Clear Sky Science · en
The 2024 Brain Tumor Segmentation Challenge Meningioma Radiotherapy (BraTS-MEN-RT) dataset
Why mapping brain tumors matters
When someone is treated for a brain tumor, doctors must aim radiation precisely: enough to kill tumor cells, but not so much that it harms healthy brain tissue. For a common type of brain tumor called a meningioma, this aiming step depends on experts tracing the tumor by hand on three-dimensional MRI scans. That work is slow, painstaking, and can vary from one specialist to another. This article describes a large new international dataset that captures how experts outline meningiomas for radiotherapy, laying the groundwork for computer programs that can do this job faster and more consistently.
A common brain tumor with complex treatment
Meningiomas arise from the coverings of the brain and are the most frequent primary brain tumors in adults. Many are slow-growing and noncancerous, but others can return after surgery or behave aggressively, making radiotherapy an important part of care. Before giving radiation, clinicians must define the “target volume” on MRI: the full extent of the tumor and, in post-surgery cases, the surgical cavity where tumor cells may remain. This is more complicated than it sounds. Scar tissue, changes from surgery, metal implants, and specialized head frames used for focused radiation can distort images and make the tumor boundary hard to see, even for seasoned experts.
Building a shared picture library
To help improve this critical step, researchers from seven major centers in the United States and United Kingdom joined forces to create the BraTS-MEN-RT dataset. It contains 750 MRI exams used in real radiotherapy plans for meningioma patients, of which 570 scans and 500 expert-drawn target volumes are publicly available. The scans are contrast-enhanced 3D MRI images that closely match what doctors actually use in the planning room: they keep the original resolution and orientation and include the full head, not just the brain. Cases span a wide age range, both men and women, intact tumors and post-surgery situations, and multiple types of radiation treatment. This diversity is vital for training computer models that must work reliably across different hospitals and scanner types.
Protecting privacy while keeping what matters
Because each MRI also captures a person’s face, the team took special care to protect patient identity. They used an automated method that removes facial features from the images while leaving the skull, brain, and tumors intact. Every defaced scan was then checked slice by slice by a neuroradiologist and radiation oncologist to be sure no tumor tissue was accidentally cut away, especially for tumors near the base of the skull. If a tumor extended too far into the removed region and could not be safely restored, that case was excluded. This balance between privacy and medical usefulness is central to making large image collections safely shareable.
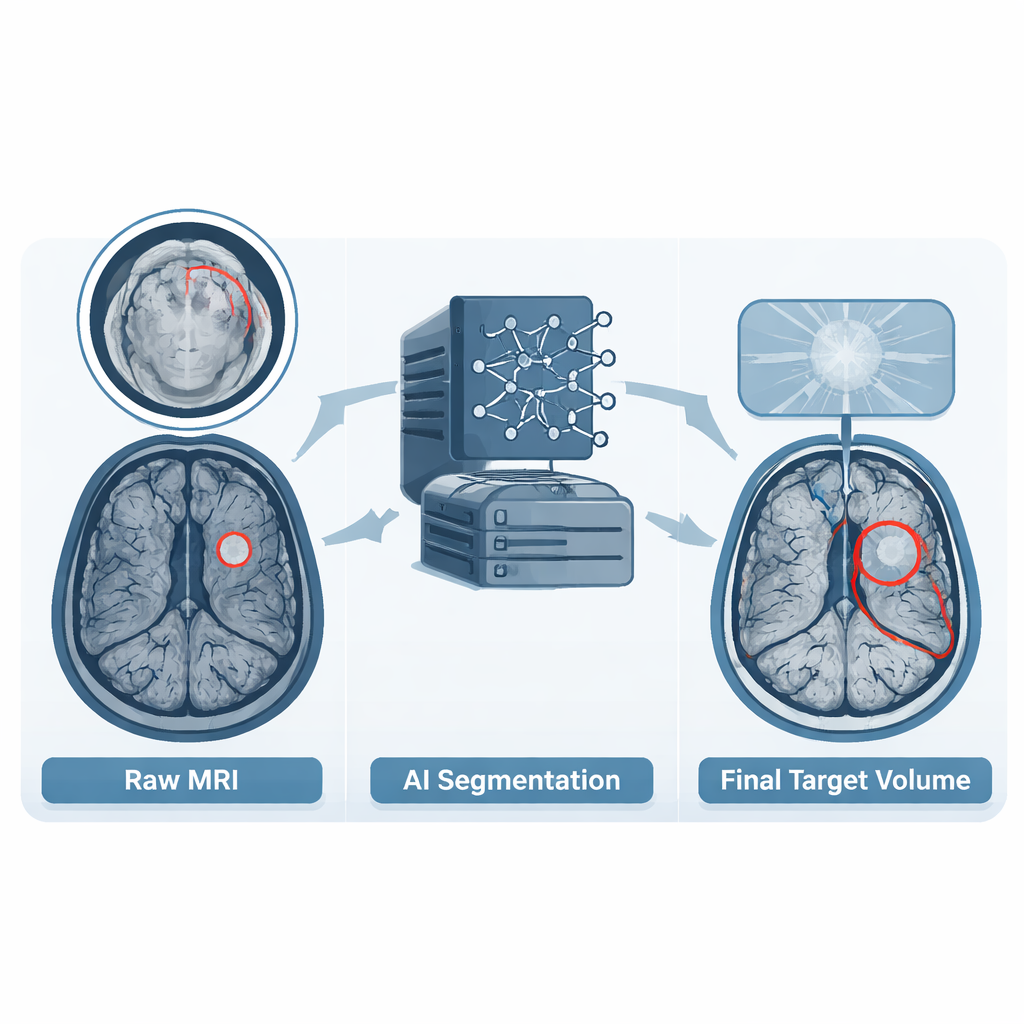
Turning expert judgment into training material
Alongside the images, the dataset includes detailed outlines of the tumor regions known as gross tumor volumes. When hospitals had already drawn these volumes in their routine planning, those outlines were used as a starting point. In other cases, or when outlines did not follow agreed guidelines, the team used a modern deep learning model to create an initial guess. A radiation oncology resident then reviewed each case slice by slice, adjusting the boundaries to match current treatment standards and adding any additional tumor deposits that had been missed. Finally, a board-certified neuroradiologist checked every outline and requested further refinements if needed. This multi-step process turned varied real-world practice into a single, consistent reference set that computers can learn from.
How this advances care for patients
By releasing this carefully curated dataset to the research community, the authors provide the missing ingredient needed to build and test algorithms that can automatically map meningioma targets on radiotherapy planning scans. If such tools prove accurate, they could save clinicians time, reduce differences between experts and institutions, and help ensure that more patients receive precisely targeted radiation. In everyday terms, the work transforms thousands of hours of specialist effort into a reusable resource that may make brain tumor treatment safer, more consistent, and more widely accessible.
Citation: LaBella, D., Schumacher, K., Mix, M. et al. The 2024 Brain Tumor Segmentation Challenge Meningioma Radiotherapy (BraTS-MEN-RT) dataset. Sci Data 13, 306 (2026). https://doi.org/10.1038/s41597-026-06649-x
Keywords: meningioma, radiotherapy, MRI, tumor segmentation, medical imaging dataset