Clear Sky Science · en
Waste-derived nano-Al₂O₃-loaded pyranopyrazole composite for high-capacity cadmium and methylene blue removal with mechanistic and DFT validation
Turning Trash into a Tool for Clean Water
Industrial dyes and toxic metals are among the most stubborn pollutants in rivers and lakes worldwide. This study shows how something as ordinary as discarded aluminum drink cans can be transformed into a powerful cleaning material that pulls both bright blue dye and poisonous cadmium out of water at the same time. By marrying this recycled metal with a tailor‑made organic molecule, the researchers create a reusable sponge‑like material that helps tackle two major water problems in one stroke.
Why Colorful Dyes and Hidden Metals Matter
Many factories that make textiles, paper, and electronics release wastewater containing both vivid dyes and invisible heavy metals. The blue dye methylene blue can damage cells and cause stress in living organisms, while cadmium, a metal used in batteries and pigments, is a known cancer risk that builds up in kidneys, liver, and lungs. These substances do not break down easily in nature, so once they enter waterways they can persist for years, moving up the food chain and eventually reaching drinking water. Conventional treatment methods can be expensive, energy‑hungry, or work well for either dyes or metals, but rarely both together. That gap has driven a search for simple, low‑cost materials that can latch onto many pollutants at once.
Designing a Dual‑Action Cleaning Sponge
The team starts by building an organic “skeleton” called Pyrano PY, assembled in a single reaction from small, readily available chemicals, including a component derived from plant biomass. This molecule is packed with useful features: nitrogen and oxygen atoms that can grip metal ions, and flat aromatic rings that can attract dye molecules. Next, they produce tiny particles of aluminum oxide (alumina) from shredded beverage cans through straightforward acid, base, and heating steps. These nanoparticles are then anchored onto the Pyrano PY framework in water, forming a hybrid material in which the organic scaffold and inorganic alumina are tightly intertwined. Microscopy, elemental mapping, and infrared spectroscopy confirm that alumina dots coat the fibrous organic surface without clogging its pores, creating many new reactive spots while preserving its open structure.
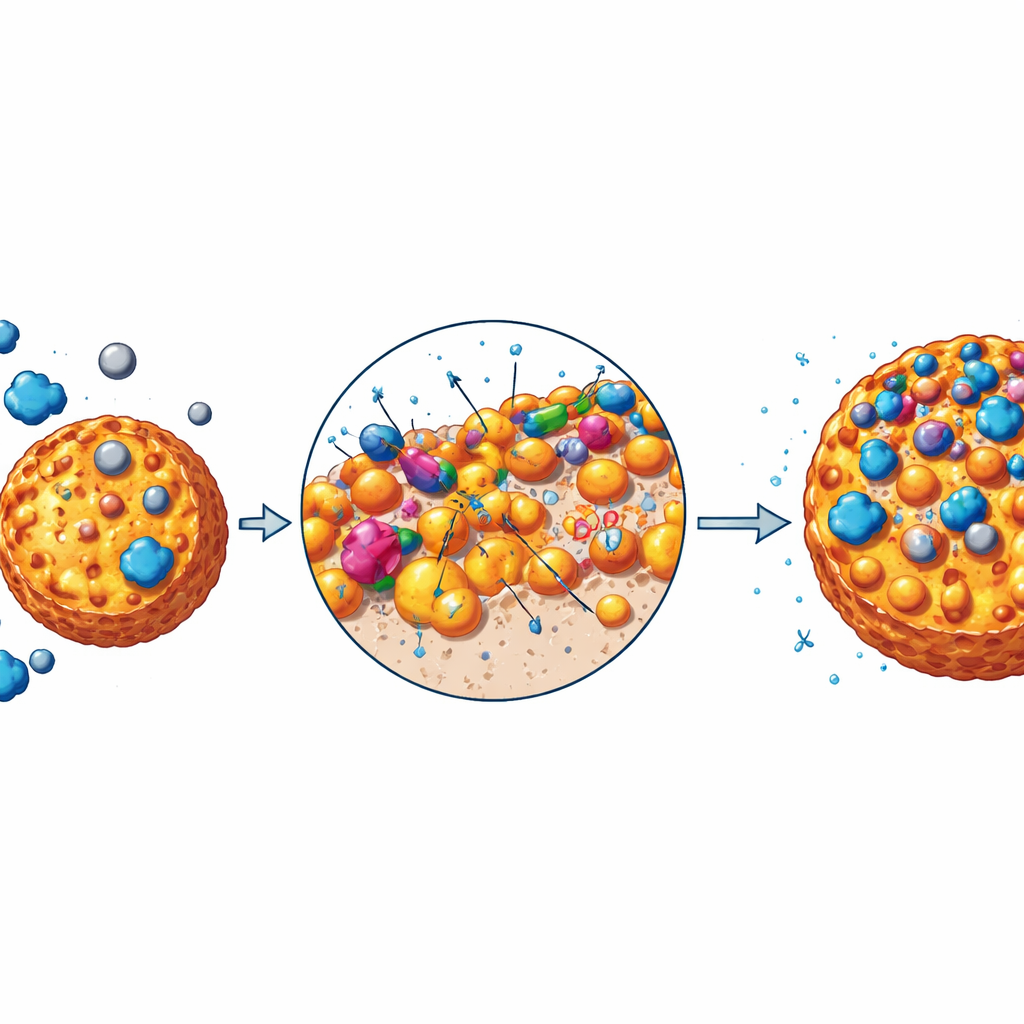
How the New Material Cleans Up Water
To test performance, the researchers shake the hybrid particles in water containing methylene blue or cadmium under different conditions. Both the pure organic material and the alumina‑loaded version remove large amounts of pollutants, but the hybrid does significantly better: up to about 190 milligrams of dye and 343 milligrams of cadmium per gram of material under optimized conditions. The process is fastest in the first two hours and works best near neutral to slightly basic pH, similar to many real wastewaters. Mathematical models show that the rate and extent of uptake are controlled mainly by chemical bonding at the surface rather than simple physical sticking. The particles behave like a rugged landscape of sites with different strengths, which helps them grab both flat dye molecules and charged metal ions. As temperature rises, removal slightly decreases, indicating that the binding is exothermic but still spontaneous and favorable across typical treatment temperatures.
Peeking Inside the Adsorption Process
The authors combine their lab tests with computer calculations based on quantum mechanics to understand why the material works so well. These simulations reveal that electrons in the Pyrano PY framework cluster around nitrogen and oxygen atoms, marking them as prime “hooks” for positively charged cadmium. The alumina phase contributes additional metal‑friendly oxygen atoms and surface hydroxyl groups, so cadmium can be clasped at several points at once. For methylene blue, negatively charged regions on the material’s surface attract the positively charged dye, while the extended flat rings in the framework allow the dye to stack like playing cards. Altogether, coordination, electrostatic forces, hydrogen bonds, and stacking interactions act in concert, explaining the high capacity and strong preference for these pollutants.
Using and Reusing the Cleaner
For any real‑world water treatment technology, reusability is crucial. The researchers show that both the pure and hybrid materials can be regenerated several times by rinsing with mild acid (for cadmium) or base (for the dye), maintaining over 90 percent of their original performance after five cycles. Because the alumina comes from waste cans and the synthesis uses common chemicals under mild conditions, the overall process is cost‑conscious and compatible with standard treatment setups such as stirred tanks or packed columns. Tests with real industrial wastewater further confirm that the hybrid material performs well outside the lab.
What This Means for Safer Water
In plain terms, this work takes a common trash item and turns it into a smart, reusable filter that can capture both a toxic metal and a stubborn dye from water, even when they are present together. By carefully designing the chemistry of the organic framework and decorating it with recycled nano‑alumina, the authors create a material whose internal “hooks” are perfectly suited to catching these pollutants. The combination of high capacity, good stability, and simple regeneration suggests that such hybrids could help make large‑scale wastewater treatment more affordable, more sustainable, and more effective at protecting people and ecosystems from hidden chemical hazards.
Citation: Abouelenein, M.G., Elfattah, M.A., Safan, N.M. et al. Waste-derived nano-Al₂O₃-loaded pyranopyrazole composite for high-capacity cadmium and methylene blue removal with mechanistic and DFT validation. Sci Rep 16, 8720 (2026). https://doi.org/10.1038/s41598-025-34070-8
Keywords: wastewater treatment, heavy metal removal, dye pollution, adsorbent materials, recycled aluminum