Clear Sky Science · en
Androgen dysregulates the follicular extracellular matrix and increases pro-fibrotic gene expression in the mouse ovary
Why This Matters for Women’s Health
Many women with polycystic ovary syndrome (PCOS) struggle with irregular periods and difficulties getting pregnant, but the hidden story takes place in the tissue architecture of the ovary itself. This study uses mouse ovaries to probe how male-type hormones, called androgens, reshape the microscopic support scaffolding around developing egg follicles. The work helps explain why ovaries in PCOS often look and feel more fibrous and stiff, and how that altered environment could interfere with normal egg development and ovulation.
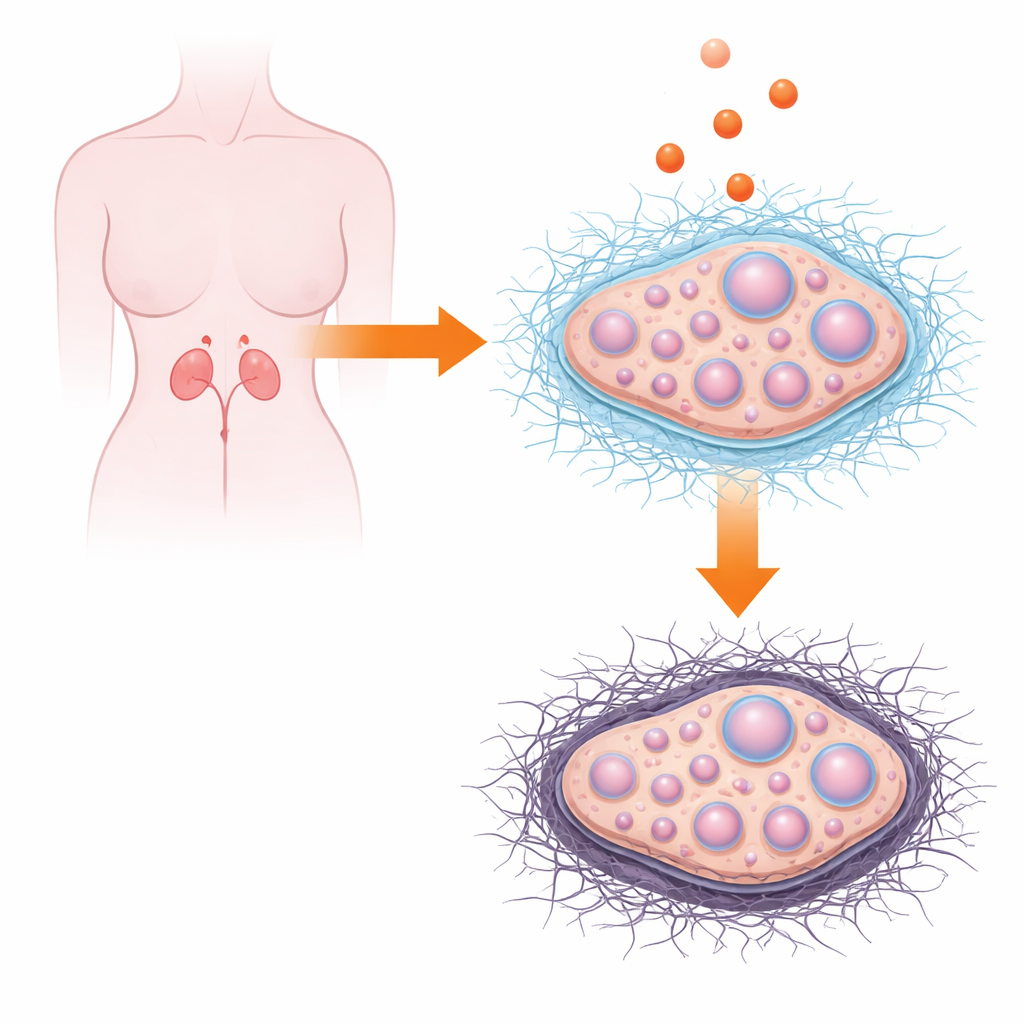
The Ovary’s Hidden Support Network
Every organ in the body is built on an intricate “molecular scaffolding” known as the extracellular matrix. In the ovary, this mesh of proteins and sugars wraps around and between follicles, the tiny fluid-filled structures that house developing eggs. The matrix is not static: it is constantly remodeled each cycle so that follicles can grow, form fluid-filled cavities, and eventually release a mature egg. Previous work in women with PCOS has shown that their ovaries often contain extra connective tissue and appear denser than normal, hinting at scarring, or fibrosis. The authors set out to identify which matrix-related genes and pathways exist in the mouse ovary and then to ask how they respond when follicles are exposed directly to androgens in the lab.
Mapping the Building Blocks in the Ovary
The researchers first drew up a panel of genes involved in structural matrix proteins, enzymes that modify those proteins, and molecules that help cells sense and respond to mechanical forces. Using mouse ovaries, they confirmed that many of these genes are active in different follicle stages, in the surrounding stromal tissue, and even in the egg cells themselves. Under the microscope, several key proteins, such as collagen, laminin, and a form of actin, formed ring-like layers around growing follicles, while others linked cells to the matrix and helped crosslink fibers. This pattern showed that the ovary’s support network is carefully organized to encase each follicle, suggesting that any disruption in these rings or in the surrounding mesh could alter how follicles grow and how stiff their environment feels.
Androgens Tip the Balance Toward Scarring
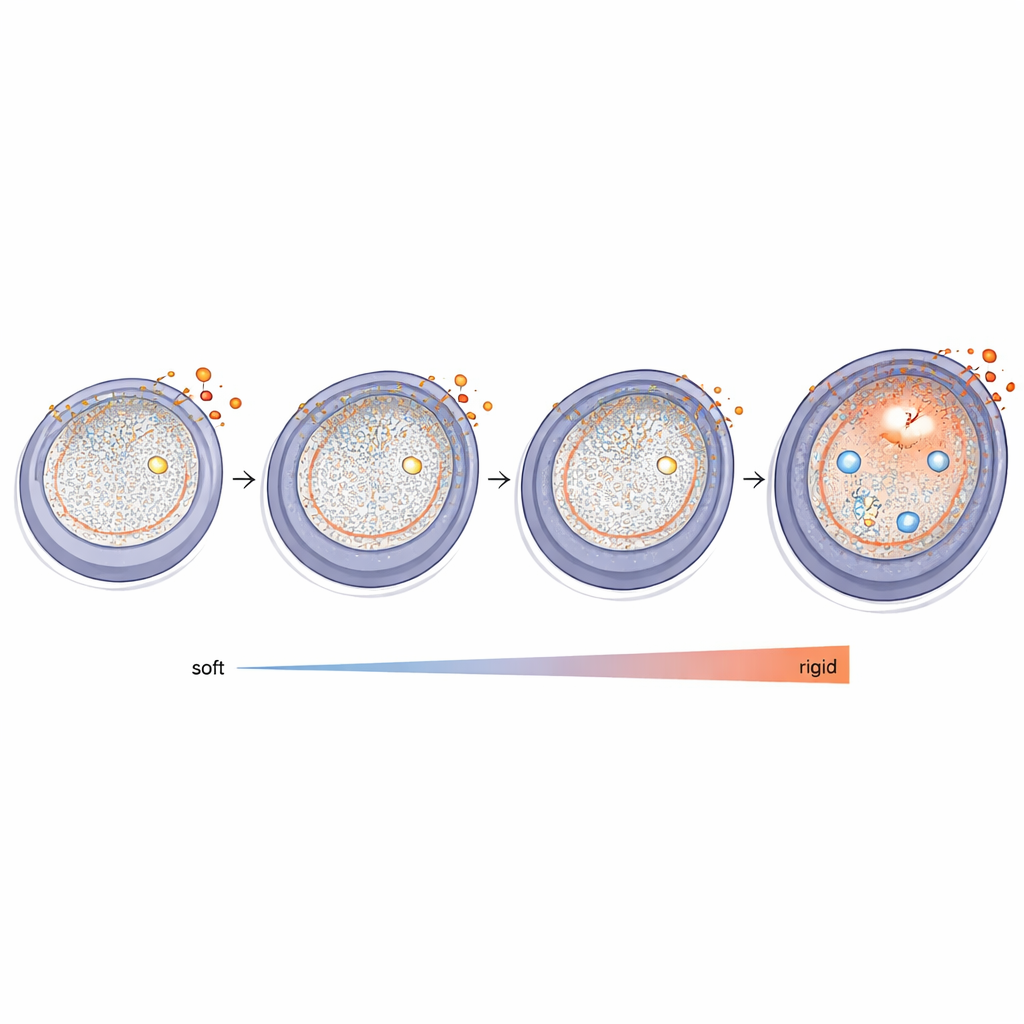
Next, the team isolated early-stage mouse follicles and cultured them with or without the potent androgen dihydrotestosterone. As expected from earlier studies, follicles exposed to androgen grew more quickly. But beneath this growth spurt, their gene activity shifted in ways that point toward a more fibrotic ovary. After one day, many structural matrix genes dropped in activity, particularly components of the thin basement membrane that forms the follicle’s outer boundary. After three days, some components rebounded or even increased, including elastin and alpha–smooth muscle actin, both associated with stiff scar-like tissue. The follicles also boosted genes for enzymes that strengthen collagen fibers or remodel sugar-rich matrix, consistent with a transition from a flexible to a denser, tougher tissue environment.
Mechanical Signals Grow Louder
The study also zoomed in on how follicles sense and translate mechanical cues as their surroundings stiffen. Androgen-treated follicles showed higher activity in several core players of the Hippo pathway, a signalling network known to respond to tissue tension and to limit organ size. Downstream targets of this pathway, including connective tissue growth factor and other matrix-associated signals, were also elevated, indicating that mechanical messages were reaching the cell nucleus and altering gene programs. In parallel, two members of the TGF-beta family, powerful drivers of fibrotic responses in many organs, were upregulated. Together, these changes suggest a feedback loop in which androgens not only change the matrix but also dial up pro-fibrotic and mechanosensitive signalling inside follicle cells.
What This Means for PCOS
To a non-specialist, the key message is that excess androgens can push ovarian follicles toward building a stiffer, more scar-like shell and can activate cellular systems that reinforce this change. In the context of PCOS, where high androgen levels and increased ovarian fibrosis frequently occur together, these findings support the idea that hormones are actively reshaping the ovary’s physical landscape, not just altering hormone levels in the blood. A stiffer ovarian environment may make it harder for follicles to develop normally and to release eggs, helping to explain some of the fertility problems seen in PCOS. Understanding these tissue-level changes could open new avenues for therapies aimed at softening the ovarian matrix or modulating mechanical signalling, complementing treatments that focus solely on hormone levels.
Citation: Hopkins, T.I.R., Lerner, A., Dunlop, I.E. et al. Androgen dysregulates the follicular extracellular matrix and increases pro-fibrotic gene expression in the mouse ovary. Sci Rep 16, 8974 (2026). https://doi.org/10.1038/s41598-025-32927-6
Keywords: polycystic ovary syndrome, androgens, ovarian fibrosis, extracellular matrix, follicle development