Clear Sky Science · en
Effect of telomere length and related gene polymorphism in signaling pathway on semen quality
Why tiny chromosome caps matter for male fertility
Many couples struggling to conceive are told that a man’s semen test looks “normal,” yet pregnancy still doesn’t happen. This study looks beyond the usual lab report and asks whether microscopic “caps” at the ends of sperm chromosomes, called telomeres, and small genetic differences in the telomere system, can help explain why some men have healthier sperm than others. Understanding these hidden factors could improve how we assess male fertility and guide earlier, more personalized care for would‑be parents.
The search for better fertility clues
Standard semen tests measure how many sperm a man has, how well they move, and how normal they look. These tests are useful, but they don’t always capture subtle problems. At the same time, research on telomeres—protective DNA repeats that help keep chromosomes stable—has suggested that they may be linked to fertility. Some studies found that shorter telomeres in sperm are associated with lower sperm counts and poorer movement, while others did not. Most earlier work also assumed a simple straight‑line relationship: longer is always better. The authors of this new study set out to test these ideas in a large group of men and to ask whether common genetic variants in telomere‑related genes also play a role.

How the study was carried out
The researchers recruited 1,349 male volunteers from a reproductive medicine center in Henan Province, China. All participants completed questionnaires and physical exams, and most provided semen samples; a subset also gave blood. The team used computer‑assisted methods to measure semen volume, total sperm number, concentration, how well sperm moved, and how normal they appeared. For 536 men, they also measured sperm telomere length using a sensitive DNA technique. In addition, they analyzed common single‑letter changes in several telomere‑related genes, including POT1, TERF1, TERT, and TERC, to see whether particular genetic patterns were linked to telomere length or semen quality.
A “just right” zone for telomere length
Most men in the study met World Health Organization benchmarks for sperm count and movement, but fewer than one in three had normal sperm shape. When the team compared sperm telomere length with semen measures, they found something striking: the relationship was not simply “the longer, the better.” Using flexible statistical models, they observed a curved pattern between telomere length and both sperm concentration and total sperm count. Men whose telomere length fell in a middle range had better sperm numbers and movement, and they were less likely to have abnormal semen results than men in the lowest group. However, very short and very long telomeres were each linked to less favorable sperm counts, suggesting there may be a “Goldilocks” range of telomere length that best supports sperm production.
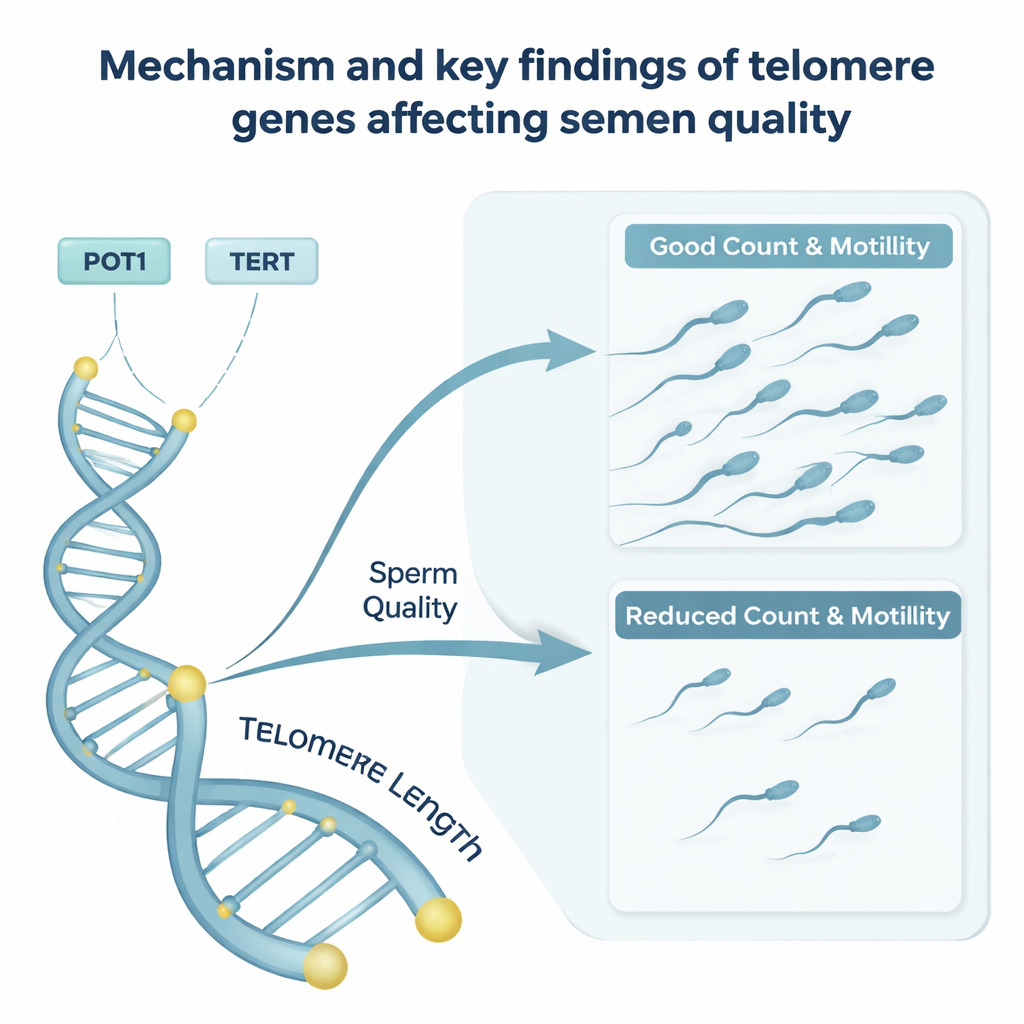
Genes that nudge telomeres and sperm quality
The study also examined whether inherited differences in telomere‑related genes help explain why some men fall inside or outside this healthy telomere window. Variants in the POT1 gene were especially notable. Men carrying a particular version of POT1 had longer sperm telomeres, yet this same variant was tied to lower total sperm counts and poorer sperm movement. Another POT1 variant was also associated with reduced sperm number and vitality. Certain versions of the TERT gene, which encodes the main component of the telomerase enzyme that maintains telomeres, were linked to changes in how straight and smoothly sperm swim. These patterns suggest that some genetic changes may push telomeres beyond their ideal range or alter how well they function, indirectly harming semen quality.
What this means for men and couples
This work supports the idea that sperm telomere length could become a useful biomarker—an additional piece of information—to help judge semen quality, especially when conventional tests are borderline or puzzling. It also shows that more is not always better: both too‑short and too‑long telomeres may be problematic, and common genetic variants in telomere genes can shift men toward or away from this optimal zone. Although the study cannot prove cause and effect and was done in a single center, it opens the door to future research and, eventually, to more refined male fertility evaluations that include the health of chromosomes themselves, not just the visible behavior of sperm.
Citation: Jia, X., Cao, J., Zhang, S. et al. Effect of telomere length and related gene polymorphism in signaling pathway on semen quality. Sci Rep 16, 6575 (2026). https://doi.org/10.1038/s41598-025-34054-8
Keywords: male infertility, sperm quality, telomere length, genetic variation, reproductive health