Clear Sky Science · en
Pan centromeric FISH enhances precision in radiation biodosimetry
Why measuring invisible radiation matters
Radiation from medical treatments, industry, or accidents can quietly damage our DNA without any immediate outward sign. In an emergency or for workers routinely exposed to radiation, doctors and safety officials need to know quickly and accurately how much dose a person has received. This paper explores a refined laboratory technique that makes hidden damage in our chromosomes easier to spot, potentially turning fuzzy guesses about exposure into more reliable, life-guiding numbers.
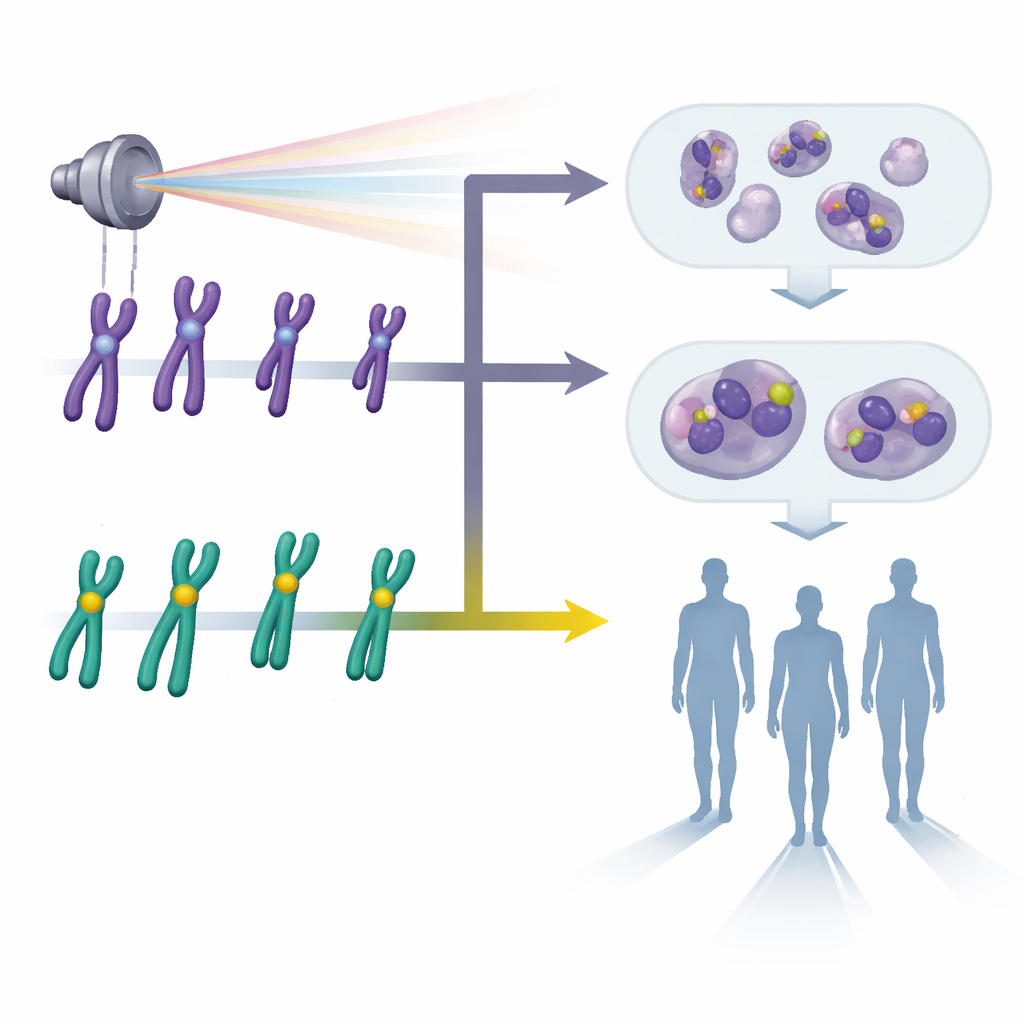
Looking for damage in the body’s instruction manual
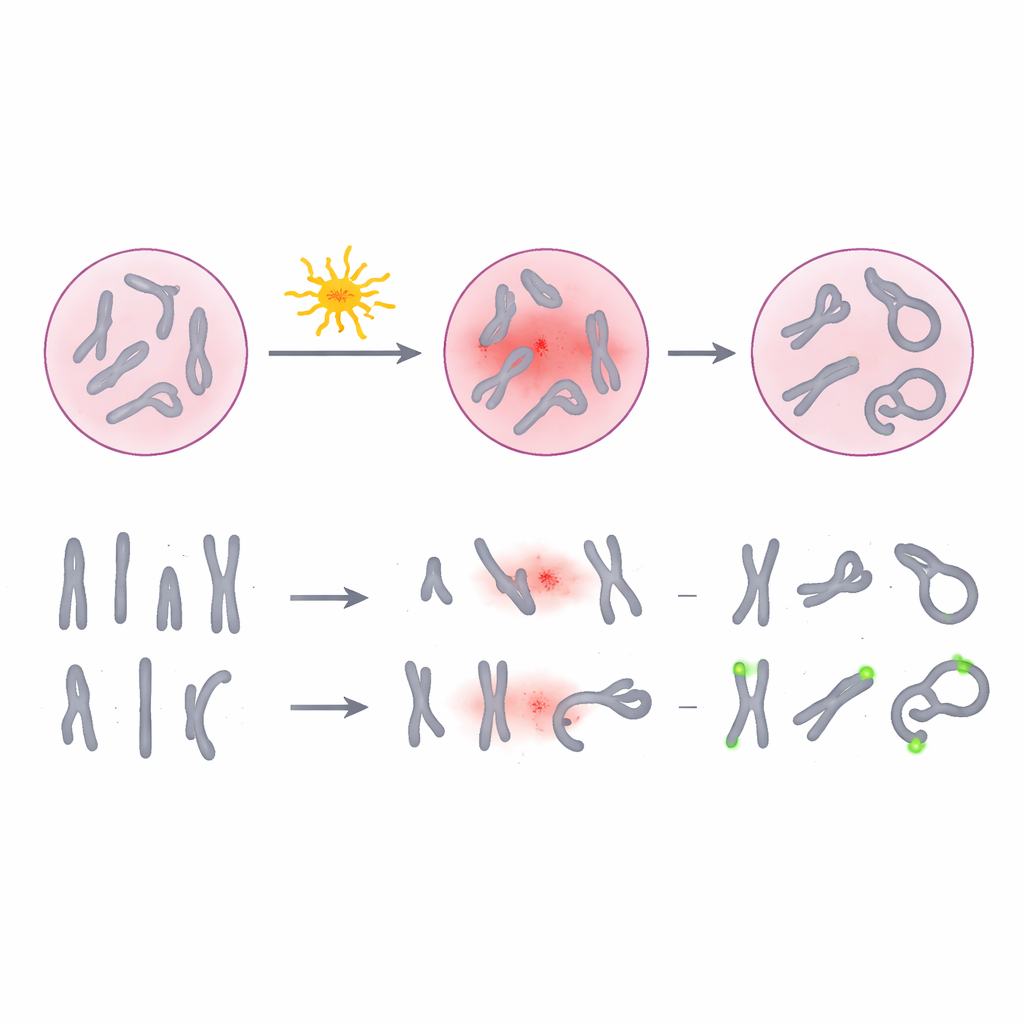
Radiation can snap and rearrange pieces of our chromosomes, the threadlike structures that carry our genetic instructions. Certain unusual chromosome shapes, called dicentrics and rings, are especially good “fingerprints” of exposure because they form mainly after radiation and appear more often at higher doses. For decades, laboratories have used a purple dye known as Giemsa to stain chromosomes in blood cells and count these telltale changes under the microscope. While this method is widely accepted and relatively inexpensive, it depends on how well a human scorer can interpret subtle shapes, especially when chromosomes overlap, are poorly spread, or look faint. At low doses—exactly where it is hardest but most important to know whether someone has been exposed—the damage can be scarce and easy to miss.
Lighting up the center of every chromosome
The researchers tested an alternative approach called pan-centromeric fluorescence in situ hybridization, or pan-cent-FISH. Instead of simply coloring entire chromosomes, this technique attaches fluorescent tags to the centromere, a small central region on each chromosome. When viewed under a special microscope, every centromere glows brightly, making it much easier to see when a chromosome has two centers (a dicentric) or forms a ring. The team collected blood from volunteers, exposed samples to controlled doses of gamma radiation from zero to three units of dose, and then prepared thousands of cell spreads using both the traditional Giemsa stain and the pan-cent-FISH method. They then carefully counted damaged chromosomes to build dose–response curves, which relate how much damage is seen to how much radiation was given.
Sharper dose estimates from brighter signals
Across more than 30,000 analyzed cells, pan-cent-FISH consistently detected more radiation-induced dicentrics and rings than Giemsa staining. The increase was most striking at low doses below half a unit, where conventional staining can easily miss rare events. When the researchers fitted mathematical curves to the data, the pan-cent-FISH curve rose more steeply, meaning it is more sensitive to changes in dose. To test practical performance, they then used both methods to estimate the dose in blinded blood samples whose true exposure was known only to the experimenters. On average, pan-cent-FISH cut the error in dose estimates roughly in half compared with Giemsa. At a very low test dose, the new method stayed within commonly accepted error limits, whereas the traditional approach drifted beyond them.
Balancing speed, effort, and real-world use
Although the fluorescent method requires special probes, a fluorescence microscope, and slightly longer preparation, it pays off during analysis. Because the glowing centromeres make abnormal chromosomes easier to recognize, scorers can move faster with fewer ambiguous cases and fewer re-checks. The technique also reduces the chances that different observers will disagree about what they see, an important benefit when many laboratories need to compare results. The authors note that Giemsa staining remains attractive for resource-limited settings due to lower costs, but argue that pan-cent-FISH offers clear advantages in situations where precision matters most, such as regulatory monitoring near legal exposure limits or triage after a large radiological incident.
Clearer chromosome pictures for safer decisions
In simple terms, this study shows that lighting up the centermost part of every chromosome gives scientists a clearer picture of radiation damage than traditional dye-based methods. By revealing more of the subtle changes in DNA structure, especially at low doses, pan-cent-FISH allows dose estimates that are closer to the truth and more consistent from one sample to the next. For workers exposed to radiation and for people caught up in nuclear or radiological emergencies, this improved clarity can translate into better medical care, more appropriate follow-up, and more confident safety decisions.
Citation: Chaurasia, R.K., Notnani, A., Vaz, D.F. et al. Pan centromeric FISH enhances precision in radiation biodosimetry. Sci Rep 16, 8020 (2026). https://doi.org/10.1038/s41598-025-34407-3
Keywords: radiation exposure, biodosimetry, chromosome damage, fluorescence in situ hybridization, radiological emergencies