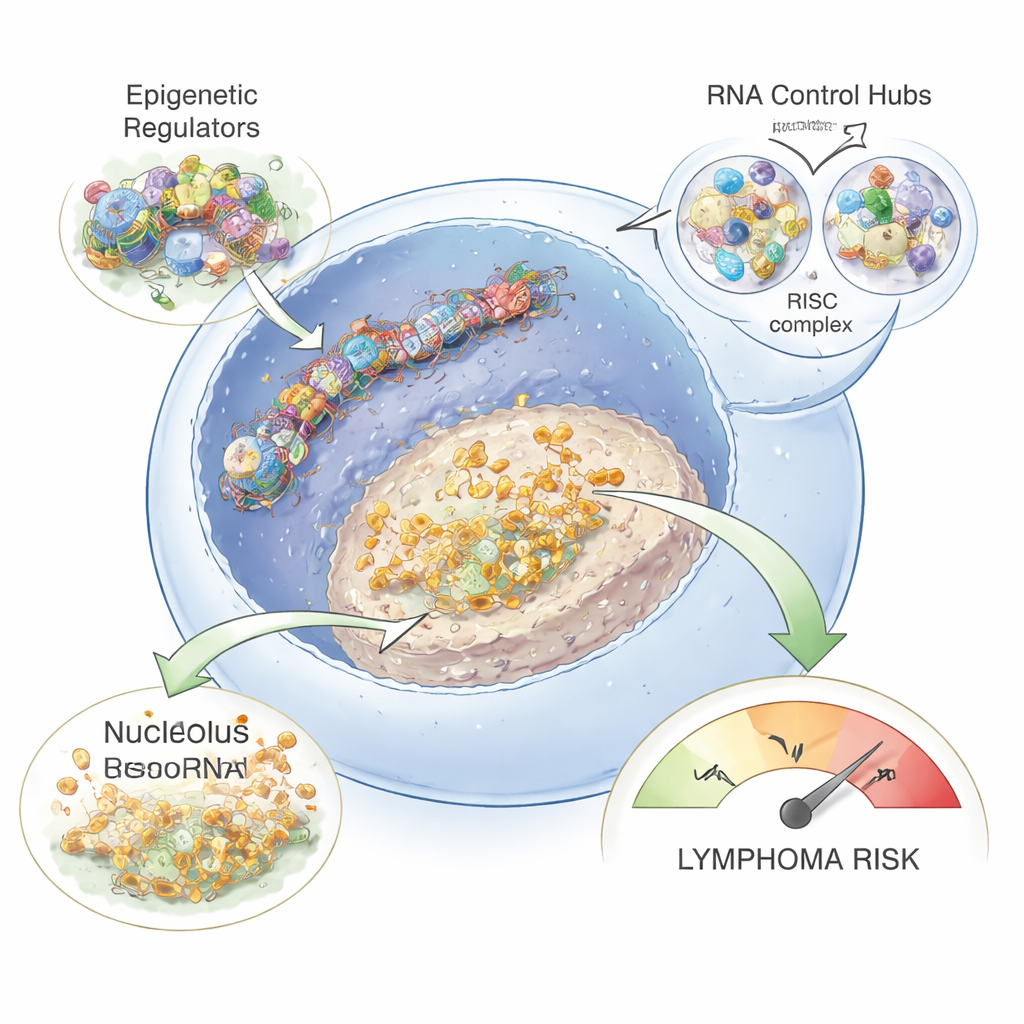
How these changes might lead to lymphoma
By correlating snoRNA levels with other genes, the study showed that upregulated snoRNAs tended to move in step with genes involved in ribosome production and ribonucleoprotein complexes. This suggests that chronic hepatitis infection may subtly rewire how B cells build ribosomes and process RNA, potentially changing which proteins are made and in what amounts. Combined with the overactive epigenetic and microRNA control systems, these shifts could create a fertile ground for mutations and misregulated growth signals that eventually tip B cells into lymphoma. Although the work does not yet prove direct infection of every B cell, the authors detected hepatitis B genetic material in at least one patient’s B cells, supporting the idea that the viruses can infect these cells and directly drive some of the observed changes.
What this means for patients and future treatments
For non-specialists, the key message is that liver viruses may influence cancer risk well beyond the liver by subtly reshaping how immune cells read their DNA and build their internal machinery. The study highlights three intertwined suspects—epigenetic regulators, RNA-silencing complexes, and snoRNAs—that appear disturbed in both infected and cancerous B cells. These pathways could eventually yield new biomarkers to flag patients at higher lymphoma risk and new drug targets that work in both virus-related and unrelated forms of the disease. For now, the findings strengthen the case for preventing and treating chronic hepatitis B and D not only to protect the liver, but also to safeguard the immune system from long-term cancer-promoting changes.
Citation: Henning, A.N., Budeebazar, M., Boldbaatar, D. et al. Hepatitis virus-associated B cell non-Hodgkin’s lymphoma involves dysregulated epigenetic and RNA-mediated regulatory gene expression and altered snoRNA transcription. Sci Rep 16, 5003 (2026). https://doi.org/10.1038/s41598-026-35041-3
Keywords: hepatitis B, hepatitis D, B cell lymphoma, epigenetic regulation, snoRNA