Clear Sky Science · en
Hepatic transcriptomic and functional responses to ketogenic diet intervention in MASLD-Induced male albino rats
Why this matters for everyday health
Many people worried about fatty liver and weight loss experiment with low-carb or ketogenic diets, often without knowing exactly what these diets do to the liver. This study used a controlled experiment in rats to explore how a ketogenic diet affects a common modern liver condition now called metabolic dysfunction–associated steatotic liver disease (MASLD), a fatty liver disease linked to obesity and diabetes. By looking not only at blood fats and organ function but also at changes in liver genes, the work offers a peek under the hood at how this popular diet might help—or in some cases complicate—liver health.
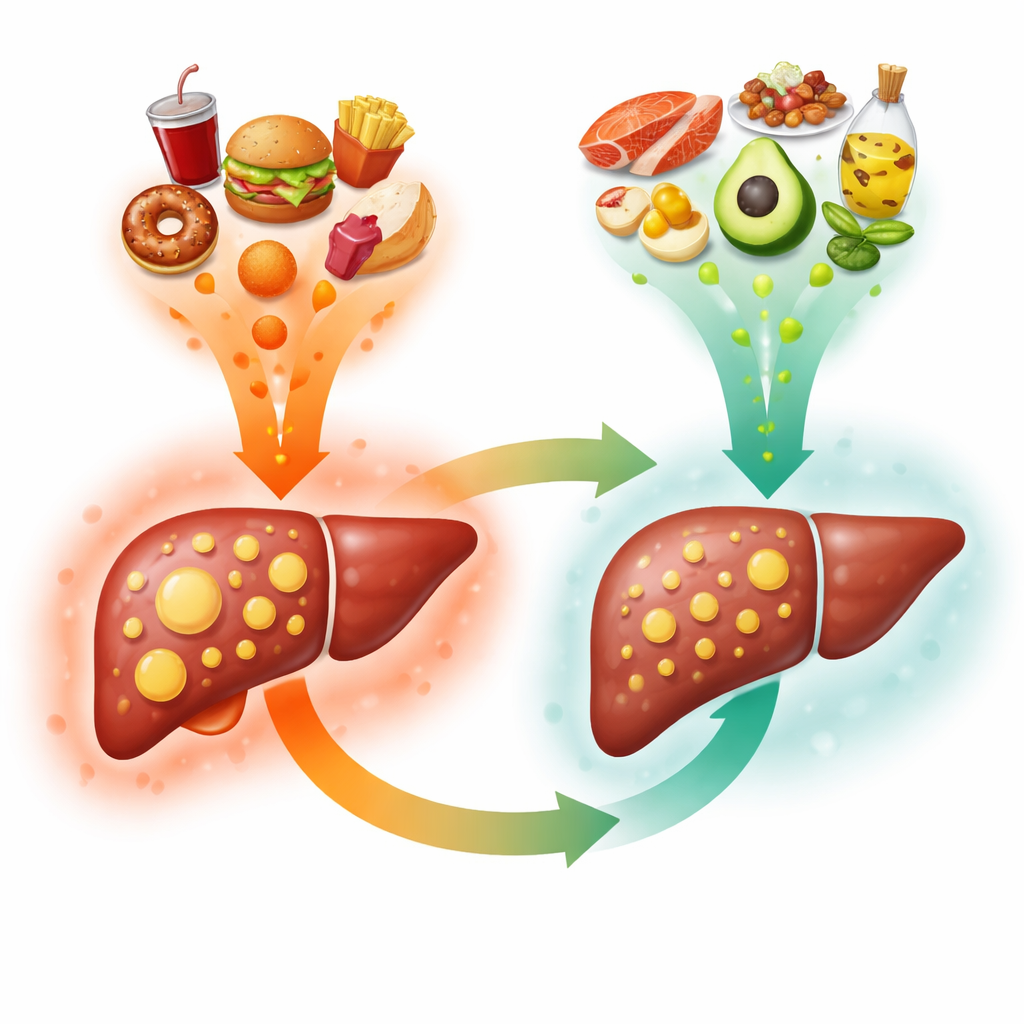
From sugary diets to fatty livers
MASLD has become the most widespread chronic liver problem worldwide, tightly tied to sedentary lifestyles and calorie-dense diets. In its early stages, fat quietly accumulates inside liver cells; over time this can trigger inflammation, scarring, and even liver cancer. Because there is no approved drug treatment, doctors often recommend lifestyle changes, but there is debate over which diet is best. The ketogenic diet, which is very low in carbohydrates and high in fat, has gained attention for its ability to lower liver fat and body weight in some small human studies. However, its deeper effects on liver biology and long-term safety remain uncertain.
How the experiment was set up
To probe these questions, the researchers studied forty male rats divided into four groups. One group ate a standard diet. A second group was fed a high-fructose, high-fat regimen designed to mimic a sugary, ultra-processed human diet and to induce MASLD. A third group received only the ketogenic diet, composed of about two-thirds fat, one-quarter protein, and very little carbohydrate, for 16 weeks. The fourth group first developed MASLD on the high-sugar, high-fat diet and then was switched to the ketogenic diet. The team monitored blood markers related to body fat hormones, kidney function, and cholesterol, examined liver tissue under the microscope, and measured the activity of many genes that control fat handling, inflammation, and cellular stress.
What happened to hormones, blood fats, and organs
Rats on the MASLD-inducing diet developed clear signs of metabolic trouble. They had higher levels of leptin, a hormone linked to body fat and inflammation, and lower amounts of adiponectin, a hormone that usually protects metabolism. Their blood triglycerides and “bad” LDL cholesterol climbed, while “good” HDL cholesterol fell. Kidney waste products such as urea and creatinine also rose, hinting at strain on the kidneys. In contrast, rats fed the ketogenic diet alone showed the healthiest overall profile: leptin stayed lower, adiponectin remained near normal, and kidney and blood fat measures were generally better than in the MASLD group. Animals that were switched from the unhealthy diet to the ketogenic diet landed in between—improved compared with MASLD animals, but not as favorable as rats that had been on the ketogenic plan from the start.
What the liver looked like inside
Microscope studies of liver tissue reinforced the blood findings. Livers from MASLD rats were swollen with fat droplets, showed disorganized structure, ballooned and dying cells, and heavy immune cell infiltration, all classic signs of advancing fatty liver disease. In the ketogenic-only group, some mild disturbances appeared, but the damage was far less pronounced. Most strikingly, rats that developed MASLD and were then switched to the ketogenic diet showed near-normal liver architecture, with many regenerating cells and much less fat and inflammation. Staining for a protein associated with cellular stress and damage, β-amyloid, was intense in MASLD livers but faint in both ketogenic groups, suggesting the diet eased molecular injury.
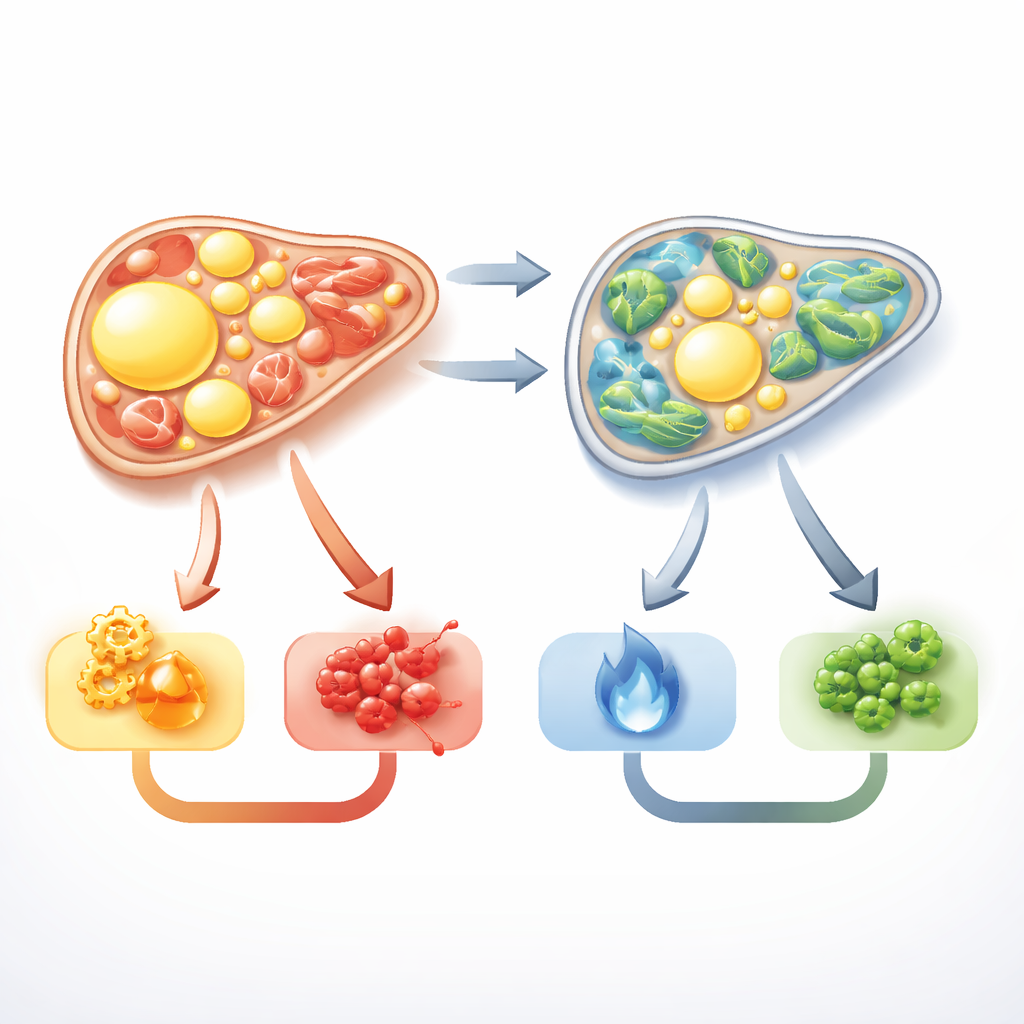
Shifts in genetic switches inside liver cells
Beyond visible damage, the team examined networks of long noncoding RNAs and microRNAs—short genetic switches that help dial pathways up or down. In MASLD rats, genes that drive fat production and uptake were highly active, while genes that promote fat burning and protect against oxidative stress were suppressed. Several regulatory RNAs previously linked to fatty liver, including GAS5, H19, NEAT1, and MALAT1, were strongly disturbed. The ketogenic diet tended to reverse many of these patterns. In MASLD animals switched to the ketogenic diet, markers of fat manufacturing fell, protective pathways such as PPARα, which supports fat breakdown, were restored, and key regulatory RNAs and their partner microRNAs shifted back toward normal. These changes map onto the observed drop in liver fat and inflammation, even though the study did not manipulate individual genes to prove direct cause-and-effect.
What this could mean for people
Put simply, the study suggests that in this rat model a ketogenic diet not only trims liver fat and improves blood measures but also calms harmful genetic programs while reawakening protective ones. For early MASLD, starting or switching to such a diet helped move the liver toward healing, though some kidney markers and cholesterol components changed in complex ways that call for caution. Because the work was done in animals and did not directly test each genetic pathway, it cannot be taken as proof that ketogenic diets are a cure for human fatty liver disease. Still, it offers a mechanistic roadmap hinting that carefully monitored, medically supervised ketogenic eating might one day be part of a broader strategy to protect the liver in people at risk.
Citation: Abdelsalam, H.M., Diab, A., El-Dawy, K. et al. Hepatic transcriptomic and functional responses to ketogenic diet intervention in MASLD-Induced male albino rats. Sci Rep 16, 9272 (2026). https://doi.org/10.1038/s41598-025-26659-w
Keywords: ketogenic diet, fatty liver disease, MASLD, liver metabolism, nonalcoholic steatohepatitis