Clear Sky Science · en
Nanomagnetic picolylamine- based complex of palladium as an efficient heterogeneous catalyst for selective reduction of nitroarenes in water
Turning Problem Chemicals into Useful Ingredients
Many industrial chemicals that make our medicines, dyes, and plastics start life as something much less friendly: toxic, sometimes explosive compounds called nitroarenes. Chemists have long known how to transform these into safer, more useful building blocks called anilines, but doing so often requires harsh conditions, expensive materials, and creates extra waste. This study introduces a tiny, magnet-friendly catalyst that can perform this clean‑up and conversion in ordinary water at room temperature, then be pulled out with a simple magnet and reused.
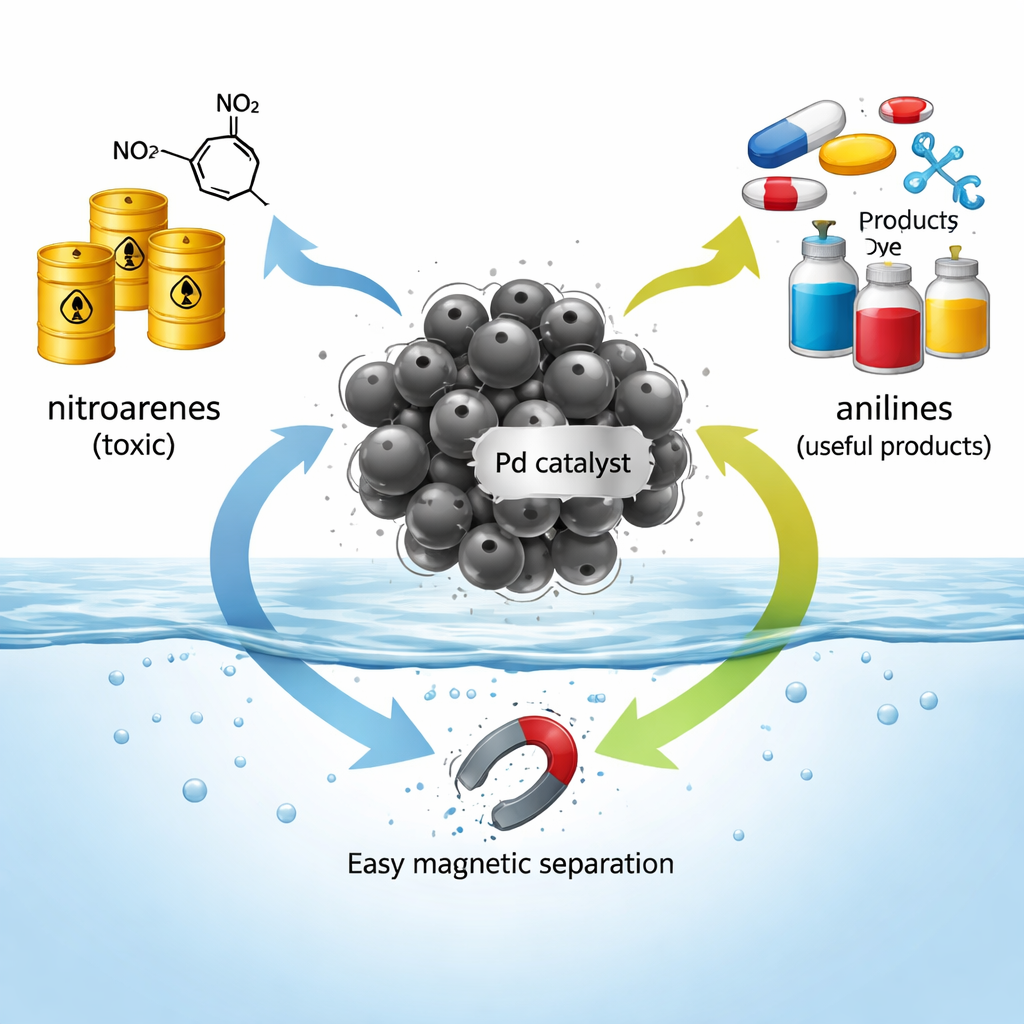
Toxic Starters and Valuable Products
Nitroarenes are aromatic rings carrying a nitro group, a chemical unit that makes them reactive but also hazardous, with links to toxicity and even cancer. At the same time, this nitro group is a gateway to many transformations that chemists rely on to build complex molecules. One of the most important steps is turning nitroarenes into anilines, which are key ingredients for polymers, colorful dyes, and many pharmaceuticals. Because anilines can be further reshaped into a wide variety of products, finding cleaner and more efficient ways to make them is important not only for chemical manufacturing but also for environmental safety.
Building a Tiny Magnetic Helper
The researchers set out to design a solid catalyst that would be highly active yet easy to recover from reaction mixtures. They started with iron oxide nanoparticles, which behave like tiny magnets. First, they coated the particle surface with a silicon‑based layer that carries a reactive chlorine group. Next, they attached a small organic molecule called 2‑picolylamine, which acts like a claw to hold metal atoms in place. Finally, they bound palladium—a metal well known for speeding up hydrogen‑based reactions—to this modified surface and chemically reduced it to its active metallic form. The end result is a nanometer‑sized iron oxide core covered with a thin shell that anchors palladium sites, creating a magnetically controllable catalyst.
Seeing and Measuring the New Material
To confirm what they had built, the team used a suite of standard materials‑science tools. Infrared spectroscopy showed the expected signatures of the iron oxide core, the silicon‑based coating, and the 2‑picolylamine layer, indicating that each construction step had succeeded. X‑ray diffraction revealed that the iron oxide crystals remained intact and that metallic palladium was indeed present on the surface, with an overall particle size on the scale of a few tens of nanometers. Electron microscopy images displayed mostly spherical nanoparticles that tended to form clusters, while elemental mapping highlighted a uniform spread of palladium across the surface. Magnetic measurements showed that, although the coating slightly reduced the magnetization compared with bare iron oxide, the particles still responded strongly and reversibly to a magnetic field, enabling rapid separation from water.
Fast, Green Reactions in Water
With the material in hand, the researchers tested it on the reduction of nitroarenes to anilines using sodium borohydride, a common source of hydrogen in the laboratory. They systematically varied the amount of catalyst, the solvent, and the amount of borohydride. Water turned out to be the best medium: it gave very high yields in short times, likely because both the catalyst surface and the reducing agent interact well in this environment. Under optimized conditions—room temperature, water as the only solvent, and very small amounts of palladium—the catalyst converted a wide range of nitroarenes, including both electron‑rich and electron‑poor examples, into their corresponding anilines in good to excellent yields. Even more complex molecules with multiple nitro groups or bulky shapes could be transformed, though they reacted more slowly.
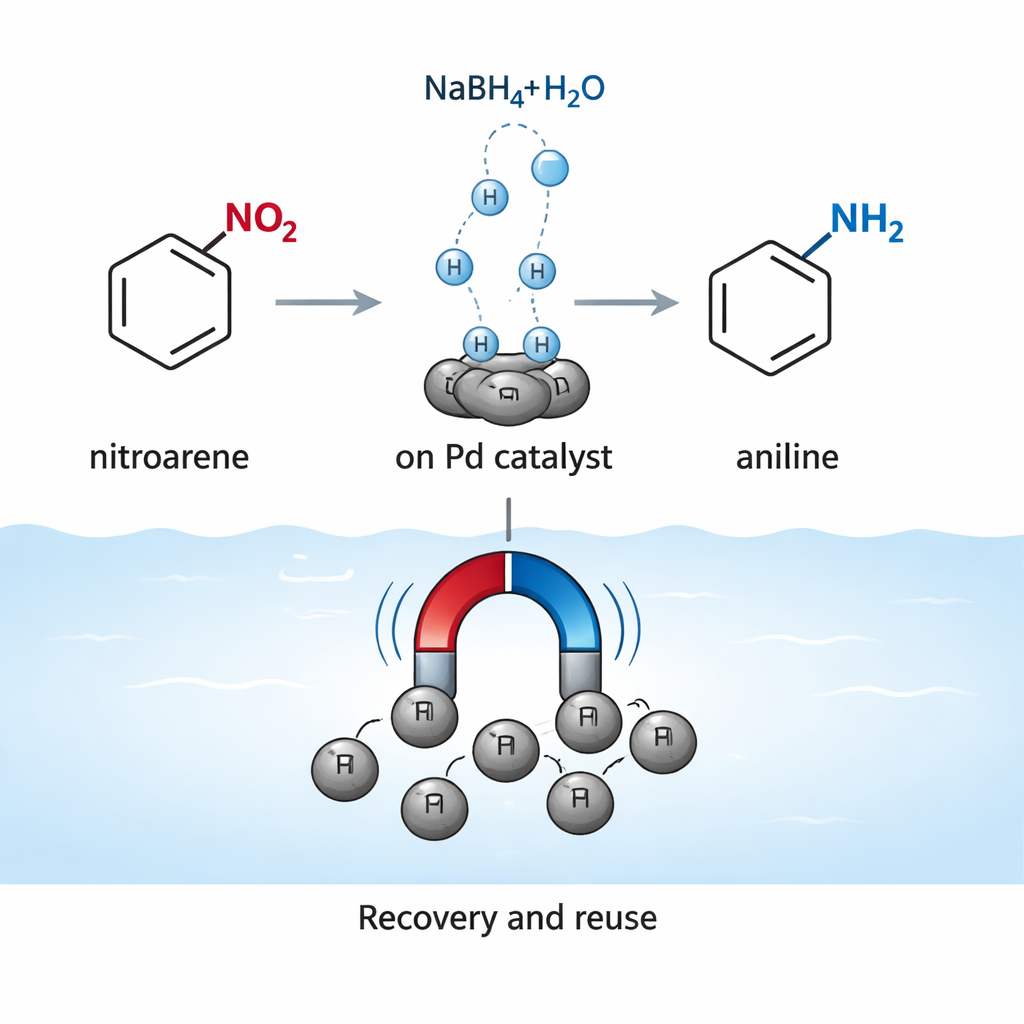
Reusable and Resistant to Wear
Modern green chemistry values not only efficiency but also reusability. The team showed that, after each reaction, the catalyst could be collected from the mixture simply by holding a magnet to the outside of the reaction vessel. After washing and drying, it was used again with almost no loss in performance over at least five cycles. Tests designed to detect dissolved palladium in the liquid phase showed only minor losses of metal, confirming that the active sites largely stay attached to the solid particles. A “hot filtration” experiment—where the solid is removed mid‑reaction—showed that the reaction almost stops once the solid is gone, another sign that the catalysis is truly happening on the particle surfaces rather than from free metal drifting in solution.
Why This Matters
For non‑specialists, the takeaway is that the study delivers a practical way to turn hazardous starting materials into useful products using a process that is simpler, safer, and more sustainable than many older methods. By combining the power of palladium chemistry with the convenience of magnetic nanoparticles, the authors created a catalyst that works quickly in plain water and can be fished out and reused multiple times. Approaches like this help move chemical manufacturing toward processes that generate less waste, use fewer toxic solvents, and are easier to manage on an industrial scale—benefits that ultimately impact the safety and cost of everyday products.
Citation: Ahmed, A.Y., AlMohamadi, H., Zabibah, H.S. et al. Nanomagnetic picolylamine- based complex of palladium as an efficient heterogeneous catalyst for selective reduction of nitroarenes in water. Sci Rep 16, 5478 (2026). https://doi.org/10.1038/s41598-026-35038-y
Keywords: magnetic nanocatalyst, palladium catalyst, green chemistry, nitroarene reduction, aniline synthesis