Clear Sky Science · en
Molecular profiling of primary versus paired asynchronous metastatic clear cell renal cell carcinoma reveals heterogeneity in tumor immune microenvironment
Why this kidney cancer study matters
Kidney cancer often spreads to other parts of the body, and modern immunotherapies do not work equally well for every patient or every tumor. This study looks under the hood of clear cell renal cell carcinoma—the most common type of kidney cancer—to see how the immune system behaves differently in the original kidney tumor compared with its later metastases. Understanding these differences could help explain why some tumors respond better to treatment than others and may guide more precise, personalized care.
Peeking inside primary and metastatic tumors
The researchers focused on 19 patients who had their kidney tumor (the primary tumor) removed and later developed metastases in places such as the lung, bone, liver, adrenal gland, lymph nodes, or soft tissue. From these patients they collected 42 tumor samples: 19 primary tumors and 23 untreated metastases. They extracted RNA—a molecule that reflects which genes are switched on—from thin slices of preserved tissue and used high-throughput sequencing to capture gene activity across thousands of genes in each sample. 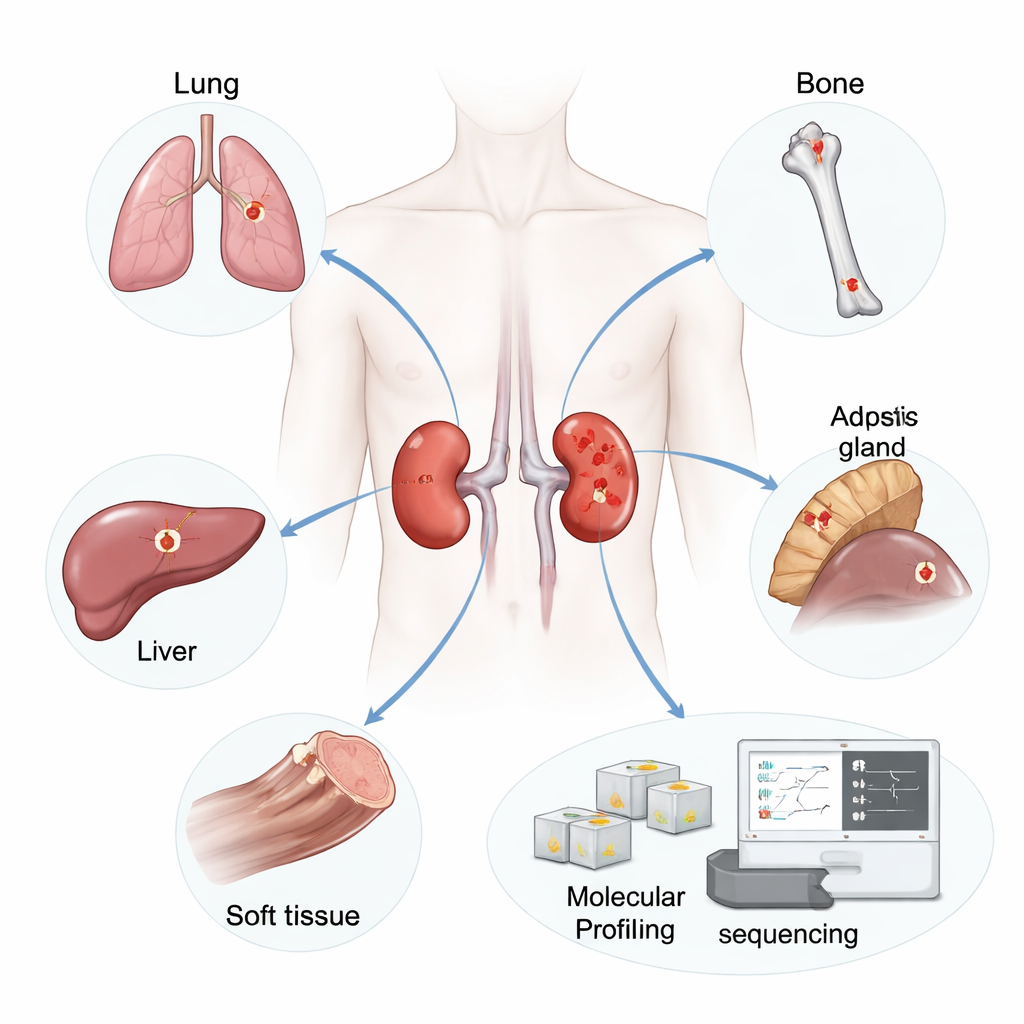
Primary and metastatic tumors are molecularly distinct
When the team analyzed patterns of gene activity, they found that primary kidney tumors tended to cluster together and looked more similar to each other than to their own metastases. In other words, a patient’s metastasis could be more different from their original tumor than from another person’s metastasis. Thousands of genes were either more active or less active in metastases compared with primaries. Metastatic tumors showed stronger signals from gene programs linked to rapid cell division and aggressive behavior, including pathways involved in cell-cycle checkpoints and protein production. In contrast, primary tumors were more enriched for stress and signaling pathways such as hypoxia (low oxygen) and inflammatory signaling, hinting at different biological pressures in the kidney compared with distant organs.
The immune landscape shifts as cancer spreads
Beyond raw gene activity, the scientists estimated which types of immune cells were present in each tumor using a computational method that deconvolves bulk RNA data. They discovered a striking difference: primary kidney tumors contained a significantly higher fraction of regulatory T cells (Tregs), a type of immune cell that dampens immune responses and can shield tumors from attack. Primaries also had more resting dendritic cells, monocytes, natural killer cells, and CD8+ T cells. In contrast, metastases were richer in so-called M2 macrophages—immune cells often associated with wound healing and a tumor-supportive, rather than tumor-killing, environment—along with more plasma cells and activated dendritic cells. 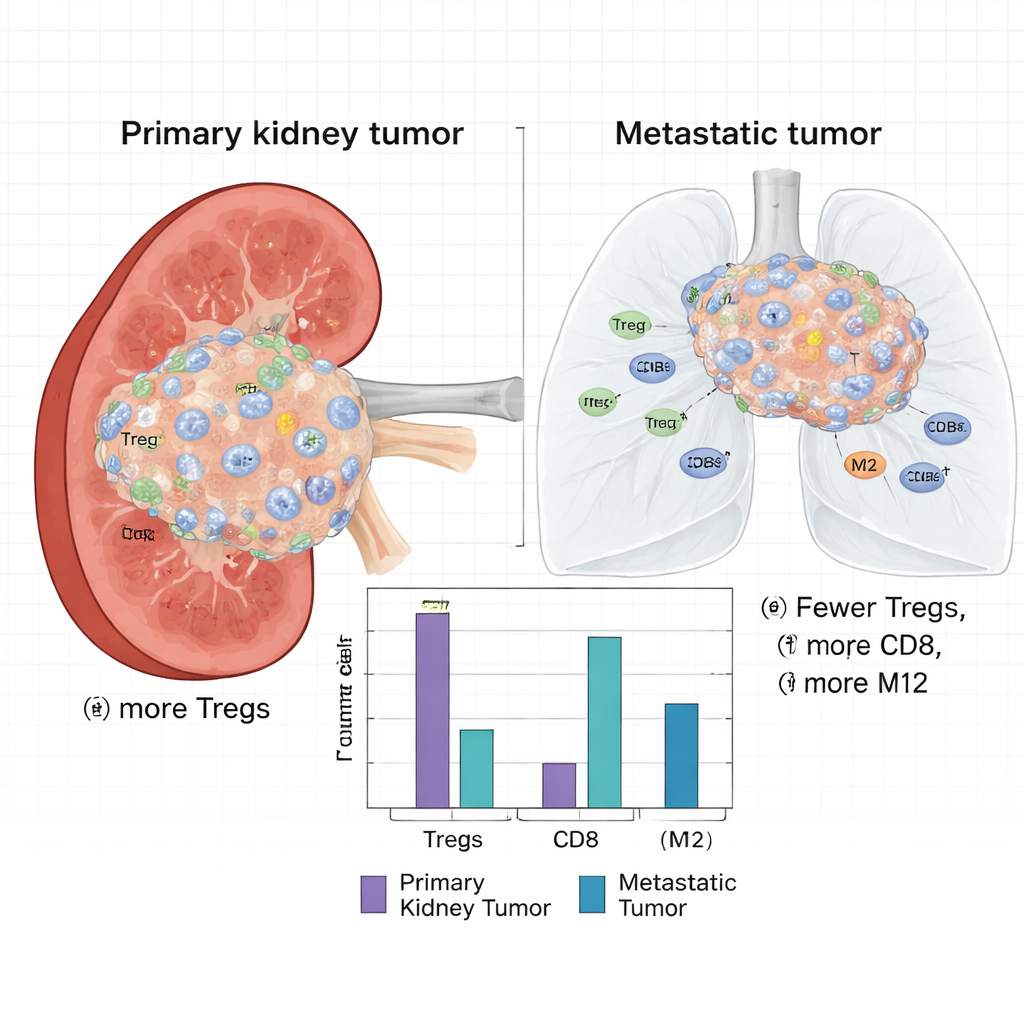
Lung and bone metastases tell different immune stories
When the researchers zoomed in on specific metastatic sites, important nuances emerged. Lung metastases showed increased activity of gene sets tied to cell division control (the G2M checkpoint) and E2F transcription factors, as well as high expression of hornerin, a protein linked to blood vessel growth in tumors. Their matched primary kidney tumors were more enriched for hypoxia-related gene programs and again carried more Tregs. Bone metastases, by contrast, stood out for stronger activation of epithelial–mesenchymal transition, a process that helps cancer cells become more mobile and invasive. These bone lesions also contained higher levels of M2 macrophages, while the primaries that gave rise to them showed increased expression of HHLA2, a recently recognized immune checkpoint molecule. Together, these findings underscore that the immune and molecular environments of metastases are not only different from primaries but also differ from one metastatic site to another.
Immune cell balance and patient outcomes
To understand what these immune patterns mean for patients, the team turned to three large public datasets of clear cell kidney cancer. They used the same computational approach to estimate Treg and CD8+ T-cell levels and then linked these to survival outcomes. Across cohorts, people whose tumors had a combination of low CD8+ T cells (the main cancer-killing T cells) and high Tregs tended to fare worse in terms of progression-free or disease-specific survival, even after accounting for age, tumor grade, and stage. While overall stage still strongly influenced survival, the balance between suppressive Tregs and attacking CD8+ cells emerged as an additional marker of risk.
What this means for future kidney cancer care
In accessible terms, this study shows that the original kidney tumor and its metastases live in different immune “neighborhoods.” The primary tumor appears especially good at recruiting peacekeeping cells that quiet the immune system, which may partly explain why kidney tumors in place often respond less well to immunotherapy than metastatic deposits. Metastases, meanwhile, evolve their own local support systems that vary by organ. These insights suggest that doctors may sometimes need to sample both the kidney tumor and metastases to fully understand a patient’s disease, and that treatments combining immune checkpoint drugs with strategies that target Tregs or M2 macrophages might improve outcomes for people with metastatic clear cell renal cell carcinoma.
Citation: Cotta, B., Nallandhighal, S., Monda, S. et al. Molecular profiling of primary versus paired asynchronous metastatic clear cell renal cell carcinoma reveals heterogeneity in tumor immune microenvironment. Sci Rep 16, 5473 (2026). https://doi.org/10.1038/s41598-026-35021-7
Keywords: kidney cancer, tumor microenvironment, immunotherapy, metastasis, regulatory T cells