Clear Sky Science · en
Identification and evaluation of tumor pyroptosis-associated antigens for design a vaccine candidate against lung cancer
Why this research matters for people with lung cancer
Lung cancer remains the deadliest cancer worldwide, and current treatments—surgery, chemotherapy, radiation, and immunotherapy—still leave many patients without long-term options. This study explores a very different idea: a therapeutic vaccine designed not to prevent infection, but to train the immune system to better recognize and attack lung tumors. Using advanced computer modeling, the authors build and test, entirely in silico, a protein-based vaccine that targets molecules linked to a fiery form of tumor cell death called pyroptosis, opening a new avenue for more precise and durable lung cancer treatment.
Turning a cell death switch into a cancer target
Instead of focusing on classic cancer markers, the researchers chose four proteins—CARD8, NAIP, NLRP1, and NLRP3—that are involved in pyroptosis, an explosive type of programmed cell death that can alert the immune system. These proteins are active in immune regulation and have been associated with better survival in lung adenocarcinoma, making them attractive targets. The team reasoned that if the immune system could be precisely trained to recognize small segments of these proteins on tumor cells, it might not only help destroy cancerous cells directly but also amplify broader immune responses against the tumor.
Building a custom vaccine piece by piece
Using immunoinformatics tools, the authors scanned the four proteins to identify short stretches, or epitopes, predicted to be seen by both T cells and B cells. From hundreds of candidates, they selected 15 epitopes that scored highly for being recognizable by immune cells, while also being predicted as non-toxic and non-allergenic. These pieces were then linked together into a single long protein, joined by carefully chosen molecular "spacers" so that each epitope would remain accessible. To boost the immune response, they added three known immune-stimulating components from bacterial proteins as adjuvants. The resulting construct, 678 building blocks (amino acids) long, was predicted to be stable, soluble, and strongly antigenic—properties that are important for a real-world vaccine.
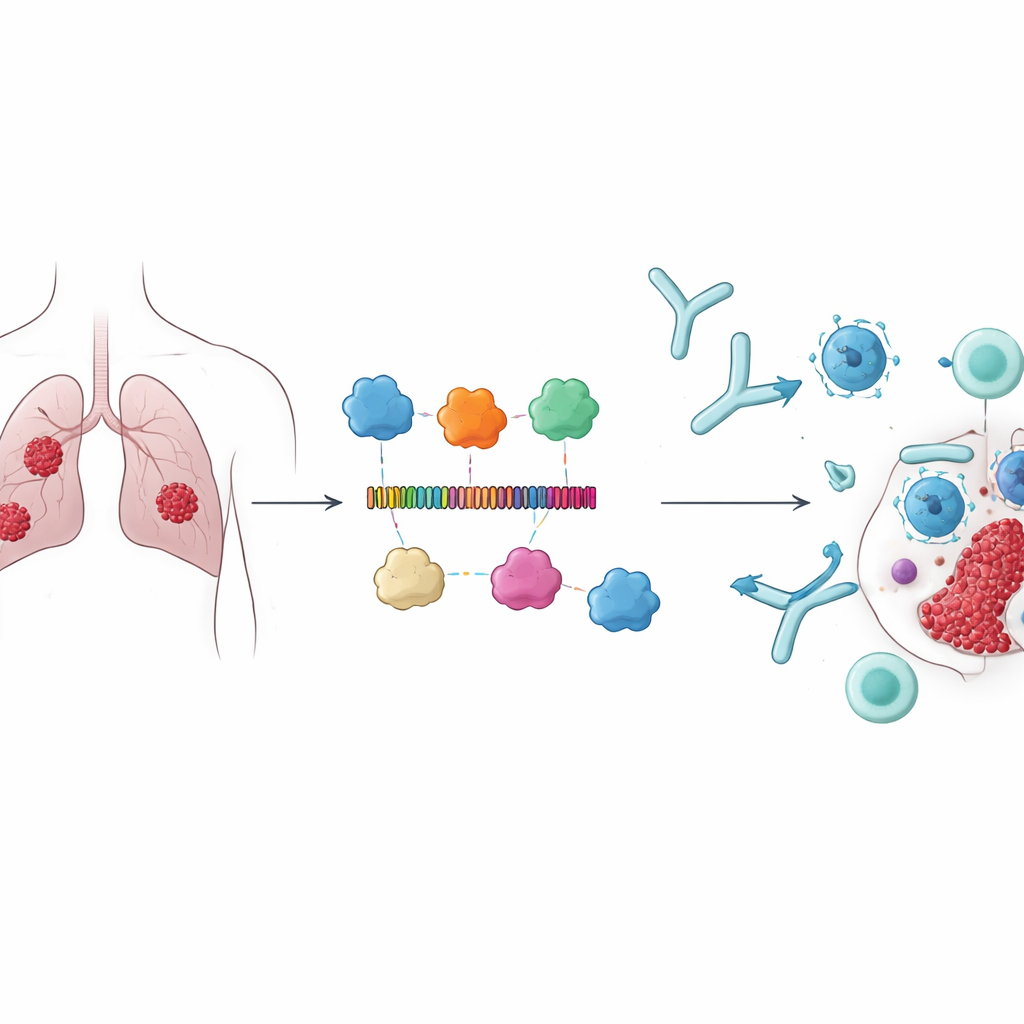
Testing the shape and strength of the design on a computer
Because a vaccine’s effectiveness depends heavily on its three-dimensional shape, the team built 3D models of the new protein using several state-of-the-art structure prediction programs. They repeatedly refined these models and checked their quality with standard structural benchmarks, settling on a final version that met strict criteria for realistic protein geometry and stability. They also mapped which surface regions would be easiest for antibodies to latch onto and engineered new internal “bridges” called disulfide bonds, predicted to further stiffen and stabilize the protein. To mimic how the vaccine would behave in the watery environment of the body, they ran detailed molecular dynamics simulations lasting 100 billionths of a second across ten independent runs. Throughout these simulations, the model protein remained compact and structurally steady, suggesting it would hold its intended form in real conditions.
Simulating how the immune system responds
The researchers next asked whether their vaccine could, in principle, talk to the immune system’s key “alarm” sensors—Toll-like receptors (TLRs) that sit on or within immune cells. Computer docking experiments showed strong, stable interactions between the vaccine and six different human TLRs, especially TLR5 and TLR8, which are known to kick off powerful immune cascades. They then simulated an entire vaccination schedule using immune system modeling software. In these virtual experiments, three doses of the vaccine led to rapid clearance of the simulated antigen, strong waves of antibodies (particularly IgM and IgG1), and a rise in long-lived memory B and T helper cells. Important signaling molecules such as interferon-gamma and interleukins also surged, indicating activation of both innate and adaptive arms of immunity—exactly the kind of balanced response desired for anti-tumor protection.
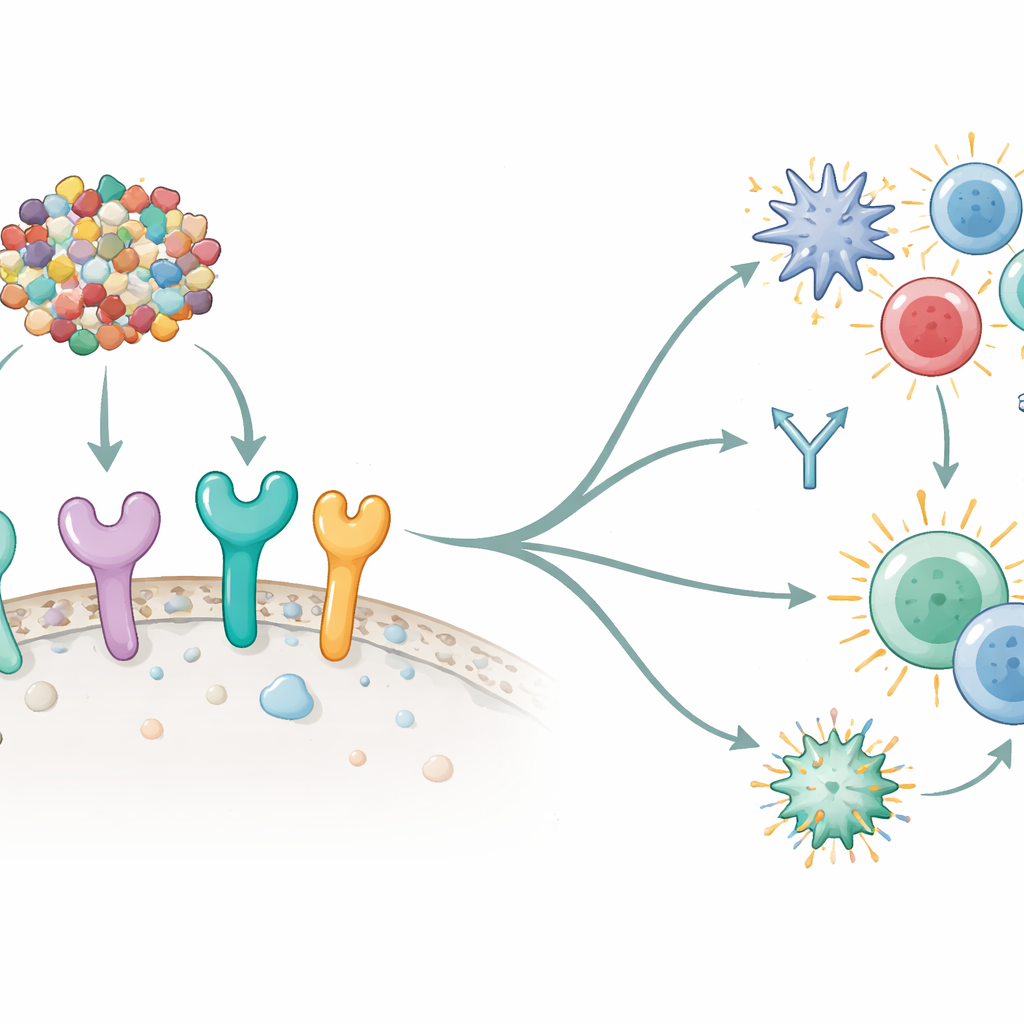
Preparing for real-world production
To move from concept to laboratory testing, the team checked whether the vaccine could be efficiently produced in a common bacterial workhorse, Escherichia coli. By adjusting the genetic code for the vaccine without changing the protein itself, they achieved an ideal pattern for bacterial protein production and a balanced chemical composition of the DNA. They then carried out a virtual cloning step, inserting the optimized gene into a standard laboratory plasmid used for protein expression. These steps suggest that, if taken into the lab, the vaccine could likely be manufactured at scale for experimental studies.
What this could mean for future cancer care
In plain terms, this work delivers a thoroughly computer-tested blueprint for a lung cancer vaccine that targets proteins tied to a particularly inflammatory form of tumor cell death. The design appears safe, stable, and capable of strongly engaging immune defenses in simulations, with broad predicted coverage across human populations. While it remains a digital prototype and must still be validated in cells, animals, and ultimately people, the study illustrates how modern computational tools can compress years of trial-and-error into a single integrated design. If future experiments bear out these predictions, such a vaccine could one day complement existing treatments, helping patients’ own immune systems more reliably recognize and attack lung tumors.
Citation: Nguyen, T.L., Kim, H. Identification and evaluation of tumor pyroptosis-associated antigens for design a vaccine candidate against lung cancer. Sci Rep 16, 9559 (2026). https://doi.org/10.1038/s41598-024-84792-4
Keywords: lung cancer vaccine, pyroptosis, multi-epitope immunotherapy, computational vaccine design, tumor immune response