Clear Sky Science · en
Targeted hypoxia-inducible factor 1-alpha (HIF1A) stabilization during in vitro maturation of bovine cumulus-oocyte complexes increases blastocyst rates
Why tweaking oxygen sensing in eggs matters
Modern cattle breeding relies heavily on laboratory-made embryos, but success is far from guaranteed: only about a third of egg cells reach the crucial blastocyst stage, when they are ready to be transferred to a cow. This study asks a deceptively simple question with broad relevance for animal breeding and assisted reproduction: if we more closely mimic the low-oxygen conditions that eggs experience naturally inside the ovary—by gently boosting a key oxygen-sensing protein—can we help more embryos develop successfully?
Life begins in a low‑oxygen niche
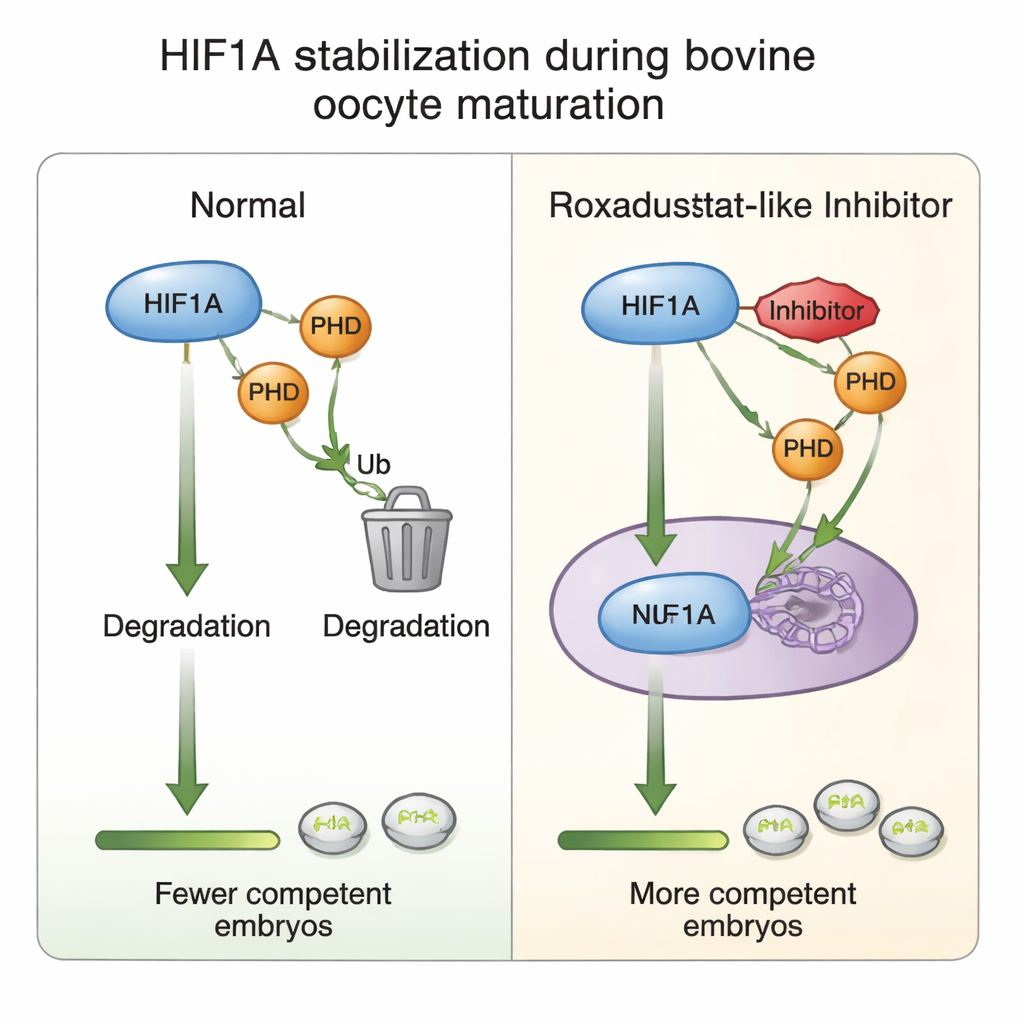
Inside the ovary, each developing egg is wrapped in a supportive cluster of cells known as the cumulus, together forming a cumulus–oocyte complex. Although the ovary becomes more richly supplied with blood as follicles grow, blood vessels stop short of the egg’s immediate surroundings. Oxygen must diffuse in, creating a mildly low-oxygen, or hypoxic, pocket around the maturing egg. In this niche, an intracellular sensor called HIF1A helps cells adapt by adjusting metabolism and hormone production. Earlier work showed that blocking HIF1A harms egg maturation and drastically reduces embryo formation, highlighting this protein as a central coordinator of egg competence—the ability of an egg to be fertilized and develop into a healthy embryo.
Using a medicine to fine‑tune the egg’s oxygen sensor
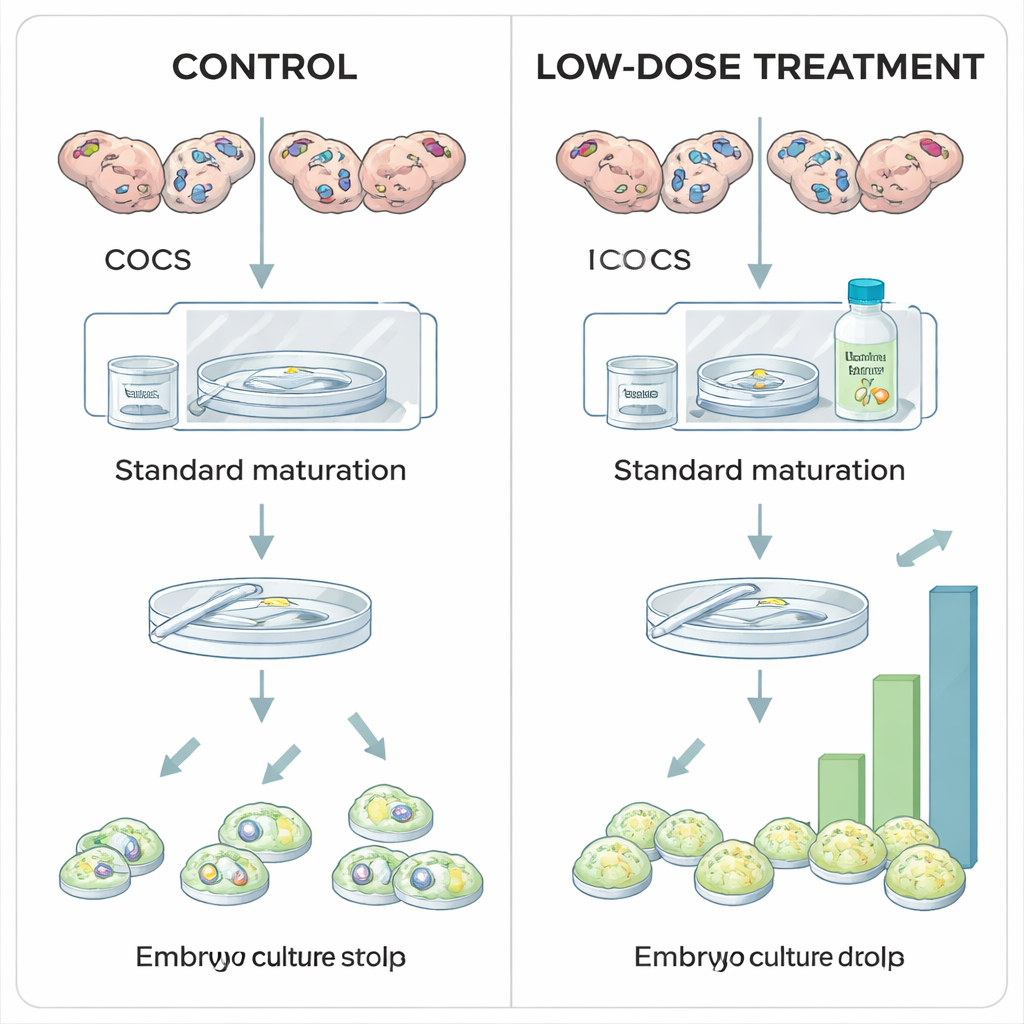
The researchers explored whether moderately stabilizing HIF1A during laboratory maturation of bovine eggs could boost embryo yields. In standard in vitro protocols, cumulus–oocyte complexes are matured under normal air oxygen levels, largely for practical reasons rather than biological ones. Here, eggs collected from slaughterhouse ovaries were divided into groups and exposed during their 24‑hour maturation period to different doses of Roxadustat, a drug already used in human medicine to treat anemia. Roxadustat works by blocking a family of enzymes called prolyl hydroxylases (PHDs) that normally tag HIF1A for destruction when oxygen is plentiful. By inhibiting PHDs, Roxadustat allows HIF1A protein to accumulate even at higher oxygen levels, partially recreating the hypoxic signal the egg would sense in vivo.
More blastocysts at the right dose, harm at higher levels
When the team examined how eggs and embryos fared after treatment, a clear pattern emerged. A low dose of Roxadustat (25 micromolar) did not change how many eggs completed maturation or how many fertilized eggs began dividing, but it significantly increased the proportion that reached the blastocyst stage. In other words, early steps were unaffected, yet more embryos successfully completed the demanding journey to become blastocysts—suggesting that modest HIF1A stabilization improves the intrinsic quality of the egg. In contrast, the highest dose (100 micromolar) reduced the percentage of eggs that matured at all, and when the same inhibitor was applied later, during the eight‑day embryo culture period, high doses sharply cut blastocyst formation without altering early cell division. These findings indicate that "just enough" HIF1A activity during maturation is beneficial, whereas too much or too prolonged activation is harmful.
What happens inside the egg’s support cells
To understand how this chemical tuning works, the authors probed the cumulus cells that surround each egg. They found that maturation in general raises HIF1A gene activity, but adding Roxadustat paradoxically lowered HIF1A messenger RNA while leaving HIF1A protein levels steady—a hint that cells may dampen gene output once the protein is sufficiently stabilized, a kind of built‑in brake against overactivation. Levels of PHD2, the most oxygen‑sensitive of the PHD enzymes, rose in response to the drug, reinforcing its role as part of the oxygen‑sensing loop. At higher drug doses, markers of cell proliferation and of the sticky matrix that helps cumulus cells expand around the egg were reduced, in line with the observed decline in maturation and embryo formation. All of this supports the idea that carefully balanced HIF1A activity fine‑tunes the dialogue between egg and cumulus cells that underpins later developmental success.
Implications for animal breeding and beyond
This work shows that gently stabilizing the egg’s oxygen sensor during the short window of in vitro maturation can meaningfully increase and stabilize blastocyst yields in cattle, without changing fertilization or early cleavage. In practical terms, that could translate into more reliable embryo production for breeding programs, using a targeted drug rather than crude changes in oxygen levels or more toxic chemicals. The study also underscores a broader lesson relevant to human fertility treatments: mimicking the nuanced, low‑oxygen environment of the ovary is not simply a matter of turning oxygen up or down, but of respecting the delicate balance of signaling molecules like HIF1A. Fine‑tuning that balance, rather than pushing it to extremes, may be a key route to healthier embryos.
Citation: Gübeli, M., Bleul, U. & Kowalewski, M.P. Targeted hypoxia-inducible factor 1-alpha (HIF1A) stabilization during in vitro maturation of bovine cumulus-oocyte complexes increases blastocyst rates. Sci Rep 16, 7554 (2026). https://doi.org/10.1038/s41598-025-33894-8
Keywords: in vitro embryo production, oocyte maturation, hypoxia signaling, HIF1A stabilization, bovine reproduction