Clear Sky Science · en
Profiling the epigenomic landscape of late embryonic and adult mouse hind limb muscles
Why muscle wiring matters for everyday movement
Every step you take, every jump, and even your posture depend on a finely tuned mix of “fast” and “slow” muscle fibers. Fast fibers deliver bursts of power but tire quickly; slow fibers are built for endurance. This study asks a deceptively simple question: what stretches of DNA act as on–off switches that help sculpt this mix of fiber types as muscles develop, and how do these switches differ between young and adult muscles? By mapping these control regions in mouse hind limb muscles, the researchers open a window onto how muscles develop, adapt, and potentially evolve across species.
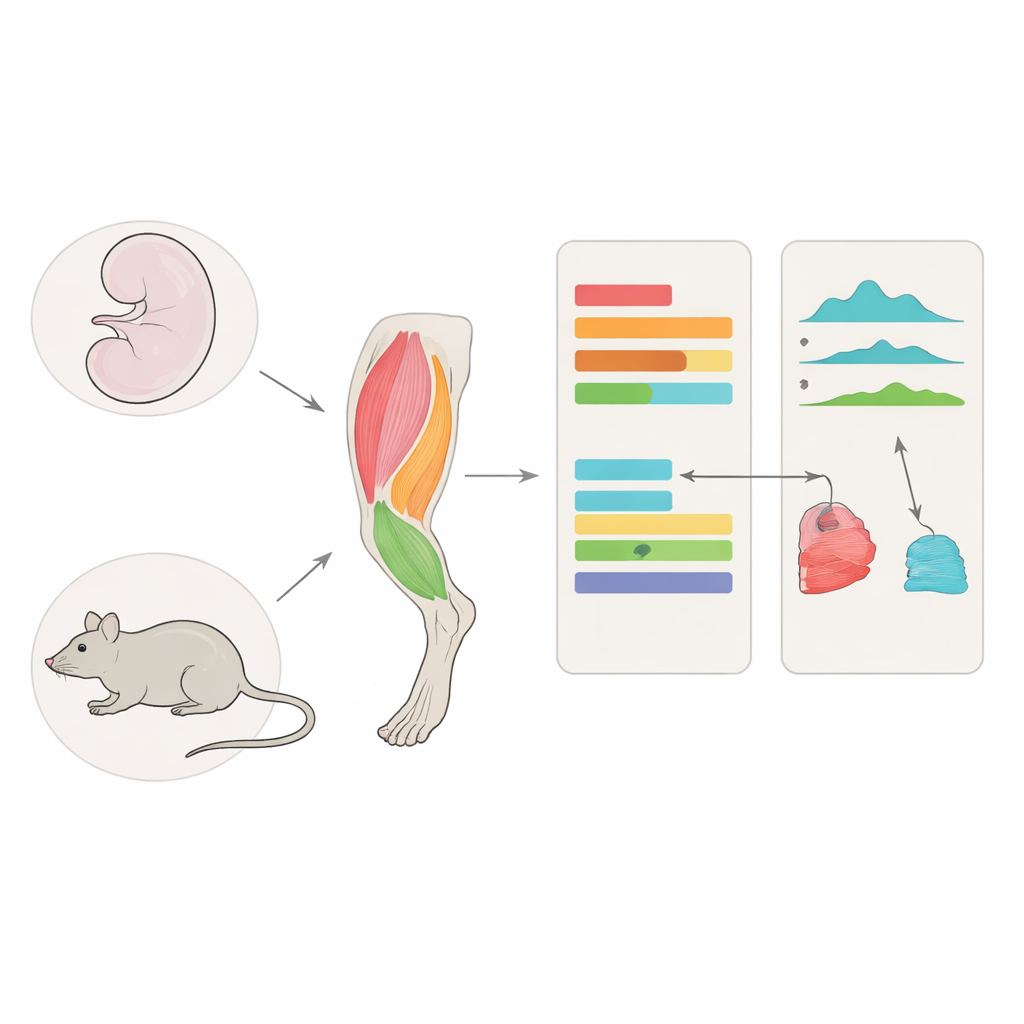
Looking inside power and endurance muscles
The team focused on four muscles in the mouse hind limb: two in the calf and two in the thigh. Each pair contained one muscle rich in slow, fatigue‑resistant fibers and one dominated by fast, powerful fibers. They examined these muscles at a late embryonic stage, just before birth, and again in adulthood. Using two genome‑wide approaches, they measured which genes were active and which parts of the DNA were physically open and accessible in the cell’s nucleus. Open regions often mark hidden control switches known as cis‑regulatory elements, which fine‑tune when and where nearby genes turn on.
From limb blueprint to working engine
In embryonic muscles, the main differences in gene activity reflected the basic patterning of the limb rather than mature muscle performance. Calf and thigh muscles still looked similar in terms of fiber type, but they differed in genes that help set up front‑to‑back and top‑to‑bottom limb segments. Key developmental muscle proteins were present, including early myosin forms that appear before birth, yet the classic markers that separate fast from slow adult fibers were relatively subdued. This suggests that, late in gestation, these muscles are still in a blueprint phase, defining where muscles go rather than how they will ultimately function.
Adult muscles reveal the split between speed and stamina
In adult mice, the picture changed dramatically. Now, gene activity split clearly between fast‑biased and slow‑biased muscles. Fast‑biased muscles showed strong activity of genes linked to rapid contraction and sugar‑burning pathways, features that support quick, powerful movements. Slow‑biased muscles instead favored genes involved in fat‑burning, mitochondrial function, and the slow myosin forms that underpin endurance. When the team compared DNA accessibility, they found many open regions near these fiber‑type‑specific genes, particularly in segments of the genome located far from gene starting points. These distant regions are prime candidates for muscle‑specific switches that shape how fast or slow a muscle behaves.
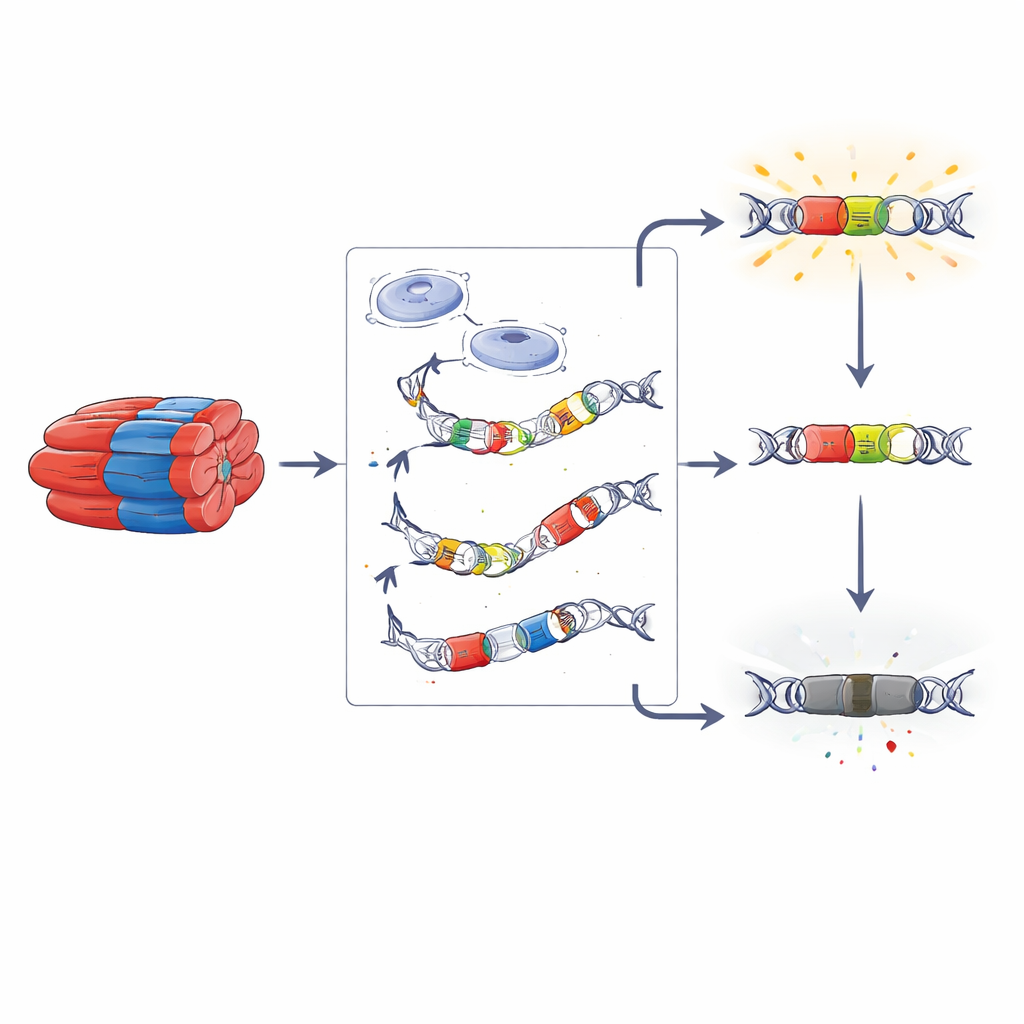
Finding DNA switches that tune muscle identity
To isolate the most muscle‑focused switches, the researchers removed open DNA regions that are also active in brain tissue, which are more likely to control general cell functions. What remained was a set of muscle‑biased control regions that differed by developmental stage and fiber bias. Some regions were shared across all muscles and ages and were more evolutionarily conserved, hinting at long‑standing roles in basic muscle identity. Others were unique to adult fast or slow muscles and showed less conservation, suggesting they may have changed more rapidly over mammalian evolution and could underlie species differences in fiber composition, such as the slow‑heavy leg muscles of humans versus the fast‑heavy muscles of many small mammals.
Testing switches that boost or dampen gene activity
The team then took a closer look at a small set of these candidate control regions that lie near genes known to influence fast or slow fiber traits. They selected twelve DNA segments and inserted each one into a simple reporter system in cultured mouse muscle cells, where the segment could either boost or suppress a light‑producing gene. Nine of these segments increased light output, acting like enhancers, while three reduced it, behaving more like silencers. Importantly, these active switches were tied to either fast‑biased or slow‑biased muscles in the original tissue, suggesting they may help push developing fibers toward power or endurance profiles.
What this means for muscles, health, and evolution
By charting when and where muscle control regions open during development and adulthood, this work shows that the genetic wiring for limb placement emerges early, while the wiring for fast versus slow performance is refined later. The discovery of conserved, muscle‑specific switches that can turn genes up or down in cells provides a starting map for understanding how everyday traits like strength and stamina are programmed in the genome. In the long term, these switches could help explain why different species—and even different people—have distinct muscle profiles, and they may one day offer targets for improving muscle function in disease, aging, or athletic training.
Citation: Queeno, S.R., Okamoto, A.S., Callahan, D.M. et al. Profiling the epigenomic landscape of late embryonic and adult mouse hind limb muscles. Sci Rep 16, 8658 (2026). https://doi.org/10.1038/s41598-025-32705-4
Keywords: skeletal muscle development, fast and slow muscle fibers, gene regulation, enhancers and silencers, mouse hind limb