Clear Sky Science · en
A novel approach to modifying eggshell-based adsorbent for the removal of acid red 1 and crystal violet dyes: kinetics, isotherm, and thermodynamics study
Turning Breakfast Waste into Cleaner Water
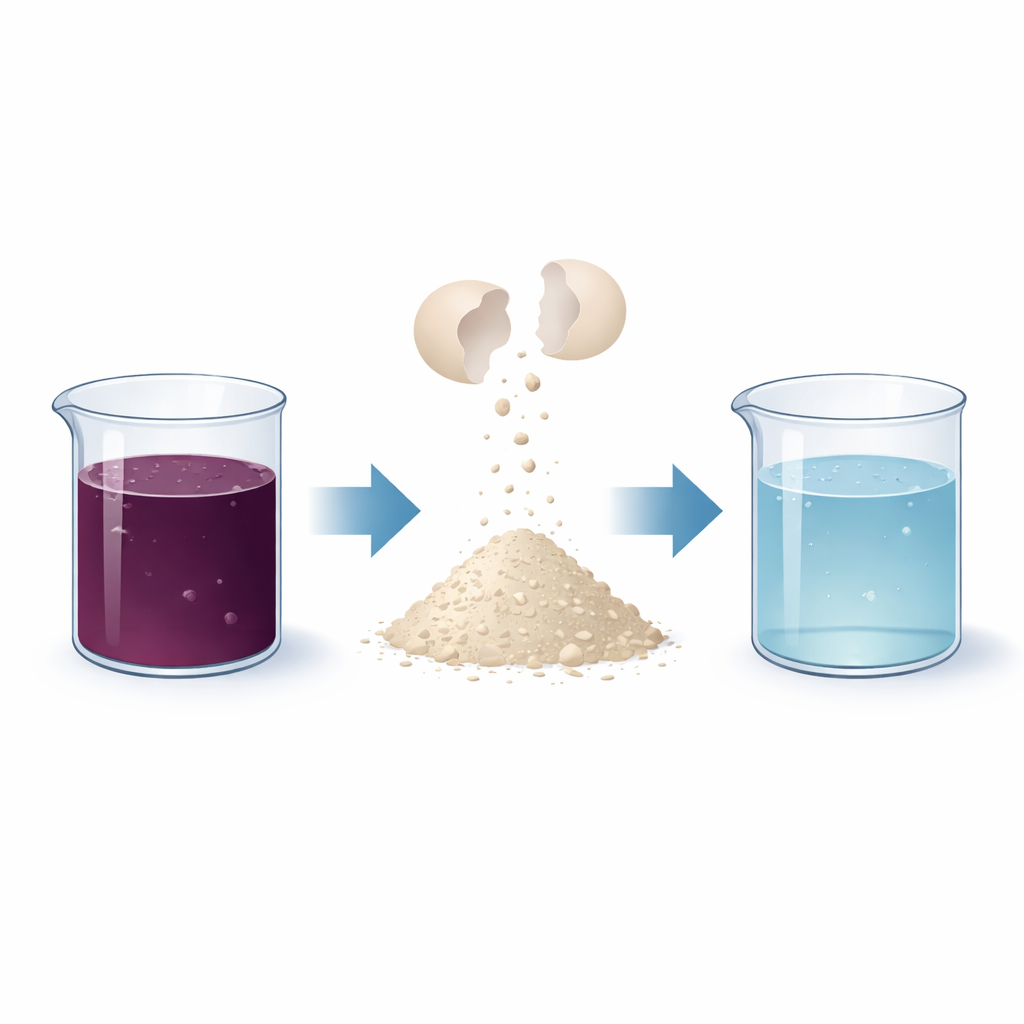
Every day, millions of eggs are cracked in homes, restaurants, and food factories, and their shells are thrown away. At the same time, textile and other industries release brightly colored dyes into rivers and lakes, where they can harm fish, plants, and even human health. This study brings those two problems together and asks a simple question: can discarded eggshells be upgraded into a cheap material that pulls toxic dyes out of water before they reach the environment?
Why Colored Water Is a Hidden Threat
Industrial dyes give clothes, paper, and many products their vivid colors, but once they enter wastewater they are difficult to remove. The two dyes examined in this work, Crystal Violet and Acid Red 1, are common in textile processing and can irritate skin and eyes, disrupt blood chemistry, and may contribute to cancer. They also block sunlight in rivers and lakes and reduce oxygen levels, disturbing entire aquatic ecosystems. Existing treatment methods can be expensive or complex, especially for factories in developing regions. That is why scientists are searching for simple, low-cost materials that can "grab" these dye molecules from water and hold onto them.
From Eggshells to a Powerful Filter
Eggshells are mostly calcium carbonate, a mineral that already has some ability to attract charged molecules. In this study, the researchers collected waste eggshells from restaurants, cleaned and finely ground them, and then treated the powder with a solution of ferrous sulfate, a common iron salt. This chemical step coats and partly alters the surface of the shells, creating more tiny pores and new reactive spots where dyes can attach. Using techniques such as X-ray diffraction, electron microscopy, and surface-area measurements, the team confirmed that the treated shells became more porous, gained iron-containing features, and offered a larger active surface than the raw material.
How the Modified Shells Catch Dyes
To test performance, the scientists mixed tiny amounts of the modified eggshell powder with dyed water under controlled conditions and tracked how fast and how completely the color disappeared. They varied the water’s acidity, the starting dye concentration, contact time, and temperature. For the purple Crystal Violet, removal worked best in mildly basic water; for the red Acid Red 1, it worked best in acidic water. This is because the surface of the eggshells can be positively or negatively charged depending on pH, which in turn attracts oppositely charged dye molecules. Within about half an hour, the modified shells were able to capture up to 138 milligrams of Crystal Violet and 124 milligrams of Acid Red 1 per gram of powder—roughly doubling or more the capacity of unmodified shells.
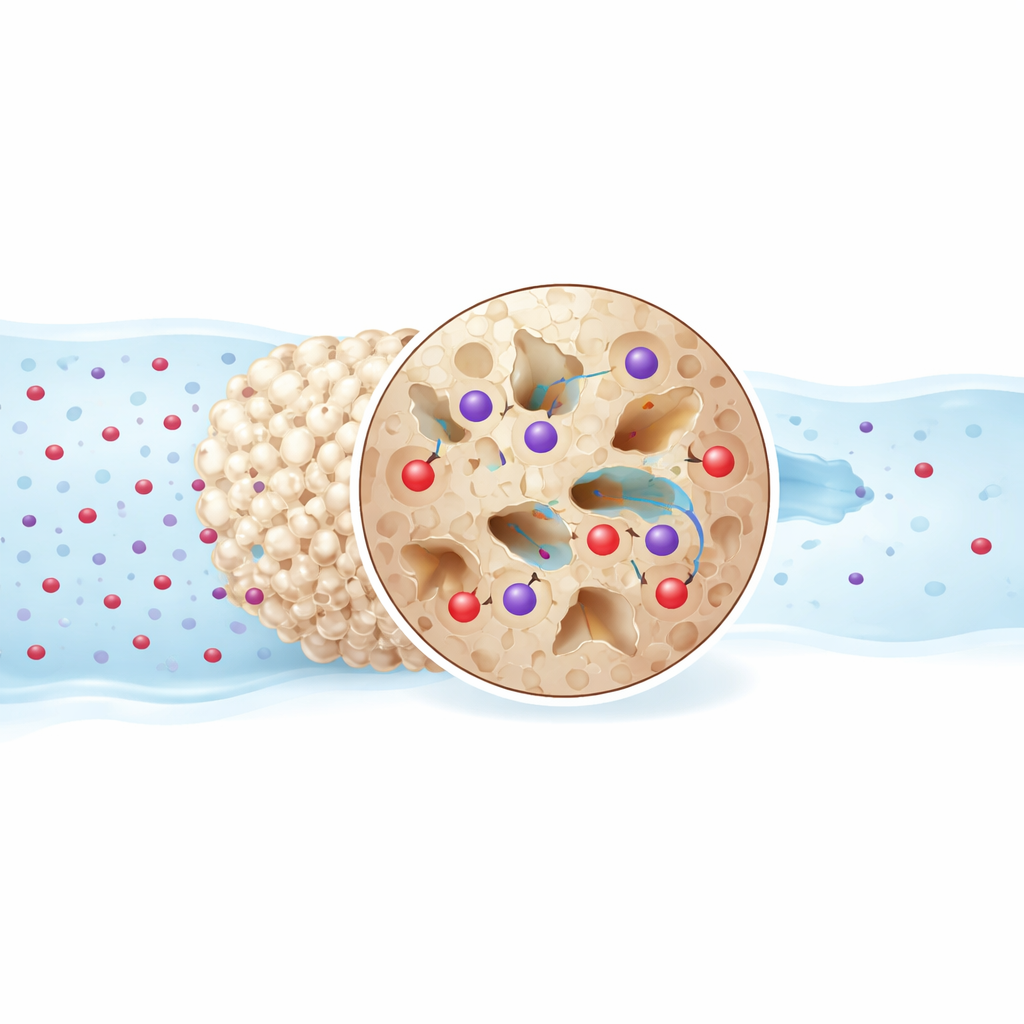
What Happens at the Microscopic Level
Closer analysis shows that dye molecules first move through the water to the outer surface of the grains and then gradually work their way into the pores. The data suggest that Crystal Violet sticks through a stronger, more chemical-like attachment, while Acid Red 1 follows a somewhat simpler, more physical pattern. Both, however, rely on a mix of attractions between opposite charges and the ability of certain chemical groups on the shell surface to form weak bonds with the dyes. The process releases heat, so it works better at room temperature than at higher temperatures, and it becomes less favorable as the water warms up. The team also tested whether the same batch of modified shells could be washed and reused, finding that they still removed substantial amounts of dye over several cycles, although the capacity gradually declined.
Cost, Reuse, and Real-World Promise
Because eggshells are a plentiful kitchen waste and the iron treatment uses relatively simple chemicals, the overall cost of the new adsorbent is low. When the researchers compared it with commercial activated carbon, a standard but more expensive filtering material, the modified shells achieved dye removal at a much lower cost per unit of treated water. Even after accounting for energy use and regeneration steps, the approach appears economically attractive. While this work was done in laboratory flasks rather than full-scale treatment plants, it points toward a practical way to turn a troublesome waste stream—discarded eggshells—into a useful tool for cleaning up another, more dangerous one: dye-contaminated water.
Everyday Impact of the Findings
In plain terms, this study shows that something as ordinary as an eggshell, when cleverly modified, can help strip harmful dyes from water efficiently and cheaply. The treated shells grab different types of dye under different pH conditions, hold large amounts of them, and can be reused several times. If scaled up, this strategy could offer factories and municipalities in many countries an affordable way to reduce water pollution while also cutting down on solid waste—bringing us a step closer to safer rivers and more sustainable use of everyday materials.
Citation: Azeem, A.A., Khalek, M.A.A. & Hamid, E.M.A. A novel approach to modifying eggshell-based adsorbent for the removal of acid red 1 and crystal violet dyes: kinetics, isotherm, and thermodynamics study. Sci Rep 16, 8721 (2026). https://doi.org/10.1038/s41598-025-34343-2
Keywords: wastewater treatment, textile dyes, eggshell adsorbent, crystal violet, acid red 1