Clear Sky Science · en
Combination of bacteriophage and antibiofilm compounds from phyllosphere bacteria as a comprehensive strategy for aquaculture and food pathogen control
Why slimy bacteria layers matter to your food and fish
Whether it is fish from aquaculture ponds or ready-to-eat products in a supermarket, both can be threatened by invisible bacterial communities that cling to surfaces as tough, slimy layers called biofilms. These biofilms help harmful bacteria survive cleaning, preservatives, and antibiotics, contributing to food poisoning and disease outbreaks in fish farms. This study explores a nature-inspired way to tackle those stubborn layers by combining viruses that attack bacteria with natural compounds made by harmless bacteria living on plant leaves.
Hidden shields on factory lines and in fish ponds
Biofilm-forming bacteria such as Bacillus cereus, a cause of food poisoning, and Vibrio harveyi, a serious fish pathogen, are especially hard to control because their slimy coating protects them from harsh conditions and common treatments. This shield, called an extracellular matrix, is made of sticky sugars, proteins, and DNA that glue the cells together and to surfaces in pipes, tanks, and processing equipment. As aquaculture and seafood consumption rise worldwide, these resilient films pose growing risks for public health and economic losses. Current methods often rely on antibiotics or chemical preservatives, which may be ineffective against biofilms and raise concerns about resistance and residues.
Borrowing help from leaf-dwelling microbes and their viral enemies
To find new tools, the researchers turned to phyllosphere bacteria—microbes that naturally live on plant leaf surfaces and compete with other bacteria in that harsh, exposed environment. Two such strains, Pseudomonas fluorescens JB 3B and Proteus myxofaciens JB 20B, produce mixtures of small molecules in the liquid around them, known as supernatants. The team tested these supernatants alongside two highly concentrated bacteriophages, viruses that infect and burst specific bacteria: one targeting B. cereus and another targeting V. harveyi. Instead of simply checking whether these treatments killed free-floating bacteria, the researchers focused on how well they could prevent biofilms from forming and how effectively they could break apart mature biofilms once established.
Breaking down biofilm walls in different ways
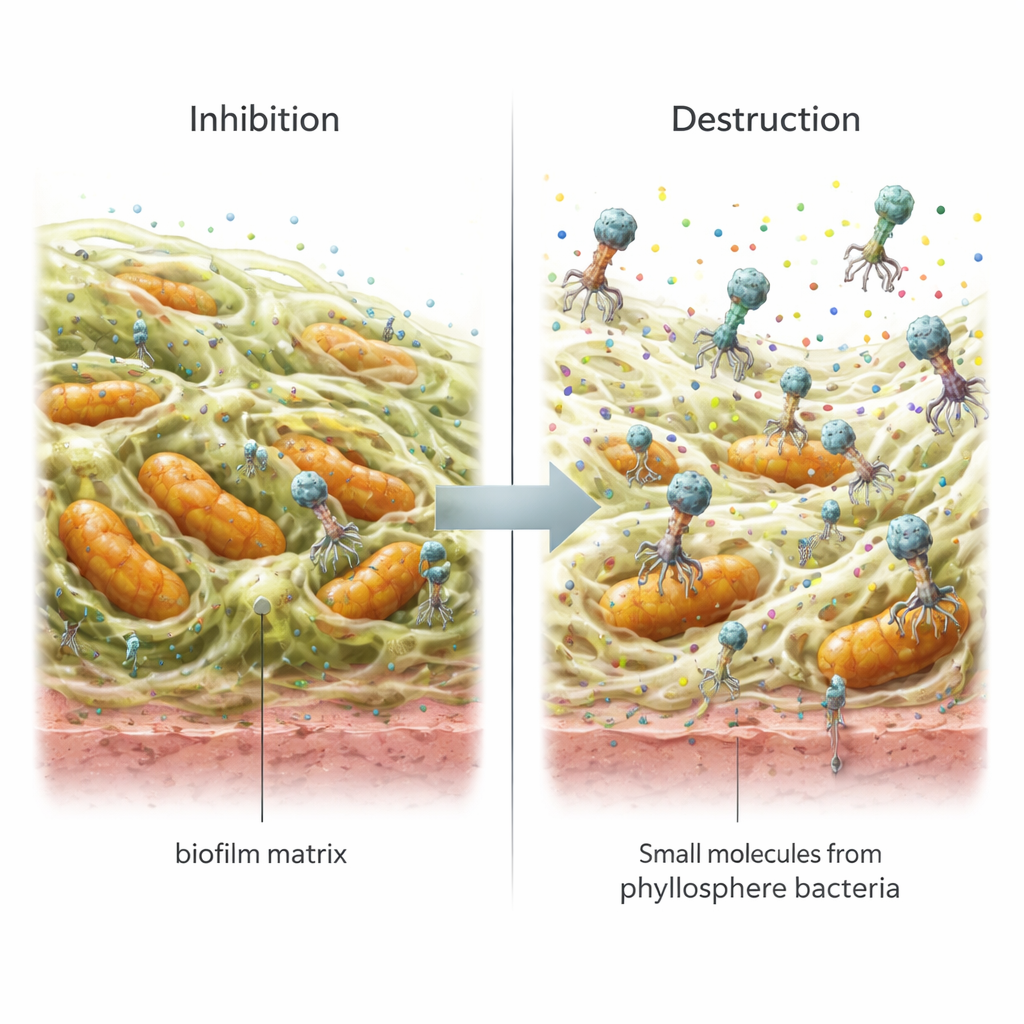
The leaf-bacteria supernatants did not behave like classic antibiotics: they did not create clear kill zones on test plates and did not block bacterial “quorum sensing,” the chemical signaling often used to coordinate biofilm building. Yet, when the scientists grew biofilms in small test wells, these supernatants significantly reduced both the formation of new films and the destruction of existing ones for both target species. For B. cereus, the supernatant from strain JB 3B alone cut biofilm buildup by about 41% and broke down mature films by about 55%. The phages alone also showed strong activity. When combined with the supernatants, the effects on B. cereus were similar or slightly better, suggesting the two tools could sometimes work together. In contrast, for V. harveyi, the best results often came from single treatments—either the phage or the supernatant—while combining them actually reduced performance, showing that one universal recipe does not fit all species.
What the microscope and chemistry reveal
Light microscopy and scanning electron microscopy offered a visual before-and-after snapshot of what these treatments did to the biofilms. Untreated samples showed thick, compact layers of cells embedded in a dense matrix. Treated samples, whether by supernatant, phage, or both, displayed thinner, patchier films with clear gaps and a disrupted structure, matching the drops measured in biofilm mass. Chemical analysis of the leaf-bacteria supernatants using gas chromatography–mass spectrometry identified several small molecules—acetic acid, sarcosine, 4‑octadecenal, and in one strain erythritol—that are known or suspected to weaken bacterial surfaces, interfere with attachment, or damage cell components. These findings suggest that instead of poisoning bacteria outright, the mixtures loosen the glue and undercut the stability of the biofilm matrix, making it easier for phages and other stresses to act.
What this could mean for safer food and healthier fish
To a non-specialist, the main message is that nature offers promising tools to manage stubborn bacterial films without relying solely on traditional antibiotics or harsh chemicals. Leaf-dwelling bacteria produce mild compounds that can weaken biofilms, and bacteriophages can then reach and kill protected cells. However, the study also shows that mixing these tools does not always lead to a stronger effect; success depends on the particular bacterial species and the exact cocktail of compounds involved. In practice, this means that future biofilm-control strategies in food processing and aquaculture may use tailored combinations of friendly microbes, their natural products, and phages, carefully matched to the target pathogen. With further work on safety and effectiveness, such approaches could help reduce foodborne illness and fish losses while easing pressure on conventional antibiotics.
Citation: May, J., Waturangi, D.E., Tan, W.A. et al. Combination of bacteriophage and antibiofilm compounds from phyllosphere bacteria as a comprehensive strategy for aquaculture and food pathogen control. Sci Rep 16, 4757 (2026). https://doi.org/10.1038/s41598-026-34989-6
Keywords: biofilms, bacteriophages, aquaculture, food safety, phyllosphere bacteria