Clear Sky Science · en
In vivo visualization of cardiac extracellular adenosine dynamics and its pharmacological modulation in zebrafish heart failure models
Why this fish heart story matters
Heart failure remains one of the leading causes of death worldwide, and many patients still get worse despite today’s best drugs. Scientists know that stressed heart cells release chemical “ distress signals ” into their surroundings, but it has been hard to watch these signals in action inside a beating heart. This study uses transparent zebrafish, whose tiny hearts closely mimic key features of human hearts, to watch those danger signals in real time and test a new way to quiet them down.
Signals from a struggling heart
When heart cells are injured, they release energy-carrying molecules such as ATP and its breakdown product adenosine into the space outside the cells. Outside the cell, these same molecules stop acting as fuel and instead behave like urgent messages that can fan the flames of inflammation. In chronic heart failure, that inflammatory response can become overactive, damaging the heart further. Until now, however, researchers lacked direct, in-the-body evidence of how these molecules behave over time in a failing heart.
Turning zebrafish into living chemical cameras
The team engineered zebrafish whose heart cells make a special fluorescent sensor protein called GRABAdo on their surface. When adenosine builds up outside the cells, this sensor glows brighter green, effectively turning each tiny heart into its own live chemical camera. The researchers then triggered heart failure in zebrafish using terfenadine, a drug known to disrupt heart rhythm and weaken pumping. As the fish developed clear signs of heart failure—slower heartbeats, enlarged and weaker ventricles, swelling around the heart, and poorer swimming performance—their heart sensors lit up, revealing a marked rise in extracellular adenosine. Chemical measurements of whole larvae confirmed that total adenosine levels were higher in the failing hearts.
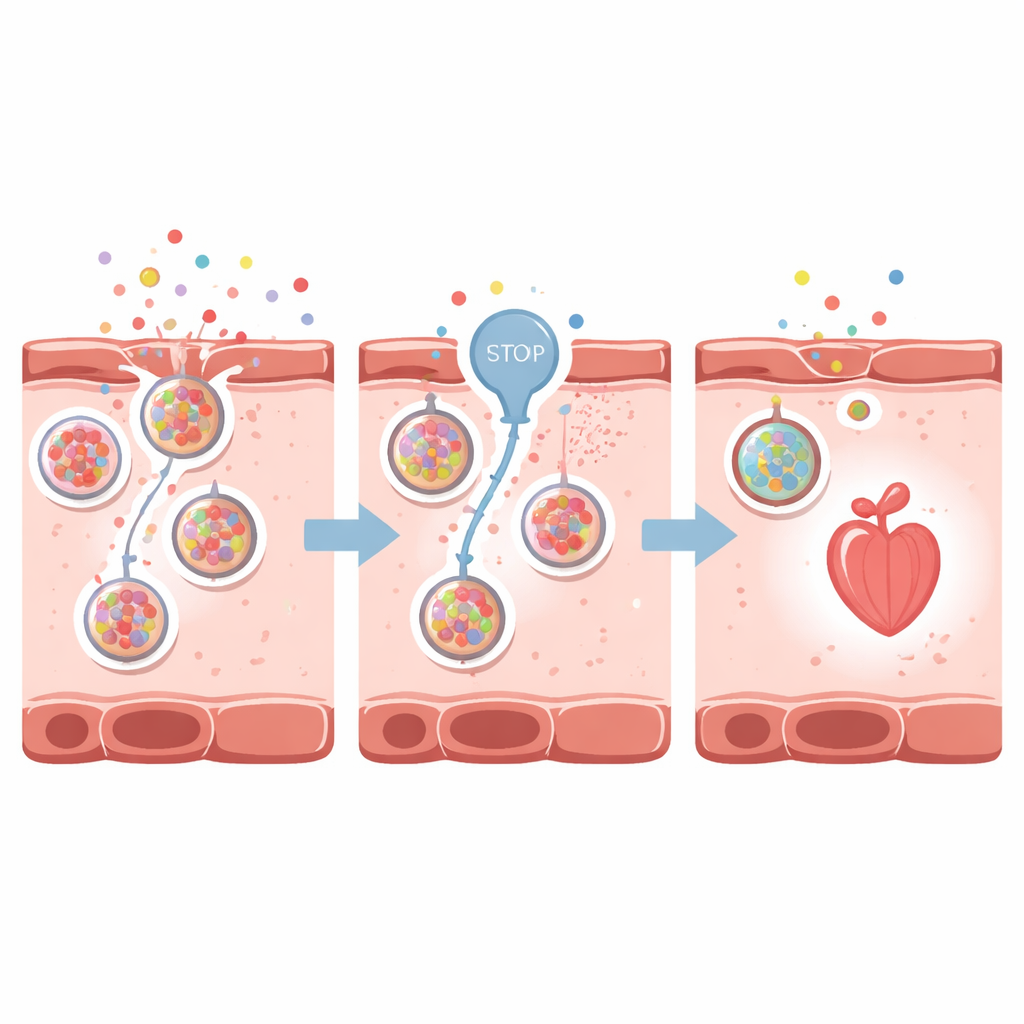
Blocking the release valve
The scientists next asked whether dialing down these danger signals could protect the heart. They focused on VNUT, a transporter that loads ATP into small cellular packets (vesicles) for release outside the cell. Using clodronate, a drug that blocks VNUT, they reduced ATP release and, indirectly, adenosine buildup around heart cells. In zebrafish with drug-induced heart failure, VNUT inhibition preserved heart shape, reduced chamber enlargement, improved heart rate and pumping strength, and maintained better blood flow. The glowing adenosine sensor dimmed, showing that less of the distress signal was present outside the cells. Similar protective effects appeared in adult zebrafish with an acute, rapidly developing form of heart failure.
Multiple protective levers working together
Heart failure involves more than one pathway, so the researchers tested how VNUT blockade interacts with other known players. Drugs that block certain adenosine receptors and ATP receptors, as well as a compound that limits passive ATP leakage through membrane channels, each helped the zebrafish hearts. When combined with VNUT inhibition, many of these treatments produced additive benefits, suggesting that turning down purine signaling at several points can be especially powerful. VNUT inhibition also helped restore calcium balance in heart cells, which is essential for normal beating, and reduced both cell death and the activity of genes linked to inflammation. Together, these results paint VNUT as a central switch that links ATP release, inflammation, disturbed calcium handling, and progressive heart damage.
From fish tanks to future therapies
For non-specialists, the key message is that the study directly visualizes a chemical stress signal rising in failing hearts and shows that blocking its release can protect heart function in a living animal. While zebrafish hearts are simpler than human hearts, they share enough biology to offer a powerful testbed for new ideas. By revealing VNUT as a promising target at the crossroads of inflammation and electrical instability, this work suggests that future heart failure therapies may do more than just support blood flow—they may actively calm harmful cellular conversations that push the heart toward failure.
Citation: Phurpa, P., Apolinario, M.E.C., Umeda, R. et al. In vivo visualization of cardiac extracellular adenosine dynamics and its pharmacological modulation in zebrafish heart failure models. Sci Rep 16, 8220 (2026). https://doi.org/10.1038/s41598-025-30303-y
Keywords: heart failure, adenosine signaling, zebrafish model, inflammation, cardiac protection