Clear Sky Science · en
A full-length mtDNA dataset for studying genetic variations across generations and complex family structures
Following Family Lines Through Tiny Powerhouses
Every one of us carries a small ring of DNA inside our cells’ powerhouses, the mitochondria, that comes almost entirely from our mothers. This genetic ring can reveal family history, help solve crimes, and shed light on disease—if we can read it accurately. The study described here delivers a carefully validated collection of complete mitochondrial genomes from real families across several generations, offering a new reference map for researchers who want to track how this special DNA changes as it is passed down.
Why Mitochondrial DNA Matters
Mitochondria act as tiny energy factories in our cells, and they have their own DNA separate from the DNA in the cell nucleus. Because mitochondrial DNA is inherited almost exclusively from the mother and exists in many copies per cell, it has become a key tool in fields as diverse as evolutionary biology, medical genetics, and forensic science. It can survive in damaged or old samples where ordinary DNA fails, and its strict maternal inheritance makes it a natural tracer of family lines and human migrations over time.
The Problem of Genetic Echoes in the Wrong Place
Reading mitochondrial DNA in full is not straightforward. Over evolutionary time, fragments of mitochondrial DNA have been copied and pasted into our nuclear chromosomes. These look very similar to true mitochondrial sequences and are scattered throughout the genome like misleading echoes. When scientists use standard short-read sequencing, these nuclear look-alikes—called NUMTs—can be mistaken for real mitochondrial variants, muddying the picture of which changes genuinely belong to the mitochondrial genome, especially when looking for rare mutations or trying to reconstruct complete maternal lineages.

A New Way to Read the Whole Ring at Once
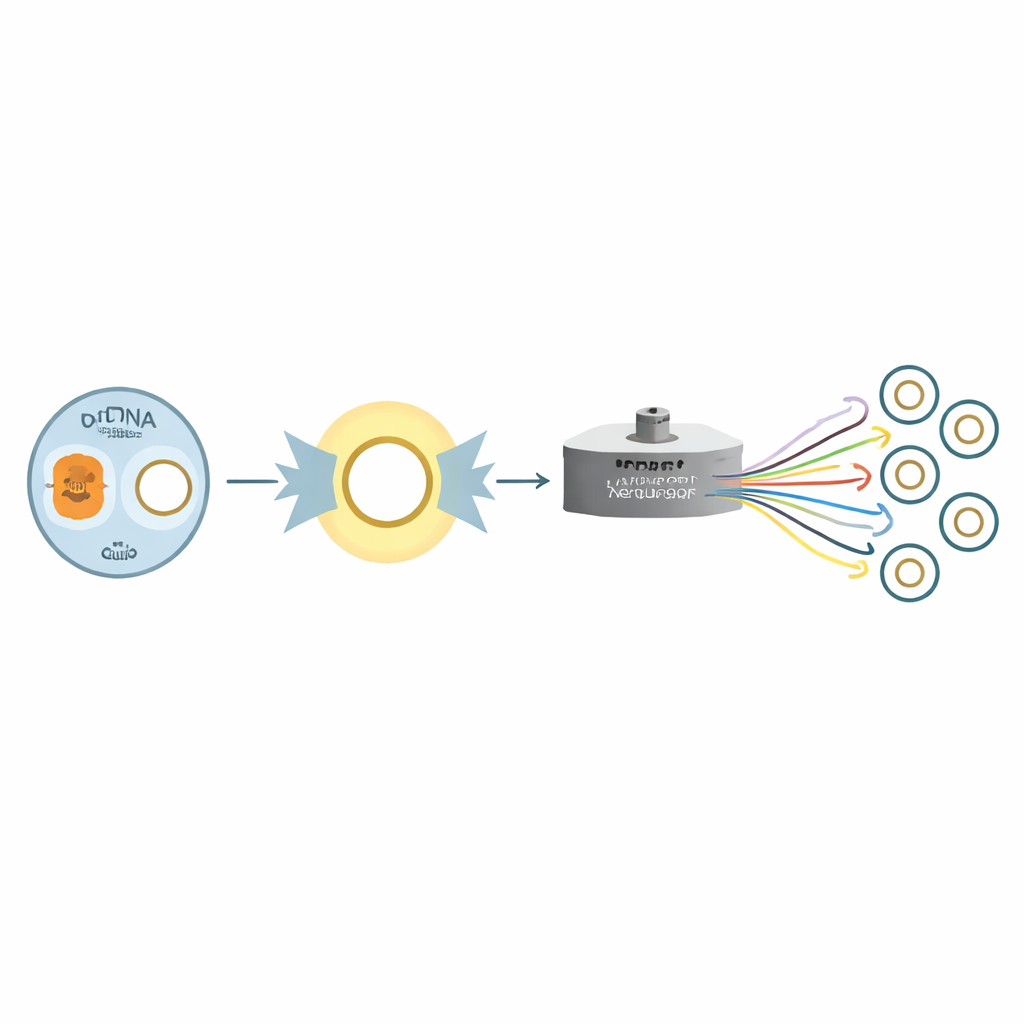
The researchers tackled this challenge using a third-generation nanopore sequencing platform combined with a clever one-piece amplification strategy. Instead of chopping the mitochondrial ring into many small fragments, they used a single pair of primers to copy almost the entire circular molecule in one long piece. This design favors real circular mitochondrial DNA over nuclear echoes and produces long reads that span the whole genome. They applied this approach to blood samples from 106 people belonging to eight families, including multi-generation households and more complex patterns such as half‑siblings, creating a rare dataset in which maternal relationships are known and can be checked.
Building and Checking a Family-Based Reference Set
After sequencing, the team put the data through a transparent, step-by-step analysis pipeline. They filtered out reads that were too short or too long, checked overall quality, and aligned the remaining sequences to a standard mitochondrial reference. Coverage of the mitochondrial genome reached 100 percent in all individuals, with very high mapping rates. They then used specialized software to identify variants, assign mitochondrial lineages (haplogroups), and reconstruct each person’s full mitochondrial sequence. Because the samples came from real families, the scientists could test whether mothers and their children carried matching mitochondrial patterns. In 73 out of 74 maternal lines, the assigned haplogroups agreed with the recorded family relationships, and the one mismatch likely reflected a labeling error rather than a biological surprise.
Keeping an Eye on Hidden Sources of Error
To make sure misleading nuclear echoes were not corrupting the results, the researchers also aligned the long reads to the full human genome and looked for reads that hit both mitochondrial and nuclear locations. Such events were rare and fell mainly in known NUMT regions, supporting the idea that their strategy greatly reduced this source of confusion. They further checked for large structural changes in the mitochondrial genome and found none above their detection threshold, consistent with the expected stability of this DNA in healthy individuals. At the same time, they cautioned that the underlying sequencing technology still has a modest error rate, and that ultra-rare variants and very long nuclear echoes may remain hard to distinguish without additional confirmation.
What This Means for Future Studies
In the end, this work does not claim to solve every technical hurdle in mitochondrial genetics, but it does provide something researchers have lacked: a well-documented, family-based collection of full-length mitochondrial genomes produced with a modern long-read platform. Because the data are openly shared along with detailed methods and quality checks, other scientists can use this resource to test new analysis tools, explore how mitochondrial mutations appear across generations, refine ancestry inference, or benchmark forensic methods. For non-specialists, the takeaway is that we are getting better at reading this tiny maternal thread of DNA accurately and responsibly, opening new windows onto health, history, and identity.
Citation: Liu, Y., Yang, Q., Xuan, Y. et al. A full-length mtDNA dataset for studying genetic variations across generations and complex family structures. Sci Data 13, 442 (2026). https://doi.org/10.1038/s41597-026-06824-0
Keywords: mitochondrial DNA, maternal inheritance, family pedigrees, long-read sequencing, forensic genetics