Clear Sky Science · en
Uncovering the role of integrated stress in Alzheimer’s disease through single-cell and transcriptomic analysis
Why Stress Inside Brain Cells Matters
Alzheimer’s disease is best known for memory loss, but deep inside brain cells another, less visible change is taking place: a cellular “stress response” that tries to keep cells alive when they are under pressure. This study looks at that internal alarm system in unprecedented detail, zooming in on individual brain cells and their genes. By doing so, the researchers aim to uncover new warning signals and treatment targets for Alzheimer’s long before symptoms become severe.
Looking at the Brain One Cell at a Time
Instead of averaging signals across whole pieces of brain tissue, the team used single-cell RNA sequencing, a technique that reads out which genes are switched on in thousands of individual cells. They analyzed data from two large public datasets: one with nearly 180,000 single cells from brains of people with and without Alzheimer’s, and another with gene activity measured in bulk brain samples from more than 250 people. Advanced software grouped the cells into nine major types, including nerve cells, immune cells known as microglia, and endothelial cells, which form the inner lining of blood vessels in the brain. This fine-grained view allowed the scientists to ask a crucial question: in which cell types is the internal stress alarm most strongly activated in Alzheimer’s?
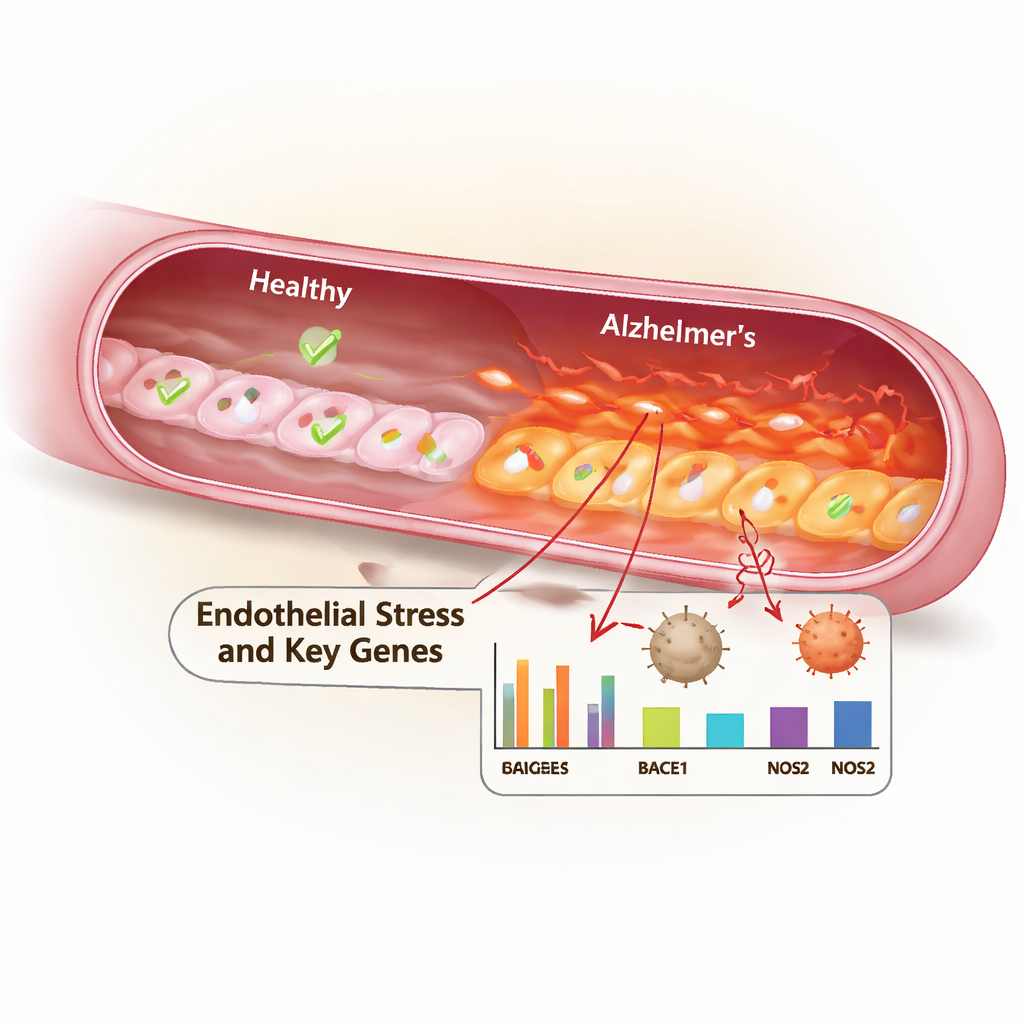
Blood Vessel Cells Under Heavy Pressure
The researchers focused on the “integrated stress response,” a built-in program cells use to cope with threats such as misfolded proteins and lack of nutrients. Using a scoring method that tallied the activity of 129 stress-related genes, they discovered that endothelial cells in Alzheimer’s brains showed a markedly higher stress response than those in healthy brains. Follow-up analyses revealed 202 genes that were turned up or down in these endothelial cells in Alzheimer’s. When they modeled how different cell types talk to each other through signaling molecules, stressed endothelial cells emerged as highly connected hubs, sending and receiving many stress and immune signals across the brain’s cellular network.
Six Genes That Stand Out
To narrow the search to the most informative genes, the team applied two machine-learning approaches—LASSO regression and random forests—to the list of altered genes. The overlap between these methods yielded six key genes: BTG1, EPB41L4A, HERPUD1, SLC3A2, SLC7A11, and SLC7A5. These genes are involved in controlling cell growth, handling misfolded proteins, and transporting amino acids that help maintain antioxidant defenses. When the scientists examined which biological pathways these genes participate in, they repeatedly intersected with inflammation-related routes, especially the NF-κB and TNF signaling pathways, which are known drivers of chronic inflammation and have been linked to Alzheimer’s pathology.
Links to Immunity and Real Patients
The study did not stop at computer analysis. The researchers examined immune cell patterns and found that several immune cell types, such as M2 macrophages, neutrophils, and certain memory T cells, were more abundant in Alzheimer’s, while other protective cells were reduced. The six key genes were closely tied to these shifts in immune balance, suggesting that stressed endothelial cells may help reshape the brain’s immune environment. To test whether the same genes change in living people, the team recruited ten Alzheimer’s patients and ten matched healthy volunteers. In blood cells from these participants, one gene (HERPUD1) was elevated and the other five were reduced in patients, mirroring the patterns seen in brain data. The authors used these genes to build a prediction model that could distinguish Alzheimer’s from controls with promising accuracy.
What This Means for Future Care
For readers outside the lab, the message is that Alzheimer’s is not only about plaques and tangles but also about how different brain cells, especially blood vessel cells, respond to long-term stress and communicate with the immune system. By pinpointing six genes that sit at the crossroads of cellular stress and inflammation, this work offers potential new biomarkers for earlier diagnosis and new targets for drugs that could calm harmful stress signals in the brain. While larger clinical studies are still needed, these findings open a window onto hidden processes that may help explain why brain cells fail in Alzheimer’s and how we might slow or prevent that decline.
Citation: Sheng, N., Wang, HY., Song, K. et al. Uncovering the role of integrated stress in Alzheimer’s disease through single-cell and transcriptomic analysis. Sci Rep 16, 4779 (2026). https://doi.org/10.1038/s41598-026-34997-6
Keywords: Alzheimer’s disease, integrated stress response, endothelial cells, neuroinflammation, single-cell sequencing