Clear Sky Science · en
Safety and feasibility of liver maximum capacity assessment in patients undergoing transarterial chemoembolisation for hepatocellular carcinoma
Why this matters for people with liver cancer
Many people with liver cancer already have fragile livers from long‑standing disease. Treating tumors without pushing the liver into failure is a constant balancing act. This study explores whether a quick, bedside breath test called LiMAx can more precisely measure how well the liver is working in patients receiving a common treatment called transarterial chemoembolisation (TACE), potentially helping doctors choose safer, more tailored care.
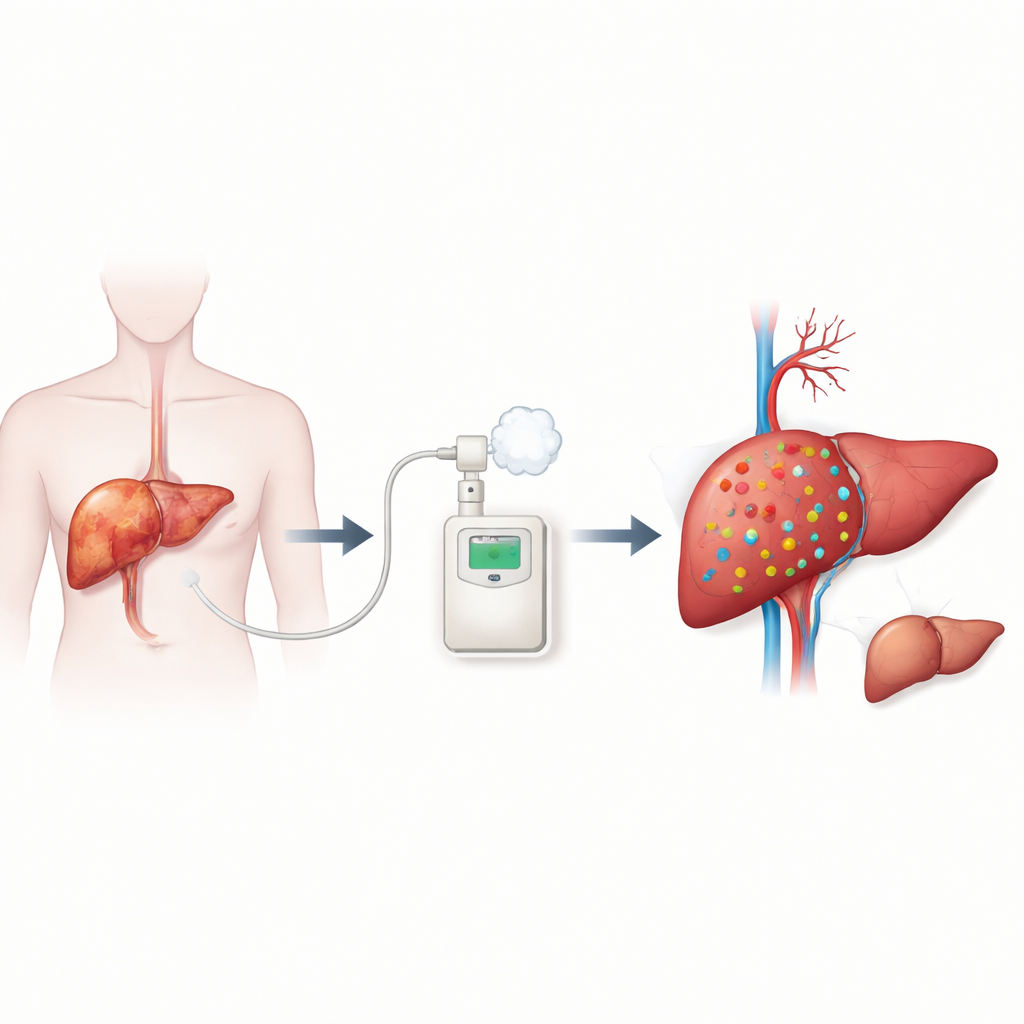
A targeted but stressful liver treatment
TACE is a minimally invasive procedure used for people with liver cancer who are not candidates for surgery. Doctors thread a small tube into the arteries that feed the tumor and deliver chemotherapy together with tiny particles that block the blood supply. This can shrink or control tumors, but it also stresses the rest of the liver, which may already be scarred from cirrhosis or other chronic diseases. If the remaining healthy liver tissue cannot cope, patients can develop liver decompensation, a serious and sometimes irreversible decline in liver function. Current blood‑based scoring systems estimate risk but often struggle to capture how much healthy liver reserve an individual patient truly has.
A breath test to gauge liver strength
The LiMAx test offers a different window into liver health. After a short fast, patients receive a small dose of a harmless compound labeled with a special carbon atom. Only working liver cells can break this compound down, releasing labeled carbon dioxide that appears in the breath. By measuring how quickly this labeled gas is exhaled over an hour, the test estimates the liver’s maximum working capacity in real time. The technique has already been used in Europe to help surgeons decide how much liver can be safely removed during major operations, but it has not been widely studied in patients receiving TACE.
Trying LiMAx before and after treatment
The researchers enrolled 30 people with liver cancer scheduled for TACE at a large UK hospital. Almost all had underlying cirrhosis. Each participant underwent the LiMAx breath test one to two weeks before TACE, again four to six weeks afterward, and once more at around three months. The team also collected standard blood tests and calculated commonly used liver scores such as Child‑Pugh, MELD, UKELD, and ALBI. They then tracked hospital stays, complications, readmissions, and survival for at least a year to see how the breath test related to real‑world outcomes.
What the test revealed about liver function
Baseline LiMAx results varied widely, from severely impaired to normal liver capacity, even among patients grouped in the same traditional score category. In general, lower LiMAx values were linked with worse blood‑based measures of liver function, but there was overlap: some people who looked relatively well on standard scores had markedly poor LiMAx readings, and vice versa. After TACE, the median LiMAx value changed very little overall, suggesting that the procedure did not usually cause a major drop in liver function in this group. A few patients experienced large decreases, while nearly half actually showed improved LiMAx values at the first follow‑up, perhaps reflecting recovery after tumor control or fluctuations in liver inflammation.
Safety and early signals from patient outcomes
The primary question was whether adding LiMAx testing to the care pathway was safe and practical. All 30 patients completed the bedside breath tests without any test‑related side effects, and most returned for at least one follow‑up measurement. Complications from TACE itself were uncommon, and only one person developed clear liver decompensation after the procedure; that individual had a very low LiMAx value before treatment. Overall short‑term survival was high, and in this relatively small sample the researchers could not show a firm link between pre‑procedure LiMAx and survival or complication rates, although poorer outcomes tended to cluster among those with more severely reduced liver capacity.
What this means going forward
For lay readers, the key message is that this simple breath test appears to be a safe and feasible way to measure how strong or fragile the liver really is in people undergoing targeted treatments for liver cancer. While this early study was too small to prove that LiMAx can reliably predict who will tolerate TACE or who is at high risk of liver failure, it shows that the test works in this setting and offers more nuanced information than standard blood scores alone. Larger studies in sicker patients are now needed to determine whether LiMAx‑guided decisions can help match each person to the right treatment, maximising tumor control while protecting the remaining healthy liver tissue.
Citation: Clarke, G., Dhanaraj, S., Hull, D. et al. Safety and feasibility of liver maximum capacity assessment in patients undergoing transarterial chemoembolisation for hepatocellular carcinoma. Sci Rep 16, 8014 (2026). https://doi.org/10.1038/s41598-025-23968-y
Keywords: liver function testing, hepatocellular carcinoma, chemoembolisation, LiMAx breath test, cirrhosis