Clear Sky Science · en
Robust learning framework for a scalable remote monitoring of autonomic dysreflexia: use-case in spinal cord injury
Why sudden blood pressure spikes matter
For many people living with a spinal cord injury, the biggest daily threat is not just paralysis, but sudden, extreme surges in blood pressure that can lead to stroke or even death. These episodes, known as autonomic dysreflexia, can strike with little warning and are hard to track outside the hospital. This study shows how a combination of simple wearable sensors and advanced computer algorithms could turn everyday devices into early‑warning systems, offering continuous protection at home instead of short, occasional checks in the clinic.
Hidden danger after spinal cord injury
Spinal cord injury often disrupts the body’s automatic control of blood pressure and heart rate. In people with injuries high on the spine, even minor triggers such as a full bladder can cause blood pressure to soar within minutes. Because these events are unpredictable and not always felt by the patient, they are frequently missed until serious symptoms appear. Traditional monitoring depends on blood pressure cuffs and clinic visits, which are too infrequent to catch most episodes. The authors set out to answer a practical question: can small, non‑invasive sensors worn on the skin pick up early signs of these dangerous spikes reliably enough to guide real‑time care?
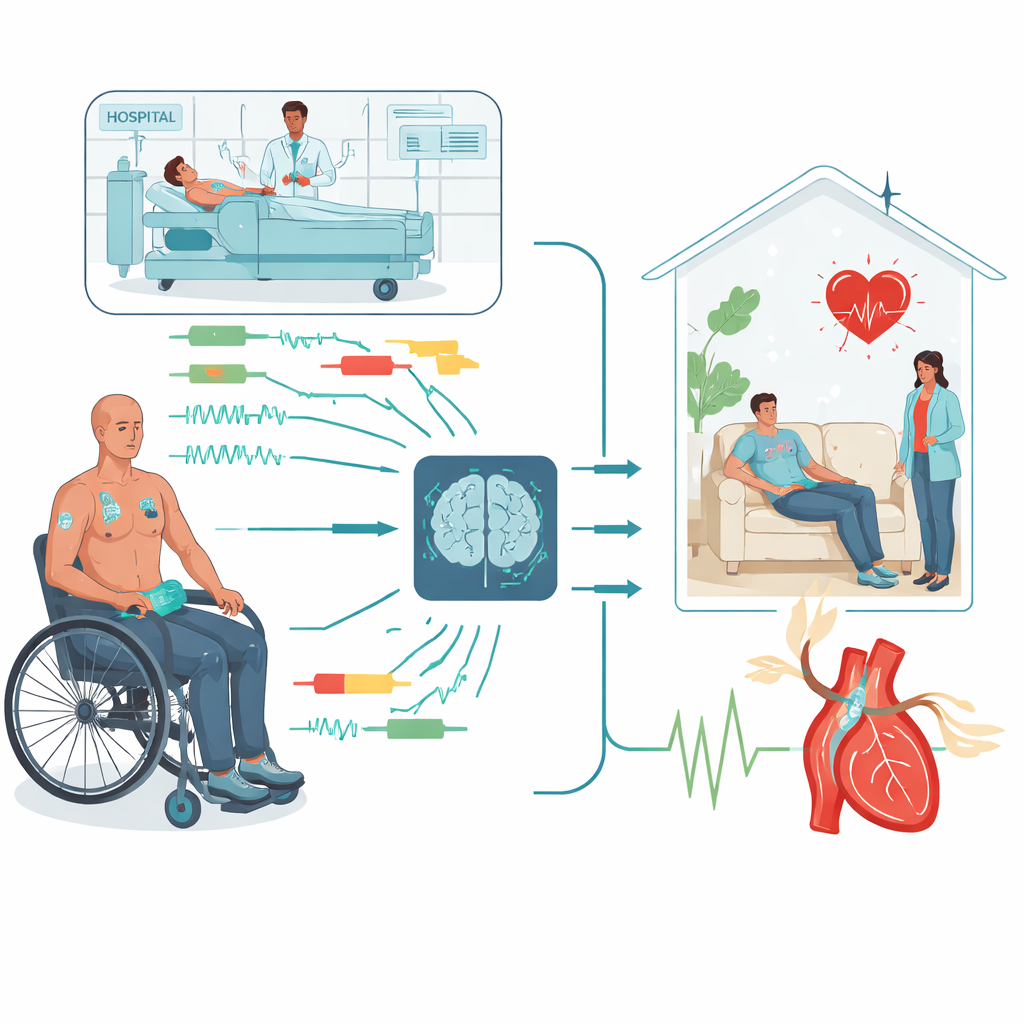
Turning body signals into a digital warning light
The research team equipped 27 people with chronic spinal cord injury with a set of wearable devices during a routine bladder test that often triggers blood pressure changes. A wristband measured pulse waves, skin temperature, heart rate, and skin conductance; a chest patch recorded electrical activity of the heart and breathing; and a separate patch measured core and skin temperature. At the same time, a medical‑grade cuff recorded blood pressure in the usual way. By aligning all of these recordings in time, the researchers built a rich picture of how the body’s signals changed before, during, and after each blood pressure surge.
Teaching computers to read the body’s rhythms
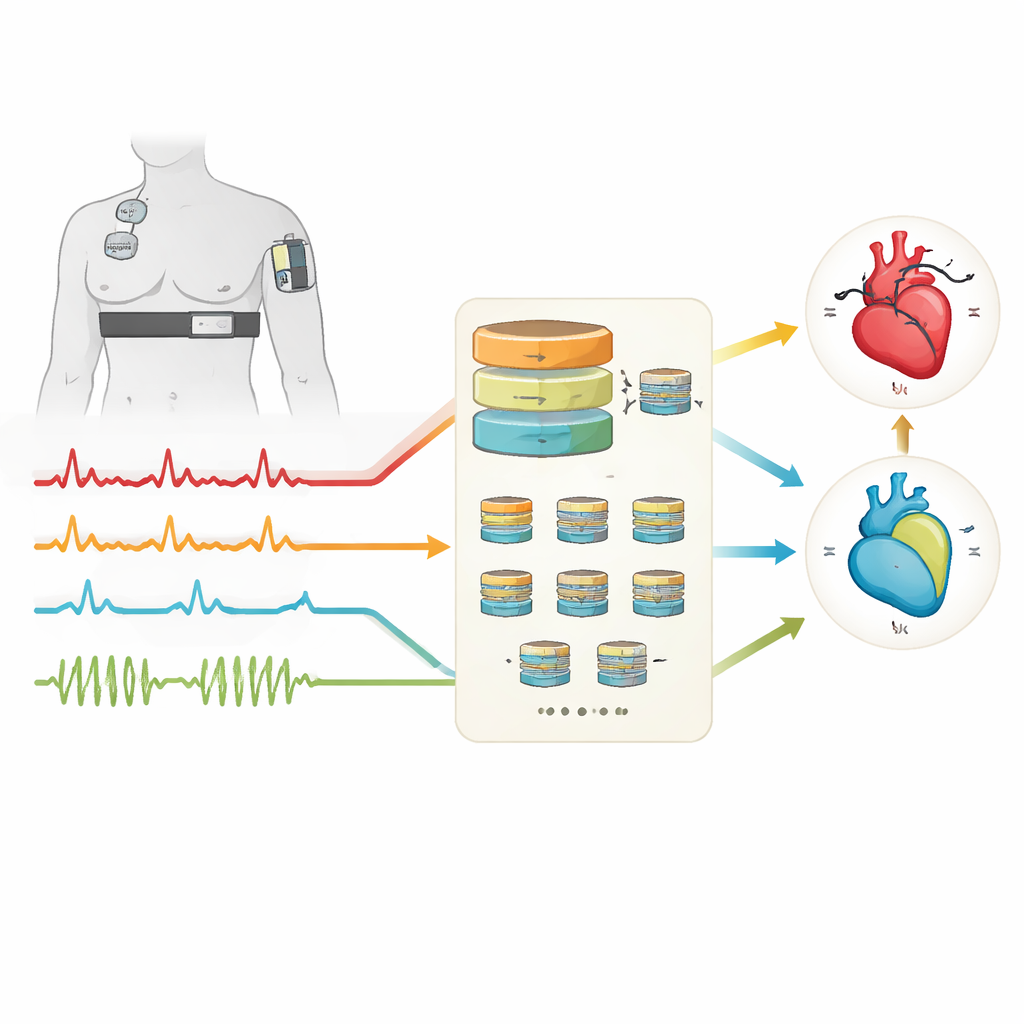
From these raw signals, the team extracted hundreds of numerical features describing how the heart was beating, how the pulse wave looked, how skin conductance and temperature were shifting, and how these patterns evolved over short time windows. They then trained an ensemble of machine‑learning models—multiple smaller classifiers that each specialize in one sensor or signal type and vote together—to distinguish dangerous episodes from normal periods. Importantly, the models were tested in a strict cross‑subject manner: they were trained on data from some people and then asked to detect episodes in people they had never “seen” before, mirroring how such a system would operate in new patients.
Which signals matter most when sensors fail
To make the system practical in real life, the authors examined not only how accurate it was, but also how it behaved when sensors were noisy or dropped out—an everyday reality for wearables. Using an interpretability method based on Shapley values, they ranked which features and signal types contributed most to correct detections. Heart‑related measures from the chest patch, including heart rate, its variability over time, and detailed shapes of the electrical heartbeat, emerged as the strongest indicators of an episode. Pulse‑wave features from the wrist added useful support, while breathing rate and temperature played more modest roles. When they simulated the loss of certain sensors, combinations that kept the heart and chest‑patch information continued to perform well, showing that the system can stay robust even with partial data.
From hospital tests to everyday protection
Overall, the best ensemble configuration correctly distinguished dangerous episodes with performance much better than chance, even though true events were relatively rare in the data. Longer observation windows—about one minute of signal at a time—helped the models capture the sustained cardiovascular shifts that characterize these spikes. While the study involved only 27 participants and was carried out in a controlled clinical setting, it provides a concrete blueprint for how wearable devices and interpretable artificial intelligence can work together to monitor an otherwise invisible risk.
What this could mean for daily life
To a layperson, the key takeaway is that ordinary‑looking patches and wristbands could one day act as a round‑the‑clock safety net for people with spinal cord injury. By carefully combining signals from the heart, pulse, skin, and temperature, and by designing algorithms that keep working even when some sensors misbehave, this framework moves remote monitoring of dangerous blood pressure surges from theory toward reality. With further testing in larger and more varied groups, similar systems could help patients and caregivers spot trouble early, act before a crisis unfolds, and ultimately reduce the risk of life‑threatening complications at home.
Citation: Fuchs, B., Ejtehadi, M., Cisnal, A. et al. Robust learning framework for a scalable remote monitoring of autonomic dysreflexia: use-case in spinal cord injury. Sci Rep 16, 9618 (2026). https://doi.org/10.1038/s41598-025-33797-8
Keywords: spinal cord injury, autonomic dysreflexia, wearable sensors, remote patient monitoring, machine learning in healthcare