Clear Sky Science · en
Structural basis for diosgenin as an inverse agonist of retinoic acid receptor-related orphan receptor γ
Why a plant compound and an immune switch matter
Many chronic conditions—from autoimmune diseases like multiple sclerosis and psoriasis to obesity and type 2 diabetes—are driven by overactive immune signals and misfired metabolic cues. Scientists have long searched for safer drugs that can dial these signals up or down with precision. This study explores how diosgenin, a natural molecule found in certain medicinal plants and yams, latches onto an immune-regulating protein called RORγ and flips it into a less active state. Understanding this interaction at an atomic level could open the door to gentler, more targeted therapies built from a familiar plant-derived scaffold.
A master dial for immunity and metabolism
RORγ is part of a large family of "nuclear receptors"—proteins inside cells that turn genes on or off in response to small molecules. This particular protein is a key controller of a group of immune cells known as Th17 cells, which produce the inflammatory signal IL-17A and have been linked to autoimmune diseases such as multiple sclerosis, psoriasis, rheumatoid arthritis and Crohn’s disease. RORγ also influences how fat cells develop and how the body responds to insulin, tying it to obesity and diabetes. Because of this broad reach, RORγ has become an attractive drug target, but many synthetic molecules that bind it have run into problems with safety, selectivity, or effectiveness in humans.
A traditional remedy yields a promising lead
Diosgenin is a steroid-like molecule extracted from plants including species of Dioscorea (wild yam) and several traditional medicinal herbs. It has been studied for anti-inflammatory, anti-cancer, anti-diabetic and cardiovascular benefits in cell and animal models. Earlier work suggested diosgenin could influence the RORα/γ pathway and suppress harmful Th17 responses, but it was not clear whether it touched RORγ directly, or how it might shape the protein’s behavior. To answer that, the authors screened a library of natural compounds using a sensitive binding assay and identified diosgenin as a strong binder to the ligand-binding region of RORγ.
How diosgenin pushes RORγ into reverse
The researchers examined how diosgenin affects RORγ’s ability to recruit helper proteins that either boost (coactivators) or dampen (corepressors) gene activity. Surprisingly, diosgenin encouraged RORγ to bind both types of partners, a pattern different from that of a known natural antagonist called ursolic acid. In cell-based reporter tests that track gene switching, diosgenin consistently lowered RORγ-driven activity in a dose-dependent way, with potency in the sub-micromolar range. This behavior fits the profile of an "inverse agonist": a compound that not only blocks baseline activity but actively drives the receptor toward an off state, even when it would otherwise be turned on.
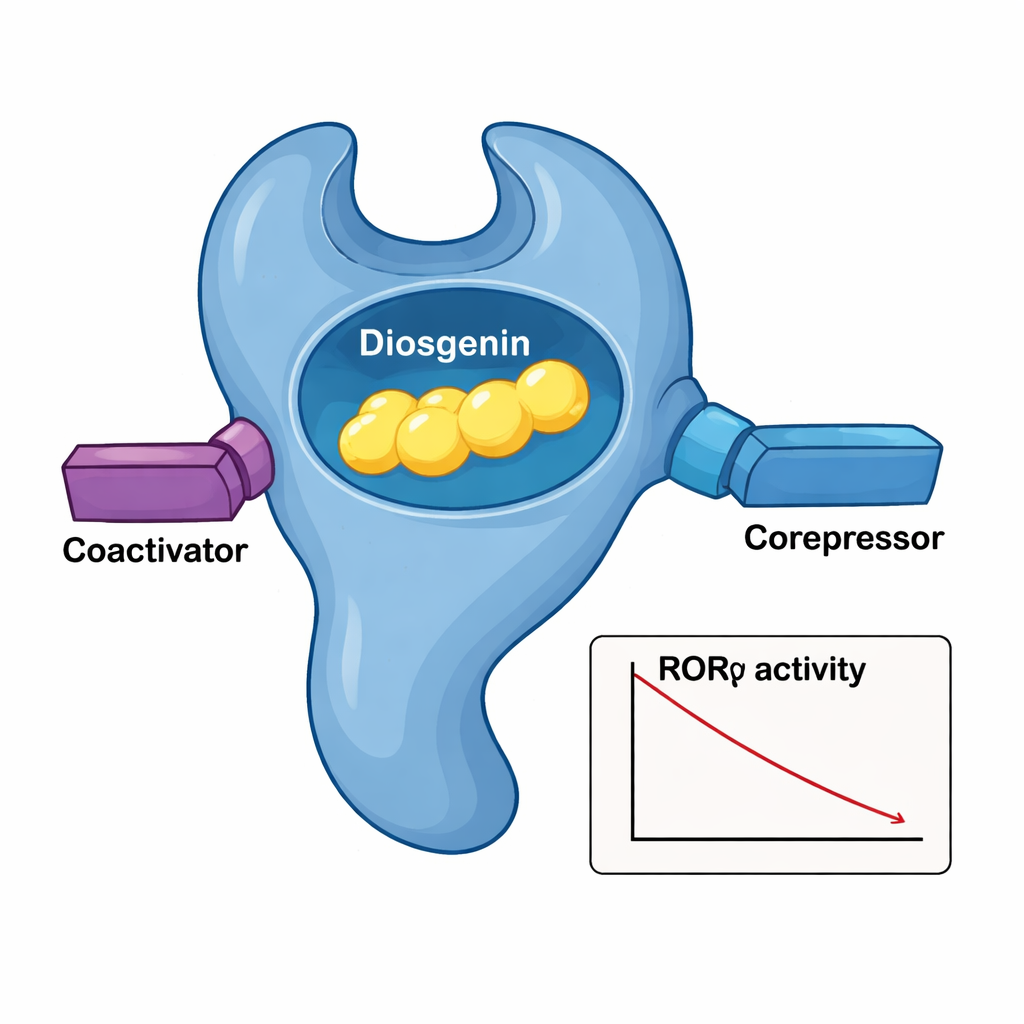
A close-up of the molecular handshake
To see exactly how diosgenin fits into RORγ, the team solved the three-dimensional crystal structure of the protein’s ligand-binding domain with diosgenin lodged inside. The receptor folds into a layered bundle of helices, forming a pocket that cradles the steroid-like body of diosgenin. A network of snug hydrophobic contacts from several amino acids acts like a tailored glove around the molecule’s backbone, while a critical hydroxyl group on diosgenin forms water-mediated hydrogen bonds with two arginine residues. When those pocket residues were altered one by one, diosgenin’s ability to silence RORγ dropped, confirming their importance. Structural comparisons with the RORγ–ursolic acid complex showed that diosgenin keeps a key tail helix (AF‑2) in an “active-like” position even as it still drives overall transcription downward, explaining why it can simultaneously draw in both coactivators and corepressors.
From crystal structure to future medicines
The detailed picture of the diosgenin–RORγ interaction highlights two notable advantages for drug design. First, the receptor’s pocket shows enough flexibility to recognize different ligand shapes, meaning chemists can fine-tune diosgenin’s scaffold to enhance selectivity for RORγ and reduce unwanted binding to other nuclear receptors. Second, diosgenin’s natural origin and known low inherent toxicity in preclinical models make it an appealing starting point compared with some fully synthetic compounds that have stalled in trials. In simple terms, this work reveals how a plant-derived molecule can latch onto a central immune and metabolic switch and push it into reverse, offering a structural template for developing safer, more effective treatments for RORγ-related diseases.
Citation: Chen, S., Tian, S., Liang, J. et al. Structural basis for diosgenin as an inverse agonist of retinoic acid receptor-related orphan receptor γ. Sci Rep 16, 4765 (2026). https://doi.org/10.1038/s41598-026-35006-6
Keywords: ROR gamma, diosgenin, autoimmune disease, nuclear receptor, inverse agonist