Clear Sky Science · en
CD13 activation assembles phosphoinositide (PI) signaling complexes to regulate the actin cytoskeleton
Cells Reaching Out to Help Their Neighbors
When tissues are injured or under stress, cells must quickly talk to one another to decide whether to repair, move, or even die. This study reveals how a little-known surface protein, called CD13, helps cells build long, thin bridges that physically connect distant cells. Through these bridges, cells can share signals such as calcium surges, potentially coordinating survival and repair across damaged tissue.
Living Cell Bridges Under Stress
The researchers worked with human cells derived from Kaposi’s sarcoma, a tumor type whose cells readily form protrusions under harsh conditions like lack of nutrients or high sugar. Under stress, normal cells made many long, elevated membrane extensions rich in actin, the structural protein that shapes cells. These protrusions rose above the surface and spanned surprisingly long distances to link cells that were not touching. When the scientists genetically removed CD13, the cells produced far fewer and shorter protrusions, which stayed close to the surface. This showed that CD13 is required for building these striking cell-to-cell bridges.
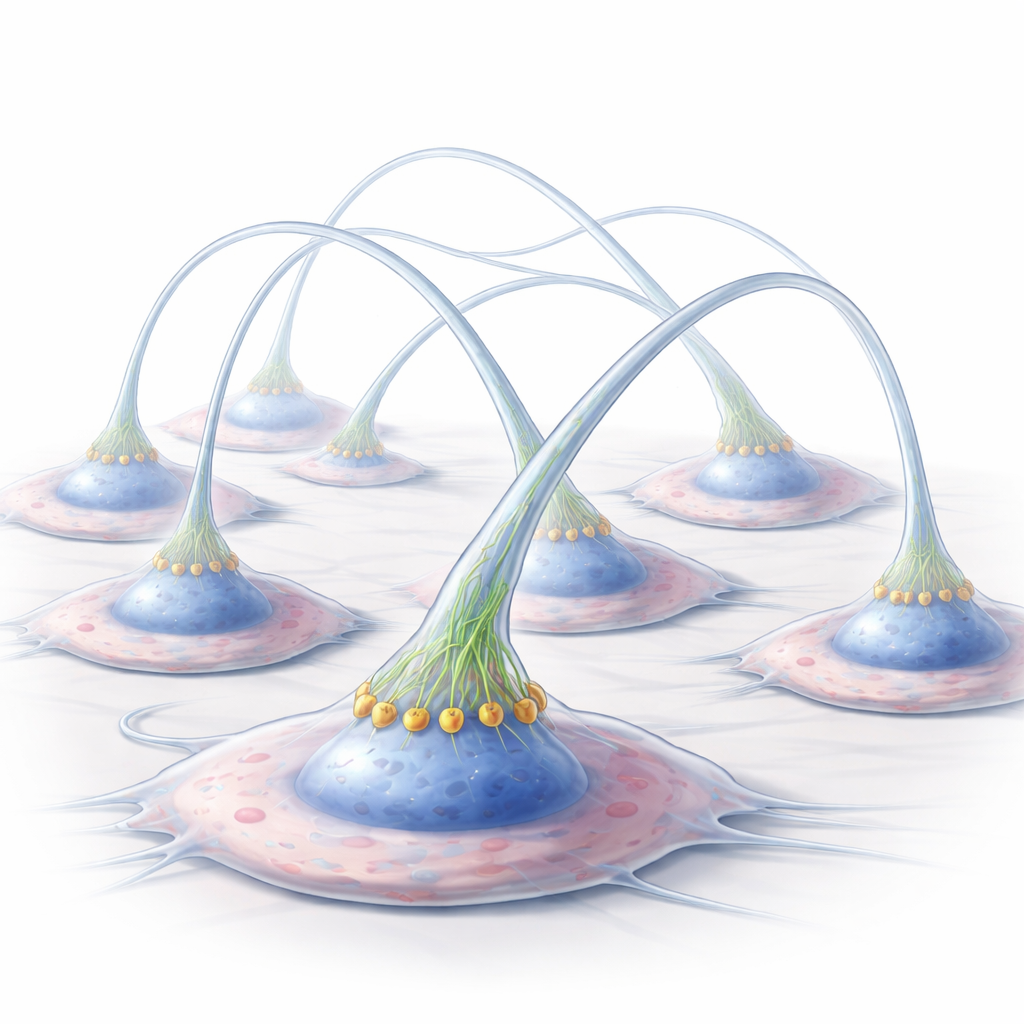
Switching On a Cellular Construction Foreman
CD13 sits in the cell’s outer membrane but has a tiny tail inside the cell. The team used a special antibody that clusters and “switches on” CD13, mimicking strong activation. This treatment greatly boosted protrusion formation in normal cells, but had no effect when CD13 was absent or when a non-activating antibody was used. Blocking CD13’s enzyme activity also did not matter, meaning its role here is as a signaling hub, not as a cutter of proteins. When scientists slipped in a short peptide designed to block interactions at CD13’s inner tail, protrusion formation dropped, even though CD13 was still present on the surface. Together, these results show that CD13 acts like a construction foreman: when activated, it assembles the right crew of proteins at the membrane to push the cell surface outward.
From Lipids and Proteins to Growing Protrusions
Inside the cell, CD13 brings together several key signaling players, including the kinases Src and FAK, the scaffold IQGAP1, and the small GTPase ARF6. These molecules regulate both the actin skeleton and the composition of the membrane. The study shows that when CD13 is activated, it helps recruit this protein complex to specific spots, where it turns on another enzyme, PIP5K. PIP5K produces a signaling lipid called PI(4,5)P2 right in the membrane. PI(4,5)P2 is a powerful organizer: it binds actin-regulating proteins and promotes actin polymerization, which physically drives membrane protrusions. The authors found that PI(4,5)P2, CD13, and actin all accumulate at the base and along the length of these structures, revealing a tightly coordinated protein–lipid engine that pushes the membrane outward.
Cellular Phone Lines for Calcium Signals
These CD13-dependent protrusions do more than just look dramatic; they behave like private phone lines between cells. Using a dye that reports calcium levels, the team mechanically stimulated one cell and watched a wave of calcium rise in its neighbors. In normal cells rich in protrusions, the calcium signal spread quickly and efficiently to many connected cells even when external calcium was blocked. In contrast, cells lacking CD13, and thus lacking robust protrusions, showed much poorer spread of the signal. The protrusions carried the gap junction protein Connexin 43, which forms channels between cells, supporting the idea that these are “closed-ended tunneling nanotubes” specialized for transmitting internal signals, not for freely exchanging bulk material.
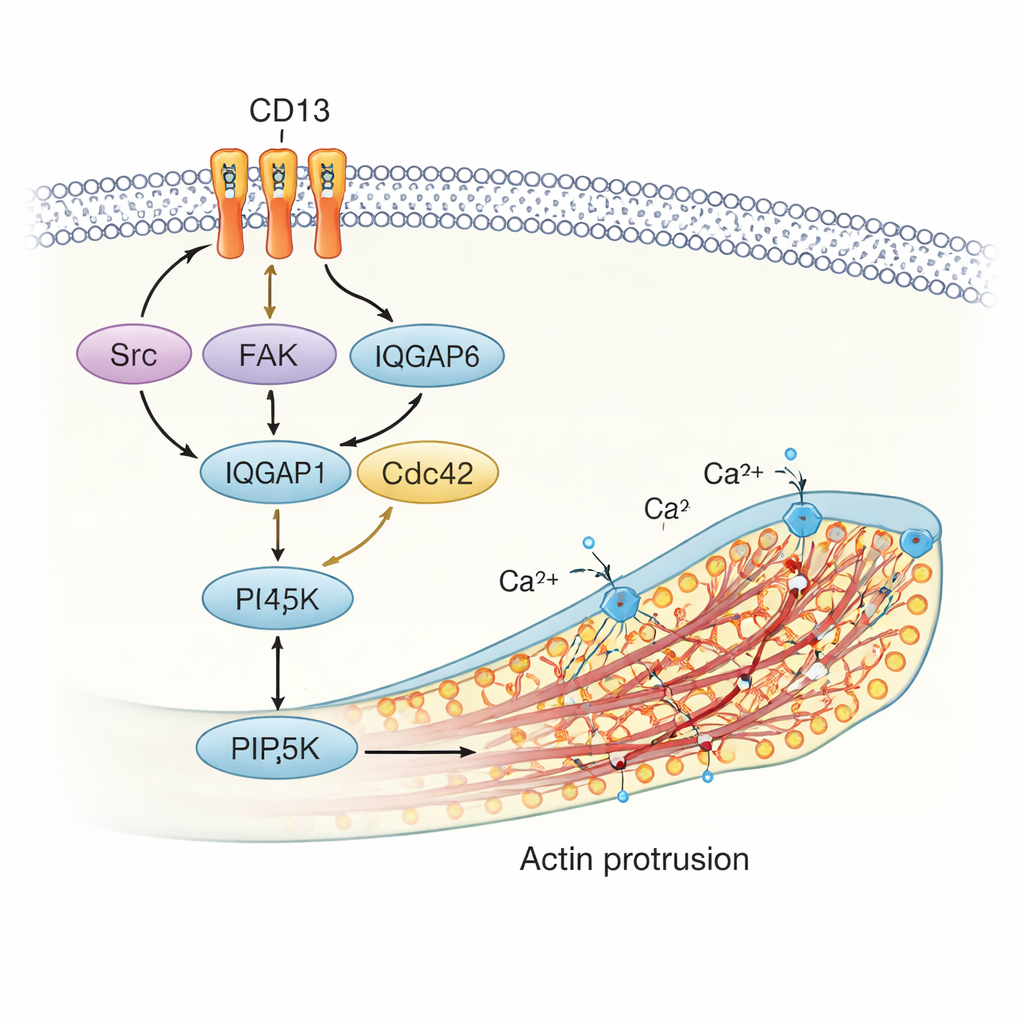
Why This Matters for Healing and Disease
By uncovering how CD13 organizes a local signaling hub to reshape the actin skeleton and membrane lipids, this work identifies CD13 as a central switch that helps cells build communication bridges under stress. For a layperson, the takeaway is that cells do not just shout chemical messages into their environment; they can lay down physical cables to send targeted signals to distant partners. Because CD13 is abundant and activated in damaged and inflamed tissues, understanding this pathway could inform new strategies to enhance tissue repair, limit harmful inflammation, or even disrupt malignant cell communication in cancer.
Citation: Meredith, E., Aguilera, B., Sharma, R. et al. CD13 activation assembles phosphoinositide (PI) signaling complexes to regulate the actin cytoskeleton. Sci Rep 16, 5191 (2026). https://doi.org/10.1038/s41598-026-35022-6
Keywords: cell communication, actin cytoskeleton, membrane protrusions, CD13 protein, phosphoinositide signaling