Clear Sky Science · en
Microbially mediated carbon utilization by a cold-water coral inhabiting methane seeps
Life in the Deep, Fed by Invisible Food
Far below the reach of sunlight, on the dark seafloor off Costa Rica, scientists have discovered a coral that seems to bend the usual rules of marine life. Instead of relying only on tiny bits of dead plankton drifting down from the surface, this coral taps into a hidden food source created by bacteria that feed on methane and sulfur-rich fluids seeping from the seabed. Understanding how this coral makes a living in such an extreme place helps reveal how deep-sea ecosystems work and why they may be especially vulnerable to human disturbance.
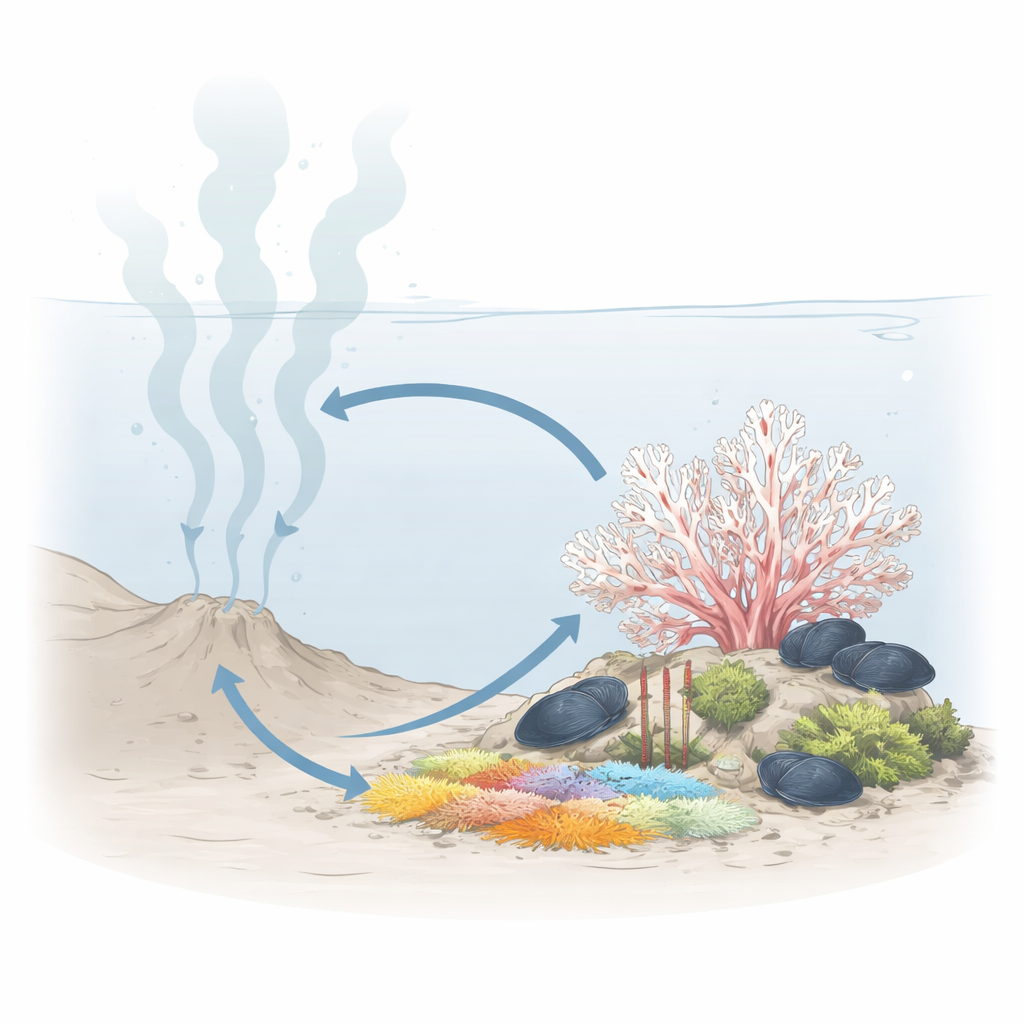
A Strange Home on a Leaking Seafloor
Methane seeps are places where gases and fluids rich in methane and hydrogen sulfide leak from deep within the Earth into the ocean. These spots host bustling communities of worms, mussels, clams, and bacterial mats that thrive without sunlight, powered instead by chemical energy. The newly studied cold-water coral, Swiftia sahlingi, lives on a mound called Mound 12 about a kilometer beneath the Pacific off Costa Rica. Rather than staying safely on the edges of this chemically harsh habitat, the coral is often found right among the classic seep dwellers, even growing on top of large tubeworms and mussel beds.
Mapping Where the Coral Chooses to Live
To understand this coral’s preferred neighborhood, the team used an autonomous underwater vehicle equipped with cameras and sonar to survey the seafloor. From tens of thousands of images, they mapped more than three thousand square meters where these corals occurred. Most colonies—about four out of five—were located in zones with clear signs of active seepage, such as mussels, tubeworms, or white bacterial mats. Habitat models showed that the presence of hard carbonate rock was essential as an anchor point, but closeness to seep animals, especially mussels, also strongly predicted where the coral was likely to thrive. This pattern suggests the coral is not just using old, inactive rocks; it is deliberately settling in areas where chemical seepage is ongoing.
Following the Trail of Carbon
Because we cannot watch corals eat in the deep sea, the researchers turned to chemical clues in their tissues. They measured the natural ratios of carbon and nitrogen isotopes in the coral and compared them with other nearby corals that do not live directly in seep zones. The seep-associated Swiftia sahlingi had much lighter carbon signatures, consistent with food ultimately derived from bacteria that oxidize methane or sulfide, rather than solely from surface-derived plankton. To probe this further, they collected live colonies and incubated them in seawater enriched with methane labeled with a rare form of carbon. After a week, one coral from an active seep area showed a dramatic shift in its carbon signature, clear evidence that methane-derived carbon, processed by microbes, had been incorporated into the coral’s own biomass.
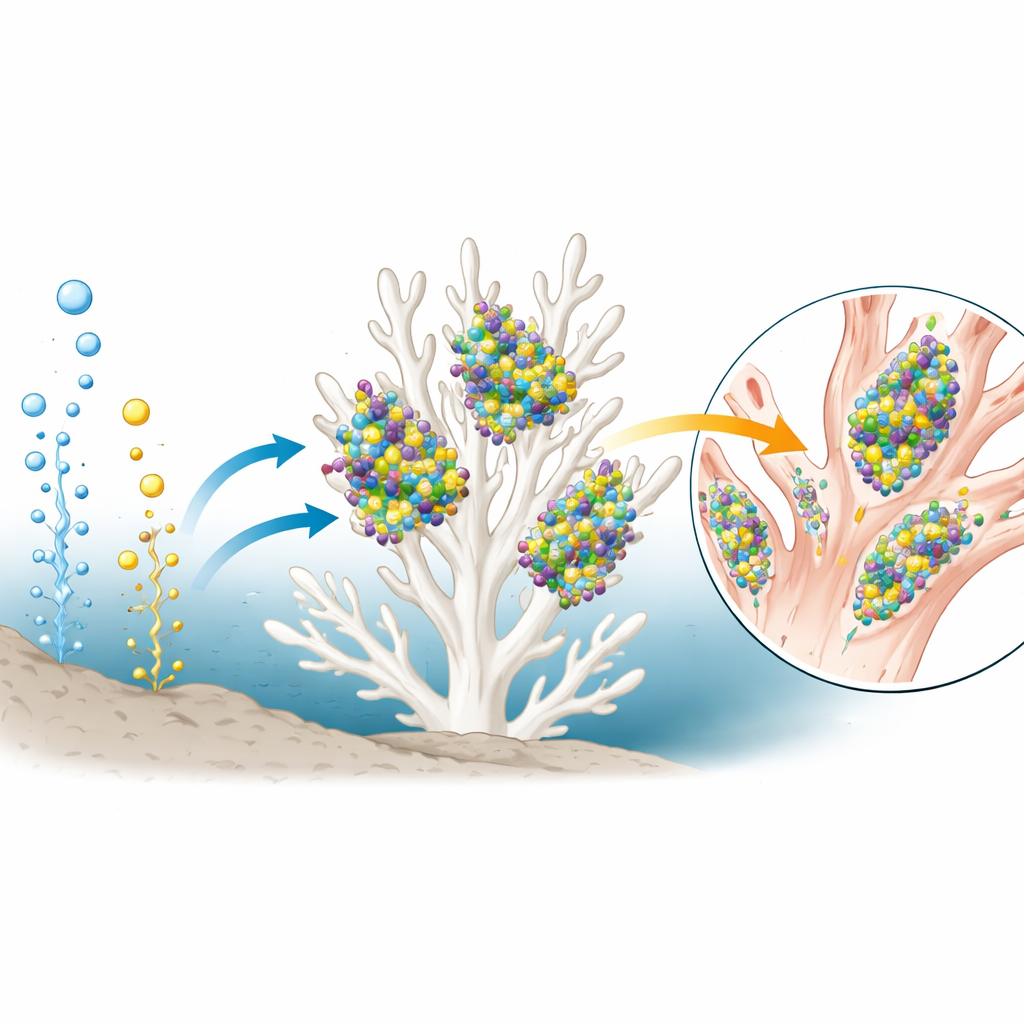
Hidden Partners: Bacteria Living with the Coral
The team also examined the coral’s microbial community by sequencing bacterial DNA from its tissues. They found that sulfur-oxidizing bacteria dominated, sometimes making up more than 90 percent of all detected bacteria. Many belonged to groups already known to power other deep-sea animals at vents and seeps. In two coral colonies, bacteria that oxidize methane were also abundant. Notably, the coral that had taken up the labeled methane in the incubation experiment hosted the highest share of these methane-eating microbes. The mix of sulfur- and methane-using bacteria, and their varying abundance from coral to coral, suggests a flexible partnership in which microbes can help feed their host in multiple ways.
Why This Flexible Lifestyle Matters
Taken together, the mapping, isotope measurements, experiments, and DNA analyses point to Swiftia sahlingi as a mix-feeding coral that draws energy from both falling surface food and from bacteria that turn seep chemicals into usable carbon. This flexible strategy may allow the coral to colonize areas once thought too extreme, expanding where cold-water coral habitats can form and supporting more deep-sea biodiversity. At the same time, it highlights how dependent these ecosystems are on fragile chemical and microbial processes that could be disrupted by activities such as deep-sea mining, trawling, or drilling. Recognizing these hidden partnerships is a key step toward protecting the deep sea’s little-known, but highly interconnected, web of life.
Citation: Stabbins, A., Goffredi, S., Gasbarro, R. et al. Microbially mediated carbon utilization by a cold-water coral inhabiting methane seeps. Sci Rep 16, 9603 (2026). https://doi.org/10.1038/s41598-025-32153-0
Keywords: methane seeps, cold-water corals, chemosynthetic microbes, deep-sea ecosystems, symbiosis