Clear Sky Science · en
A single-cell transcriptomic dataset profiling traumatic brain injury and NeuroD1-based gene therapy in mice
Why Brain Injuries Matter to All of Us
Every year, millions of people suffer blows to the head from falls, crashes, sports, or combat. These traumatic brain injuries can leave lasting problems with memory, movement, and mood, and today’s treatments mostly manage symptoms rather than truly repairing the brain. This study explores a promising gene-based approach in mice that aims not just to limit damage, but to help the brain rebuild itself from within, and it maps these changes cell by cell to create a public resource for future therapies.
Looking Inside the Injured Brain
When the brain is injured, it does not simply bruise like a muscle. A complex chain reaction unfolds: nerve cells die, blood flow is disturbed, and support cells called astrocytes and immune cells rush in. Astrocytes normally help feed neurons and keep brain signaling in balance, but after trauma they can form a dense scar and fuel long-lasting inflammation. The authors used a controlled stab wound to the cortex of mice to mimic certain forms of penetrating brain injury, then examined how every major cell type in the affected tissue responded over time. They applied a technique called single-cell RNA sequencing, which reads which genes are switched on in tens of thousands of individual cells, allowing a detailed census of how the brain’s cellular community changes after injury.
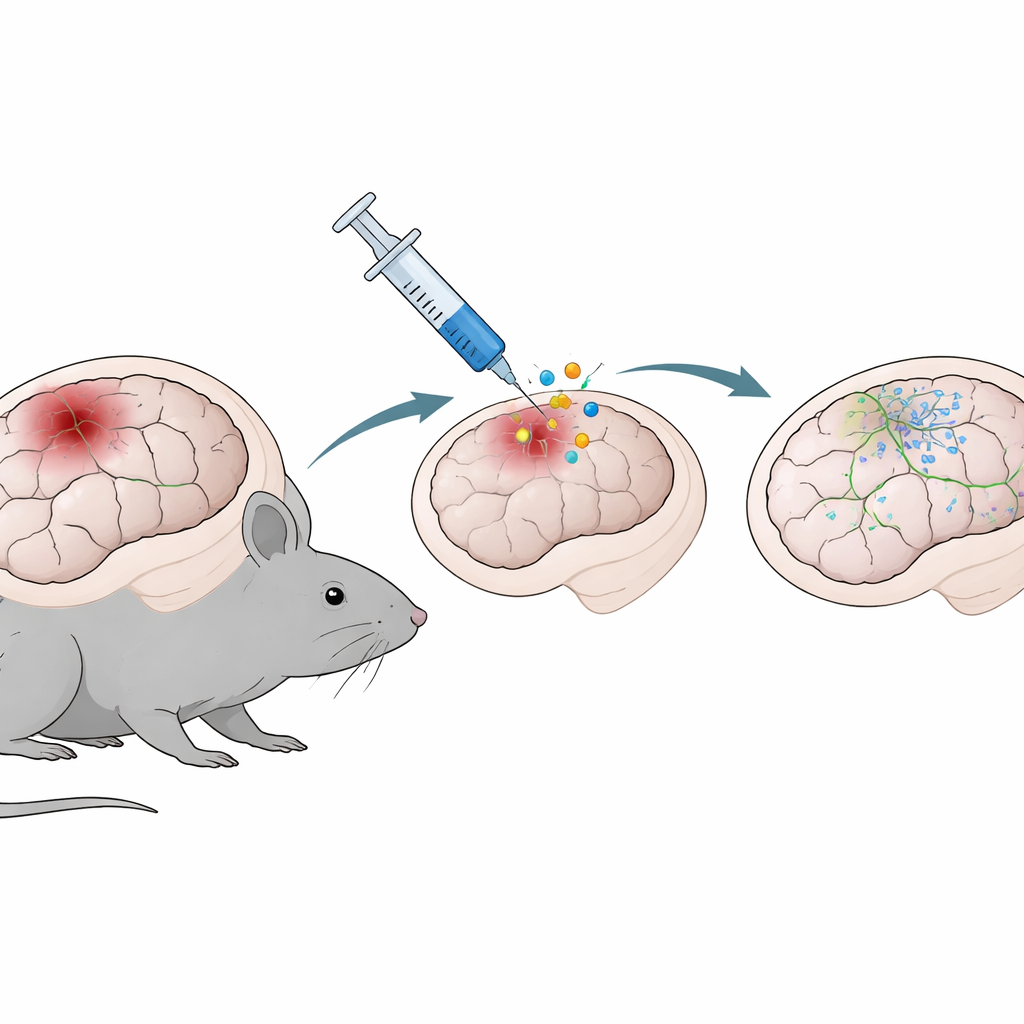
A Gene Therapy That Recruits Brain Support Cells
Because astrocytes are abundant and present right at the site of injury, they are attractive targets for repair strategies. The team tested a gene therapy based on NeuroD1, a gene known to push cells toward a neuron-like state. Using a harmless viral carrier injected into the damaged cortical area three days after injury, they delivered either a neutral marker (GFP) or NeuroD1 into astrocytes. When the brains were examined one and two weeks later, mice receiving NeuroD1 showed smaller tissue cavities where damage had occurred and fewer activated immune cells called microglia around the lesion. In other words, the therapy not only reduced visible structural damage but also calmed the local inflammatory response.
Tracking Cell Types One by One
To understand what lay beneath these improvements, the researchers compared three groups of cortical samples: healthy mice, injured mice given the control virus, and injured mice given the NeuroD1 virus. They sequenced more than 97,000 individual cells altogether and grouped them by their gene activity patterns into familiar brain residents, including neurons, astrocytes, oligodendrocytes (which insulate nerve fibers), microglia, and cells lining blood vessels and brain cavities. Injury alone shifted this balance toward more astrocytes and microglia and fewer neurons and myelin-forming cells, reflecting scarring and inflammation. With NeuroD1 treatment, this skew began to reverse: the fractions of neurons, oligodendrocytes, and choroid plexus epithelial cells increased, while astrocytes and microglia became less dominant in the injured zone.
The Hidden Work of Astrocyte Subtypes
Astrocytes turned out not to be a single uniform population. By re-analyzing them separately, the team identified seven distinct astrocyte subclusters, each with its own gene signature and behavior across the three conditions. Some subclusters were common in healthy tissue but almost vanished after injury, while others appeared only after trauma. In injured brains treated with the control virus, several astrocyte groups turned on programs involved in building and reshaping synapses—the junctions where neurons communicate—while dialing down genes tied to energy production in mitochondria. This pattern suggested that, after injury, astrocytes were pushing abnormal wiring changes while running on reduced metabolic power.
How NeuroD1 Rebalances Cell Energy and Wiring
NeuroD1 treatment reshaped these astrocyte subclusters in a different direction. In several injury-linked astrocyte groups, genes related to mitochondrial activity, cellular respiration, and general energy metabolism were boosted again, while genes linked to excessive synapse building and myelin remodeling were toned down. In other words, NeuroD1 seemed to restore the energy engines of these cells and temper runaway attempts at rewiring that might otherwise contribute to dysfunction. Some astrocyte subtypes that had expanded sharply after injury shrank in the presence of NeuroD1, while others associated with healthier functions increased. These fine-grained shifts provide clues about which astrocyte states are harmful and which might support repair.
What This Means for Future Brain Repair
This work does not yet offer a ready-made cure for human brain injury, but it does deliver two important advances. First, it shows in a living mammalian brain that a targeted gene therapy can lessen tissue loss and inflammation while nudging cell populations and energy use back toward a healthier state. Second, it makes available a rich, public single-cell dataset that other scientists can mine to pinpoint specific cell types, genes, and pathways that drive damage or recovery after trauma. For readers outside the lab, the key message is that the brain’s own support cells can potentially be recruited and reprogrammed to help rebuild injured circuits, moving us a step closer to treatments that restore, rather than simply stabilize, brain function after a traumatic blow.
Citation: Chen, R., Zhang, S., Liu, S. et al. A single-cell transcriptomic dataset profiling traumatic brain injury and NeuroD1-based gene therapy in mice. Sci Data 13, 406 (2026). https://doi.org/10.1038/s41597-026-06788-1
Keywords: traumatic brain injury, gene therapy, astrocytes, single-cell sequencing, NeuroD1