Clear Sky Science · en
Adsorption of heavy metal ions and organic pollutants from refining wastewater by magnetically synthesized silver nanoparticles coated with graphene oxide
Why Dirty Water from Oil Refineries Matters
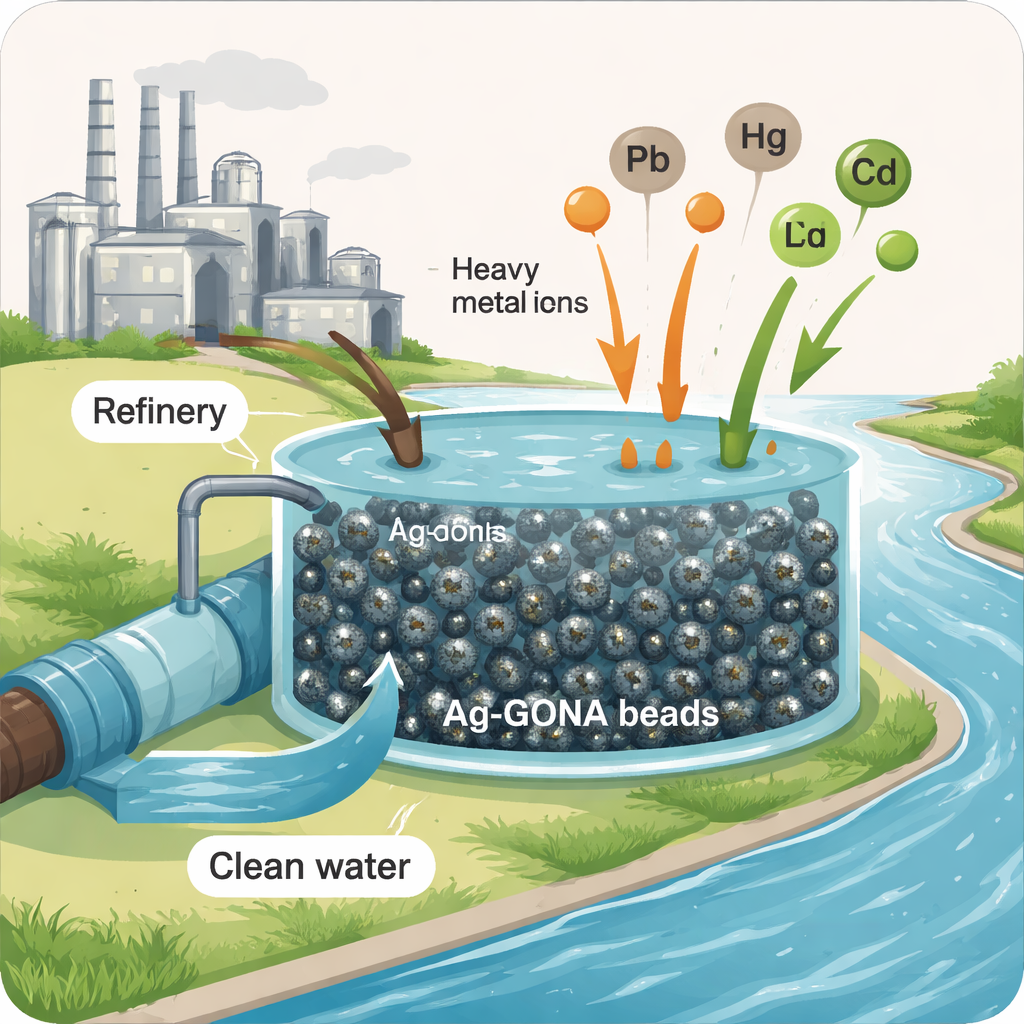
Water used in oil refineries often comes out laced with toxic metals and stubborn organic chemicals that are hard to remove and dangerous even at low levels. These pollutants can build up in fish, seep into drinking water sources, and damage ecosystems for years. This study explores a new type of tiny, reusable cleaning beads that can pull many of these dangerous substances out of wastewater at once, offering a practical tool for cleaner rivers, safer drinking water, and more sustainable industry.
Tiny Beads Built from Everyday Ingredients
The researchers created small, squishy beads by combining three main ingredients: a plant-based gel called alginate, ultra-thin sheets of carbon known as graphene oxide, and very small particles of silver. The silver nanoparticles were made in a "green" way using baker’s yeast, a common by-product of brewing. The yeast acts as a natural factory that helps turn dissolved silver into stable, nanosized particles. These particles, together with the graphene sheets, were then trapped inside the alginate gel to form dark, porous beads called Ag-GONA beads. Because the beads are magnetically responsive and relatively easy to handle, they can be mixed into dirty water and then separated again for reuse.
How the Beads Grab Metals and Chemicals
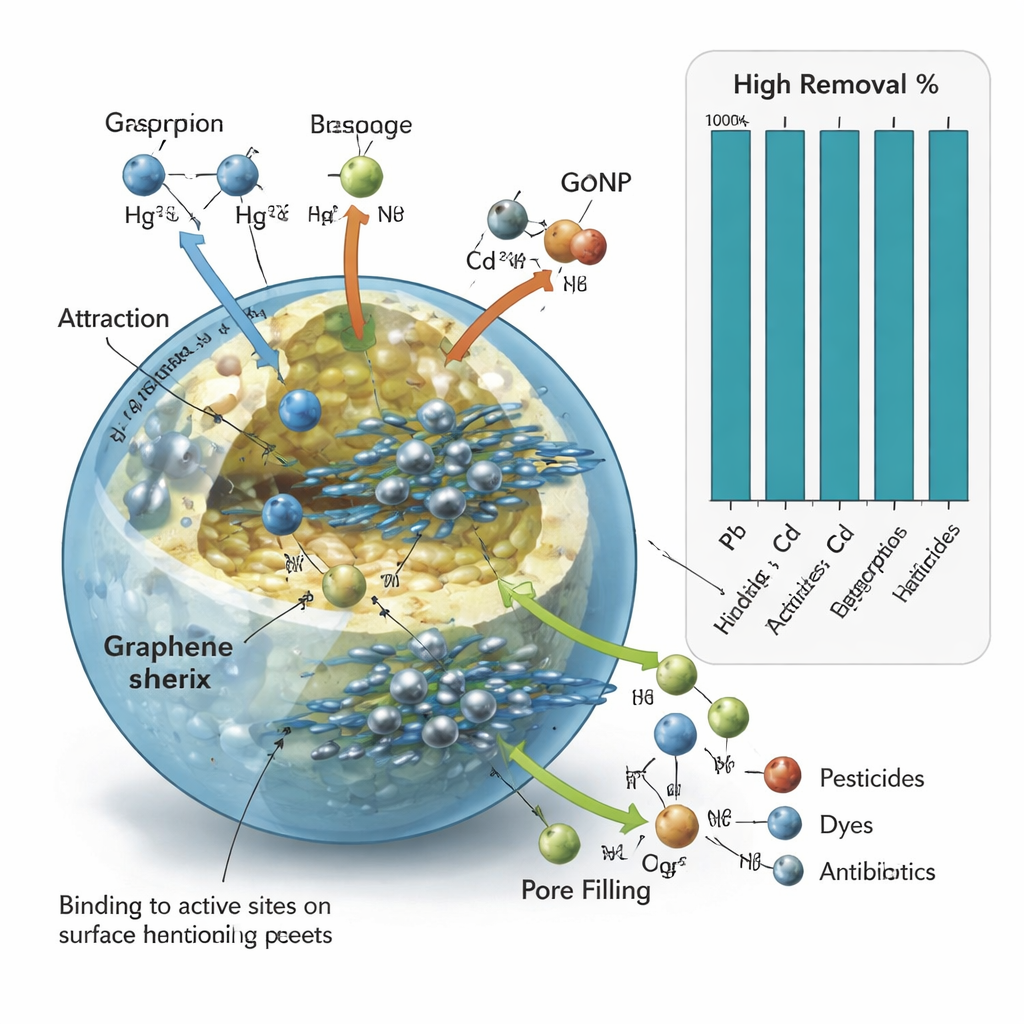
Under the microscope, the beads look like a sponge with many nooks and crannies and a rough surface coated with chemical groups that can latch onto pollutants. Tests showed that these beads are especially good at capturing three highly toxic metals—lead, mercury, and cadmium—as well as three typical oil-related organic compounds: naphthalene, phenol, and fluorene. The team found that the process worked best in water close to neutral pH (around 7), at room temperature, and after about six hours of contact. Under these conditions, the beads could hold very large amounts of metals, far exceeding many earlier materials tested for similar purposes. The pollutants stick to the beads through a mix of forces: electrical attraction, hydrogen bonding, and simple filling of pores in the material.
Finding the Sweet Spot for Cleaning Performance
The researchers carefully varied acidity, temperature, contact time, and pollutant concentration to understand how the beads perform under different conditions. At low pH (more acidic), hydrogen ions in the water compete with metal ions and organic molecules for the same binding spots, so the beads remove less pollution. As the pH approaches neutral, the bead surface becomes more welcoming to metals and organics, and removal rates surge, often above 90 percent. Higher temperatures, however, slightly reduce performance, suggesting that cooler, near-ambient conditions are better. At very high pollutant levels, the beads begin to fill up and remove a somewhat smaller fraction of contaminants, but they still capture substantial amounts overall, showing strong potential for treating real refinery streams.
Built to Be Used Again and Again
For any treatment material to be practical, it must be reusable. The team tested this by cycling the same batch of beads through multiple rounds of pollution removal and cleaning. After each use, the beads were washed with a mild acidic solution containing calcium salts to knock off the trapped pollutants and restore their structure. Over six cycles, lead removal fell from nearly complete removal to around four-fifths, while mercury, cadmium, and the organic compounds showed only modest declines. This means the beads can clean water repeatedly without falling apart, lowering both cost and waste compared with single-use materials.
What This Means for Cleaner Water
In plain terms, the study shows that a thoughtfully designed mix of everyday biopolymers, carbon sheets, and tiny silver particles can form beads that act like powerful, reusable sponges for some of the most worrisome pollutants in refinery wastewater. They work best in mildly treated water conditions that are realistic for industrial plants, and they can be regenerated several times with only a modest loss of performance. If scaled up, this approach could become part of an efficient, more environmentally friendly toolkit for keeping toxic metals and persistent organic chemicals out of natural waters and, ultimately, away from people and wildlife.
Citation: Syed, S.S., Jacob, L., Banat, F. et al. Adsorption of heavy metal ions and organic pollutants from refining wastewater by magnetically synthesized silver nanoparticles coated with graphene oxide. Sci Rep 16, 7681 (2026). https://doi.org/10.1038/s41598-025-26709-3
Keywords: wastewater treatment, heavy metal removal, nanomaterial adsorbent, refinery pollution, graphene composite beads