Clear Sky Science · en
Mechanistic insights into melanin-induced PCR inhibition and its NanoPCR-based mitigation
Why dark pigments can hide vital DNA clues
When investigators or doctors rely on DNA tests, they assume the genetic material in a sample can be copied cleanly in the lab. But dark pigments like melanin, which give hair, skin, and some tissues their color, can quietly sabotage this copying step, known as PCR. This study unpacks how melanin interferes with DNA testing and explores a nanotechnology-based workaround that could make DNA profiling more reliable in tough, pigment-rich samples from crime scenes and beyond.
How DNA copying powers modern forensics
PCR, the polymerase chain reaction, is the workhorse behind modern DNA testing. It uses a heat-stable enzyme, Taq polymerase, to make millions of copies of tiny stretches of DNA so they can be read and compared. In forensic work, this is crucial for building STR (short tandem repeat) profiles that help identify individuals from traces like hair shafts, skin fragments, or burned and decomposed remains. However, real-world samples rarely arrive pure. They often carry along chemical “troublemakers” that can block PCR and rob analysts of clear, court-ready DNA profiles.
Melanin: the pigment that gets in the way
Melanin, the same pigment that shields our skin and hair from sunlight, turns out to be one of the most stubborn PCR blockers in forensic samples. Even though it may be only a few percent of a hair’s mass, its complex, sticky structure lets it latch onto proteins and metal ions, and it can interfere with how DNA and the polymerase enzyme meet. Earlier work showed that melanin reduces PCR efficiency and causes partial or failed DNA profiles, but the precise way it disrupts the copying process was unclear. Forensic samples rich in melanin—like dark hair, pigmented tissue, or cremated remains—often yield missing DNA signals, reduced peak heights, and imbalanced alleles that lower the evidentiary value of a profile.
Zooming in on the clash between melanin and the PCR enzyme
The authors used computer modeling and laboratory tests to watch, in effect, how melanin and Taq polymerase interact at the molecular level. Detailed simulations of the enzyme’s structure revealed that melanin nestles into key regions that normally hold DNA in place and help add new building blocks during copying. In particular, melanin forms stable, non-covalent contacts with specific amino acids that line the catalytic core and DNA binding groove, subtly destabilizing the enzyme’s shape. A fluorescence assay that tracks changes around tryptophan residues confirmed that melanin binds the enzyme with moderate strength, in a reversible fashion. Together, these data support the idea that melanin acts as a mixed-mode, competitive inhibitor—it occupies space and contacts that Taq polymerase needs, slowing or distorting the copying reaction without permanently destroying the enzyme.
What this looks like in real DNA profiles
To see the practical impact, the team ran STR genotyping on DNA exposed to melanin. The results resembled a damaged barcode: some highly informative markers, such as SE33 and Penta E, vanished altogether; others showed weak signals and skewed peak heights. Overall signal intensity dropped, and the pattern varied across dye channels, consistent with uneven interference. This kind of selective fading and dropout is especially troubling in casework, where losing just a few powerful markers can blur identity resolution or complicate mixed-sample interpretation. Interestingly, a few loci sometimes showed unexpectedly high signals, which the authors attribute to the random, stop-and-go nature of PCR under stress rather than any true improvement—another reminder that inhibited reactions can mislead if judged by isolated peaks alone.
Nanoparticles and a familiar protein come to the rescue
Because directly removing melanin can also remove precious DNA, the authors explored “in-tube” helpers that neutralize the inhibitor instead. They compared three additives: plain gold nanoparticles, the common protein bovine serum albumin (BSA), and gold nanoparticles coated with BSA. Plain gold particles offered only partial recovery of signal. Free BSA, a longtime PCR helper, delivered the strongest overall restoration of peak heights and allelic balance, but only at relatively high and finicky concentrations, and with more variability between samples. The hybrid approach—BSA-coated gold nanoparticles—struck a balance: it significantly improved overall signal and marker recovery, nearly matching BSA’s performance, while using orders-of-magnitude less protein and producing more uniform, reproducible profiles. The nanoparticles serve as a stable scaffold that presents BSA in a way that efficiently soaks up melanin and protects the polymerase throughout thermal cycling.
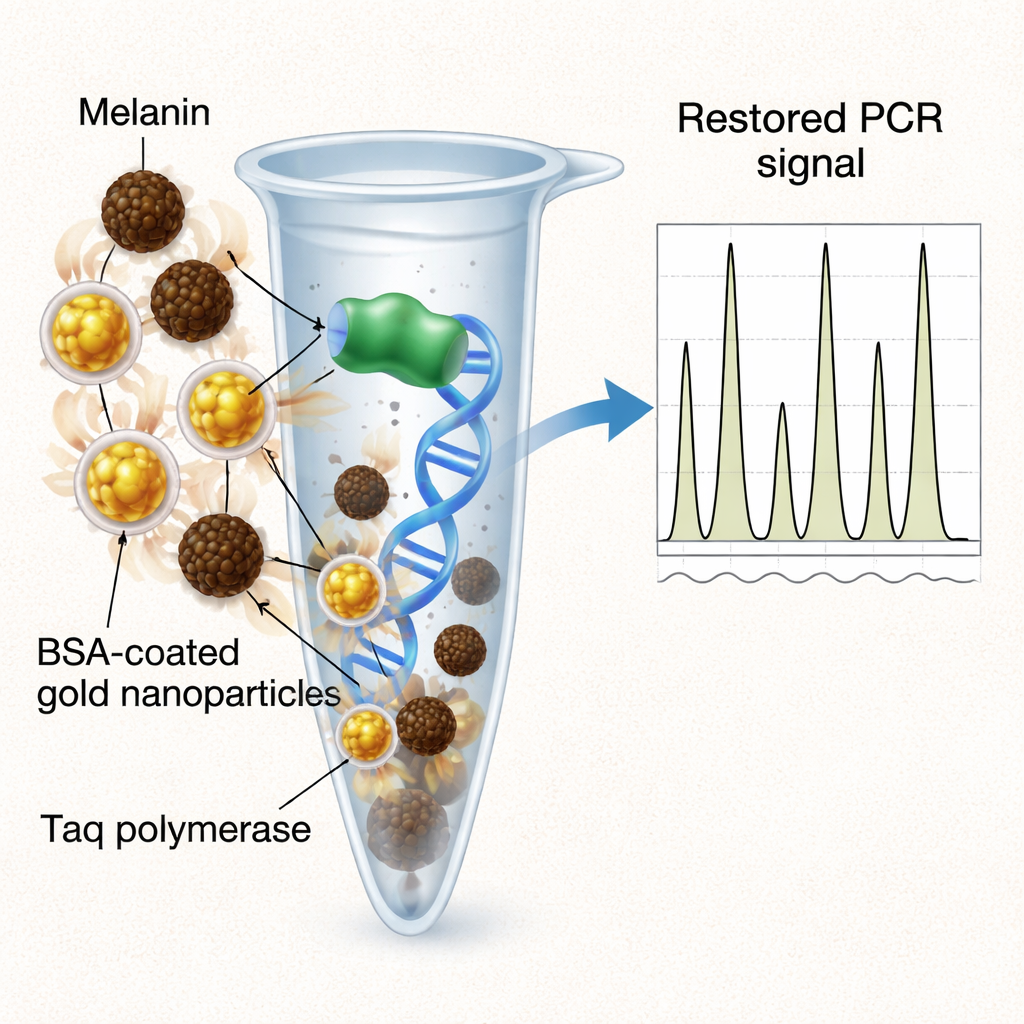
What this means for real-world DNA testing
For non-specialists, the take-home message is that dark pigment in evidence samples can quietly derail DNA tests by gumming up the very enzyme that makes testing possible. This study not only clarifies how that interference happens at the atomic level but also shows that carefully designed nanomaterials—gold particles dressed with a thin layer of a familiar protein—can rescue DNA signals without losing material or introducing artifacts. While further validation on real casework samples is still needed, the work points toward more robust, low-dose additives that could help forensic labs, medical diagnostics, and even ancient DNA studies reliably read genetic information from difficult, pigment-rich material.
Citation: Vajpayee, K., Srivastava, S., Sharma, S. et al. Mechanistic insights into melanin-induced PCR inhibition and its NanoPCR-based mitigation. Sci Rep 16, 5467 (2026). https://doi.org/10.1038/s41598-026-35010-w
Keywords: forensic DNA, PCR inhibition, melanin, gold nanoparticles, nanotechnology in genetics