Clear Sky Science · en
Evaluation of UV/TiO2/H2O2 photocatalysis for the removal of perfluorinated organic compounds from water
Why stubborn “forever chemicals” in water matter
Invisible pollutants known as “forever chemicals” have been detected in tap water, rivers, and even food around the world. One of the best known, perfluorooctanoic acid (PFOA), is used in nonstick pans, stain‑resistant fabrics, and firefighting foams. It does not easily break down in nature and has been linked to cancer, liver damage, and problems in babies and children. This study explores whether a light‑driven water treatment method can start to chip away at PFOA’s legendary resistance and what real‑world obstacles stand in the way.
A tough chemical that lingers everywhere
PFOA belongs to a family of industrial compounds often called PFAS, nicknamed “forever chemicals” because they can persist in water for decades. Their carbon–fluorine backbone makes them extremely stable and hard to destroy. As a result, tiny amounts are now found in drinking water, surface water, soil, air, and wildlife. Health studies link PFOA to liver and immune problems, developmental effects in newborns, and possible cancer. Regulators are starting to respond: in 2024 the U.S. Environmental Protection Agency set a very low legal limit for PFOA in drinking water. Yet common treatments in water plants—such as filtration and settling—mostly pass PFOA straight through.
Shining light on a promising treatment idea
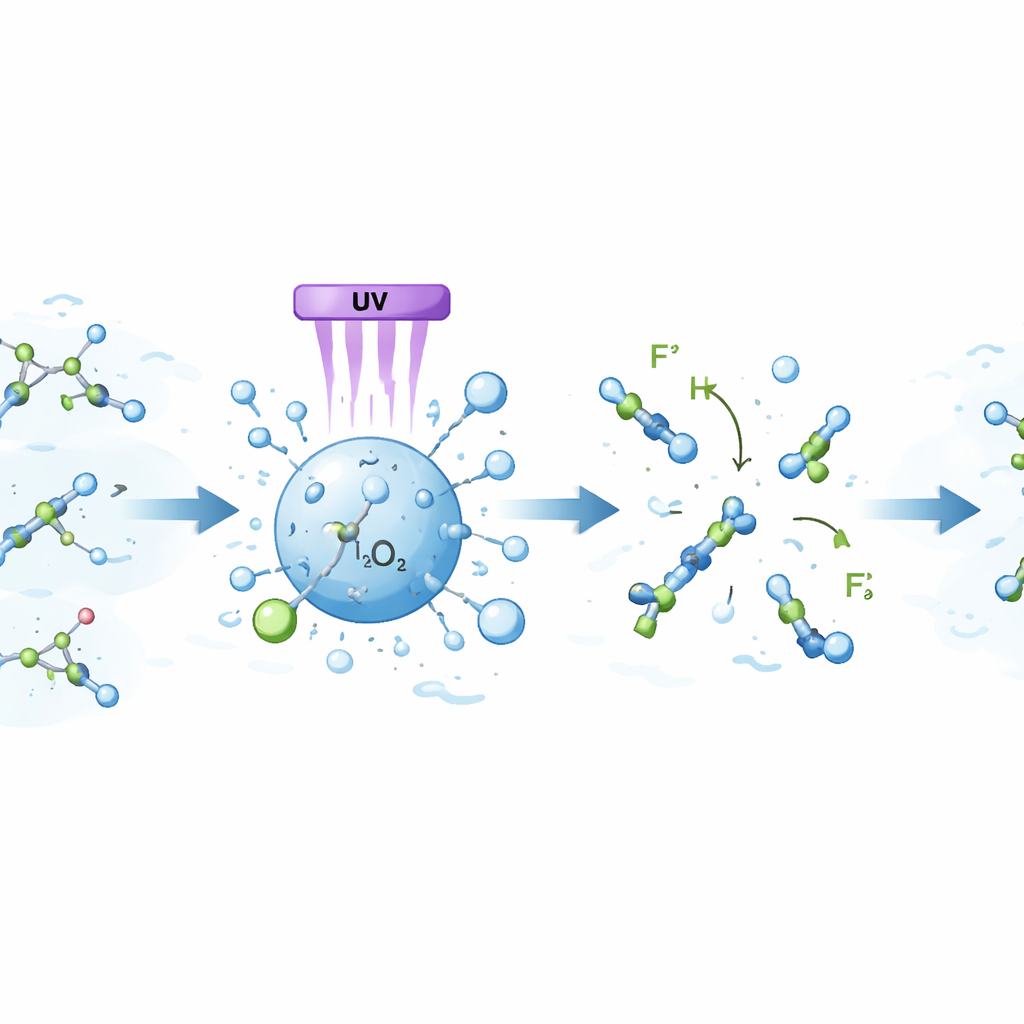
Researchers have been testing “advanced oxidation processes,” which aim not to trap pollutants but to chemically break them apart using highly reactive short‑lived species in water. In this work, the authors evaluated a combination of ultraviolet (UV) light, titanium dioxide (TiO₂) particles, and hydrogen peroxide (H₂O₂). When UV light hits TiO₂ in water, it can create energetic charges at the particle surface that, together with H₂O₂, form aggressive oxidants able to attack sturdy molecules. The team built a one‑liter glass reactor with internal UV lamps and carefully controlled stirring, temperature, and chemical doses to see how much PFOA they could remove from both ultra‑clean laboratory water and real river water.
How well the light‑driven system worked
The scientists first tuned the recipe, varying the amounts of TiO₂ and H₂O₂ and comparing two types of UV light: shorter‑wavelength UV‑C at 254 nanometers and longer‑wavelength UV‑A at 360 nanometers. They found that stronger performance came from the higher‑energy 254‑nanometer light and from moderate, not extreme, doses of both TiO₂ and H₂O₂. Under these optimized conditions, the system removed about 26% of PFOA from deionized water after five hours, and 40% after a full day. Tests without one of the three ingredients showed that neither UV light alone, nor TiO₂ alone, nor hydrogen peroxide in the dark could significantly degrade PFOA. Only when all three components were present did removal clearly improve.
Why real water makes the job harder
When the same optimized treatment was applied to river water, performance dropped: only about 20% of PFOA disappeared in five hours. Natural water contains a mix of dissolved salts and organic matter, which compete for the same reactive species that attack PFOA or block light from reaching the catalyst particles. Some ions and natural organic compounds act as “scavengers,” soaking up radicals before they can do useful work. The study also had to contend with PFOA sticking to glass surfaces, which can make it look as if more has been destroyed than really has; the authors carefully tracked this effect to avoid overestimating treatment success.
What this means for cleaning our water
For non‑experts, the bottom line is that this UV‑based treatment can slowly nibble away at PFOA but does not yet offer a quick or complete fix. Even under ideal lab conditions, most of the pollutant remained after many hours of exposure, and real river water made the process less effective. Still, the work shows that combining UV light, TiO₂, and hydrogen peroxide does help and points to ways to improve it, such as modifying the catalyst or pairing the method with stronger oxidants like ozone. Understanding exactly how and how fast these stubborn molecules break down is an essential step toward designing future systems that can truly remove “forever chemicals” from the water we drink.
Citation: Marín, M.L.M., Peñuela, G.A. Evaluation of UV/TiO2/H2O2 photocatalysis for the removal of perfluorinated organic compounds from water. Sci Rep 16, 9638 (2026). https://doi.org/10.1038/s41598-025-34613-z
Keywords: PFOA, PFAS, photocatalysis, water treatment, advanced oxidation